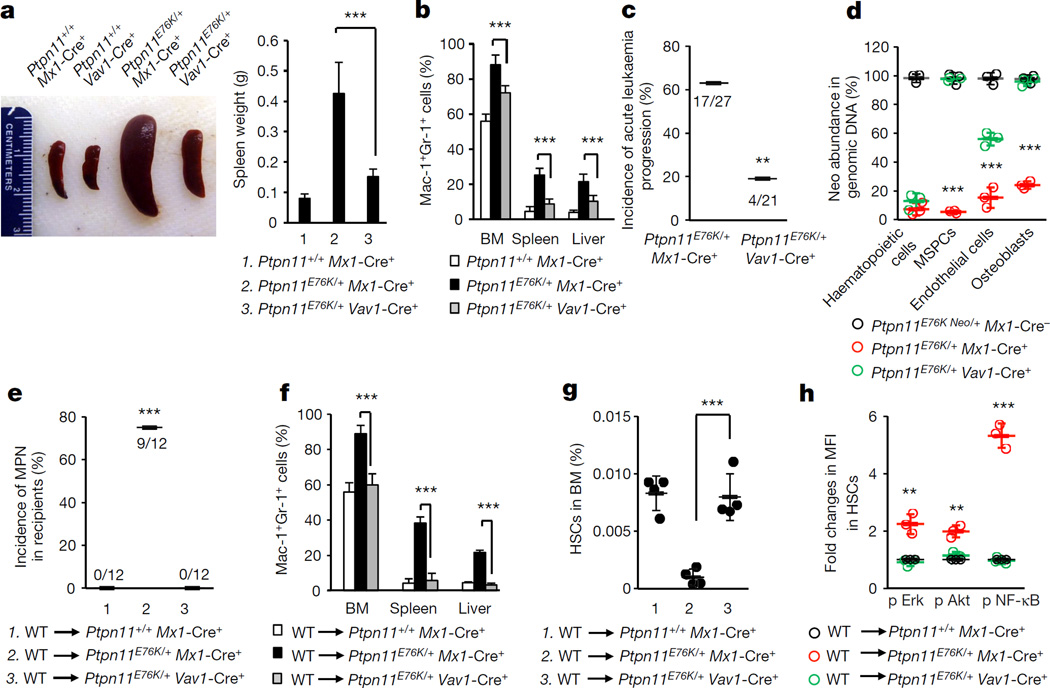
Figure 2. MPN that developed in Ptpn11E76K/+Vav1-Cre+ mice is less severe and less progressive than MPN developed in Ptpn11E76K/+Mx1-Cre+ mice.
Ptpn11E76K/+Mx1-Cre+ and Ptpn11+/+Mx1-Cre+ mice (8 weeks after pI–pC administration), along with 16-week-old Ptpn11E76K/+Vav1-Cre+ and Ptpn11+/+Vav1-Cre+ mice were killed. a, Spleen weights were determined (n = 8 mice per group). b, Cells isolated from BM, spleens and livers were assayed for Mac-1+Gr-1+ myeloid cells (n = 8 mice per group). c, Ptpn11E76K/+Mx1-Cre+ and Ptpn11E76K/+Vav1-Cre+ mice administered with pI–pC were monitored for 12 months for acute leukaemia progression. d, Haematopoietic cells (CD45+), MSPCs (Sca-l+CD140α+CD45−Ter-119−CD31−), endothelial cells (CD45−Ter-119−CD31+), and osteoblasts (Sca-l−CD140α+CD45−Ter-119−CD31−) were sorted from the BM.
The abundance of the neo cassette in genomic DNA was determined by qPCR (n = 3 mice per group). e–h, BM cells collected from wild-type BoyJ mice were transplanted into Ptpn11E76K/+Mx1-Cre+, Ptpn11+/+Mx1-Cre+ (8 weeks following pI–pC treatment), and Ptpn11E76K/+Vav1-Cre+ (16 weeks old) mice. Recipients were monitored for MPN development for 6–8 months (e). Mac-1+Gr-1+ myeloid cells in the BM, spleen and liver were examined (n = 5 mice per group) (f). The pool size (n = 4 mice per group) (g) and intracellular signalling activities (n = 3 mice per group) (h) of donor HSCs were determined 25 weeks following transplantation. Data shown in a, b, d, f–h are mean ± s.d. of all mice examined. Statistical significance was determined between Ptpn11E76K/+Mx1-Cre+ and Ptpn11E76K/+Vav1-Cre+ groups; **P < 0.01; ***P < 0.001. Source Data for this figure are available online.