FIGURE 2:
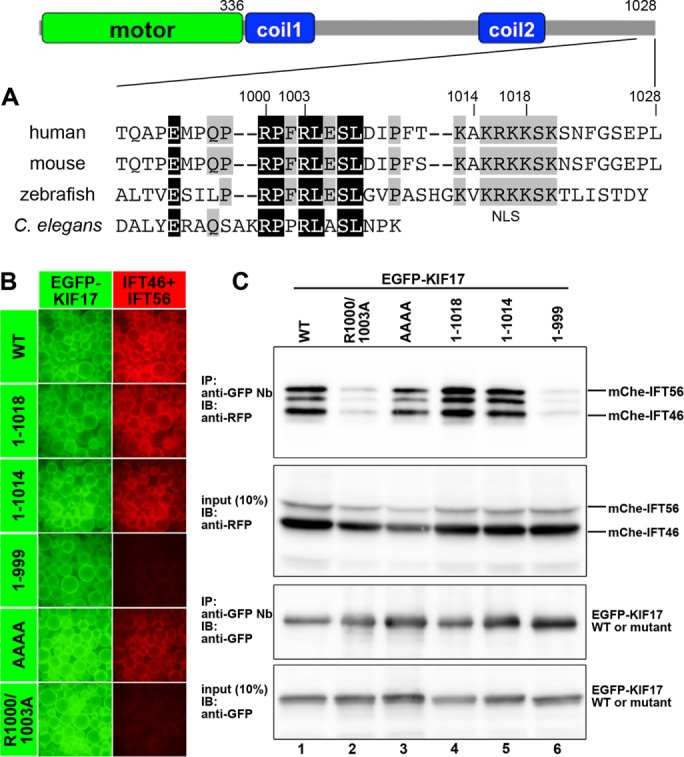
Interaction of KIF17 with the IFT46–IFT56 dimer through its conserved C-terminal sequence. (A) Schematic representation of the overall structure and domain organization of KIF17 and alignment of the C-terminal sequences of human, mouse, and zebrafish KIF17 and C. elegans OSM-3. Coil1 and Coil2 are coiled-coil regions. Residues conserved in all and three members are shown in black and gray boxes, respectively. (B, C) Involvement of the conserved C-terminal sequence of KIF17 in its interaction with the IFT46–IFT56 dimer. Lysates prepared from HEK293T cells coexpressing mChe-IFT46 and mChe-IFT56 and EGFP-KIF17(WT), EGFP-KIF17(1-1018), EGFP-KIF17(1-1014), EGFP-KIF17(1-999), EGFP-KIF17(AAAA), or EGFP-KIF17(R1000/1003A) were subjected to the VIP assay (B) or immunoblotting analysis (C).
