Fig. 6.
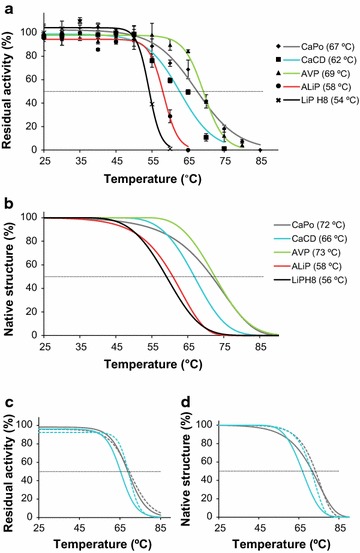
Changes in thermal stability. a, b Thermal stability of the resurrected proteins and LiPH8 estimated from enzyme activity (a) and secondary structure loss (b). The T 50 and T m values are provided in the legends. Alternative CaPo-bis and CaPo-tris showed 67 and 63 °C T 50 and 69 and 66 °C T m values, respectively. c, d Mn2+ (1 mM) addition during incubation (dashed lines) slightly increased the T 50 (c) and T m(d) values of the CaPo (gray) and CaCD (blue) ancestors. Inactivation was measured after 10 min at pH 5.5, using 2.5 mM ABTS (a) or 6 mM Mn2+ (c) as substrate. Secondary structure loss (b, d) was estimated by circular dichroism at 222 nm
