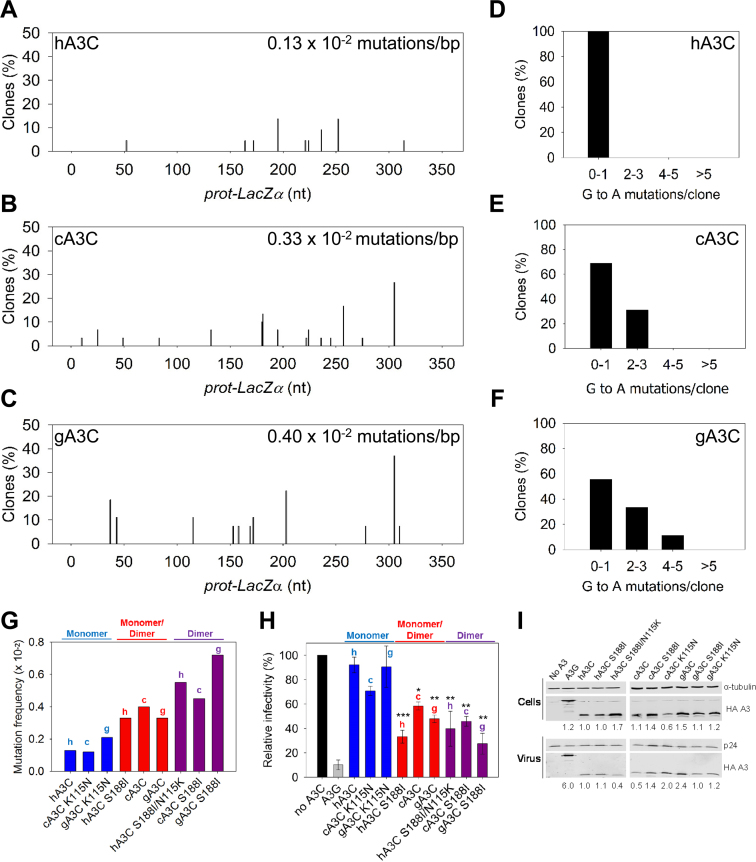
Figure 5.
Monomeric A3C induces lower levels of mutagenesis than dimeric A3C. (A–G) An in vitro HIV replication assay was utilized to determine the enzymes abilities to catalyze deaminations during proviral DNA synthesis. (A–C) Spectra of mutations are plotted as the percentage of clones containing a G→A mutation at a particular location (nt) in the 368 nt prot-lacZα construct for (A) hA3C, (B) cA3C or (C) gA3C. (D–F) Histograms illustrate the number of mutations that can be induced by (D) hA3C, (E) cA3C or (F) gA3C within individual clones. (G) Summarized G→A mutation frequency for A3C monomers (hA3C, cA3C K115N, gA3C K115N), monomers/dimers (hA3C S188I, cA3C, gA3C), and dimers (hA3C S188I/N115K, cA3C S188I, gA3C S188I). The graph denotes whether the A3C is from human (h), chimpanzee (c) or gorilla (g). Individual spectra and clonal mutation frequencies not included in Figure 5 are in Supplementary Figure S6. (H) HIV Δvif infectivity was measured by β-galactosidase expression driven by the HIV-1 5΄LTR from HeLa CD4+ HIV-1 LTR-β-gal cells infected with HIV Δvif that was produced in the absence or presence of A3G or A3C orthologs. Relative decrease in virus infectivity is shown for A3G, A3C monomers (hA3C, cA3C K115N, gA3C K115N), A3C monomers/dimers (hA3C S188I, cA3C, gA3C) and A3C dimers (hA3C S188I/N115K, cA3C S188I, gA3C S188I). The graph denotes whether the A3C is from human (h), chimpanzee (c) or gorilla (g). Results normalized to the no A3 condition are shown with the Standard Deviation of the mean calculated from at least three independent experiments. Statistical significance of HIV Δvif restriction for each A3C ortholog was determined in comparison to the monomer form (hA3C, cA3C K115N or gA3C K115N). Designations for significant difference of values were ***P ≤ 0.001, **P ≤ 0.01 or *P ≤ 0.05. (I) Immunoblotting for the HA tag was used to detect A3 enzymes expressed in cells and encapsidated into HIV Δvif virions. The cell lysate and virion loading controls were α-tubulin and p24, respectively. Quantification of the relative amount of A3 was normalized to hA3C.