Abstract
The aim of this study was to determine the distribution of metallo-β-lactamase-producing Pseudomonas aeruginosa in Japan and to investigate the molecular characteristics of resistance gene cassettes including the gene encoding this enzyme. A total of 594 nonduplicate strains of P. aeruginosa isolated from 60 hospitals throughout Japan in 2002 were evaluated. This study indicated that although the prevalence of imipenem-resistant P. aeruginosa has not increased compared to that found in previous studies, clonal distribution of the same strain across Japan is evident.
Class A, B, and D β-lactamases, as defined by Ambler et al., can hydrolyze carbapenems (1, 9). In particular, class B β-lactamases, termed metallo-β-lactamases, are an increasingly serious clinical problem because they have a very broad substrate profile that includes penicillins, expanded-spectrum cephalosporins, and carbapenems and excludes only monobactams, such as aztreonam. It has been reported that IMP-1 metallo-β-lactamase-producing Serratia marcescens was first isolated in Japan in 1991 (10). Recently, metallo-β-lactamase-producing Pseudomonas aeruginosa and S. marcescens probably have the highest incidence of isolation in Japan (7).
Most metallo-β-lactamase genes are located on integrons, which are genetic elements containing gene cassettes that can facilitate their spread and mobilize the genes to other integrons or to other sites. The gene cassettes often encode clinically important antibiotic resistance genes, including those encoding β-lactamases such as extended-spectrum β-lactamases and carbapenemases, and also aminoglycoside-modifying enzymes (12).
Little is known about the distribution of the clone(s) that produces metallo-β-lactamases in Japan. Therefore, we conducted a surveillance study covering a wide geographic area with the aim of determining the distribution of metallo-β-lactamase producers in Japan and to investigate the molecular characteristics of the resistance gene cassettes that included the gene encoding a metallo-β-lactamase.
A total of 594 nonduplicate strains of P. aeruginosa isolated from 60 hospitals throughout Japan in the year 2002 were evaluated. The susceptibility of P. aeruginosa to several antibiotics was measured with the Etest strip, and the strains were stored on Casitone medium (Eiken Chemical Co. Ltd., Tokyo, Japan) (data not shown). After 6 months, the antibiotic susceptibility of these isolates was reassessed by the National Committee for Clinical Laboratory Standards broth microdilution method with cation-adjusted Mueller-Hinton broth (Difco, Detroit, Mich.). The isolates were screened for the presence of metallo-β-lactamase by a double-disk synergy test reported by Arakawa et al. (2). Integron analysis was performed by PCR mapping (5′-conserved segment intI to 3′-conserved segment qacEΔ1) of the typical antibiotic resistance genes and integron with specific primer sets (Table 1). The specificity of the primer sets for blaIMP-1-like and blaVIM-2-like gene was confirmed with positive-control strains producing IMP-1 or VIM-2 metallo-β-lactamase. The specificity of amplicons obtained by specific primer sets (aacA4, aadA1, aadA2, and blaOXA-2) was also partially verified with the automatic sequencer ABI Prism 310 genetic analyzer (Applied Biosystems/Perkin-Elmer Biosystems). PCR with Ex Taq polymerase (Takara Bio, Inc., Tokyo, Japan) were carried out by standard methodology (13). pulsed-field gel electrophoresis analysis was performed by a modified method of the standard protocol (6). The restriction enzyme used was SpeI (15). By use of the dendrogram, isolates with a genetic relatedness of >80% were considered to represent the same pulsed-field gel electrophoresis type (4).
TABLE 1.
Nucleotide sequences of PCR primers used in this study
| Genea | Primer sequence (5′ to 3′) | Tm (°C) | Reference |
|---|---|---|---|
| intA(S) | ATC ATC GTC GTA GAG ACG TCG G | 67.4 | 11 |
| intB(AS) | GTC AAG GTT CTG GAC CAG TTG C | 66.9 | 11 |
| blaIMP-1(S) | CTA CCG CAG CAG AGT CTT TG | 62.7 | This study |
| blaIMP-1(AS) | AAC CAG TTT TGC CTT ACC AT | 59.9 | This study |
| blaVIM-2(S) | AAA GTT ATG CCG CAC TCA CC | 63.9 | This study |
| blaVIM-2(AS) | TGC AAC TTC ATG TTA TGC CG | 64.5 | This study |
| aacA4(S) | GAC CTT GCG ATG CTC TAT GAG TGG CTA AAT | 73.0 | This study |
| aacA4(AS) | TTC GCT CGA ATG CCT GGC GTG TT | 76.9 | This study |
| aadA1(S) | TGA TCG CCG AAG TAT CGA CTC | 66.3 | This study |
| aadA1(AS) | CCT TGG TGA TCT CGC CTT TC | 65.8 | This study |
| aadA2(S) | TTC GAA CCA ACT ATC AGA GGT GCT AA | 67.4 | This study |
| aadA2(AS) | AAA GCG AAT AAA TTC TTC CAA GTG ATC T | 66.4 | This study |
| blaOXA-2(S) | CAA TCC GAA TCT TCG CGA TAC TT | 66.9 | This study |
| blaOXA-2(AS) | AAG TAT CGC GAA GAT TCG GAT TG | 66.9 | This study |
| qacEΔ1 | CTC TCT AGA TTT TAA TGC GGA TG | 60.6 | This study |
(S), sense; (AS), antisense.
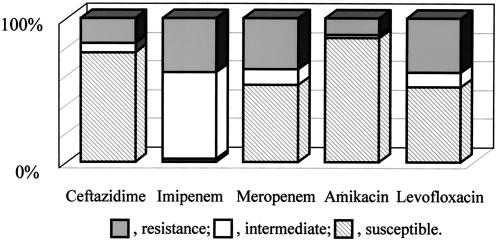
Eighty-eight (15%) of 594 isolates were not susceptible (MIC ≥ 8 mg/ml) to imipenem. Among 88 isolates, 88 (100%), 21 (24%), 41 (47%), 12 (14%), and 42 (48%) were not susceptible to imipenem, ceftazidime, meropenem, amikacin, and levofloxacin, respectively (Fig. 1). Screening of metallo-β-lactamase producers was carried out for these isolates by the double-disk synergy test. Eleven (1.9%) of 594 isolates were found to produce metallo-β-lactamase. Ten of these isolates were IMP-1-like, and the other was a VIM-2-like metallo-β-lactamase producer.
FIG. 1.
Antimicrobial susceptibilities of imipenem-nonsusceptible P. aeruginosa isolates.
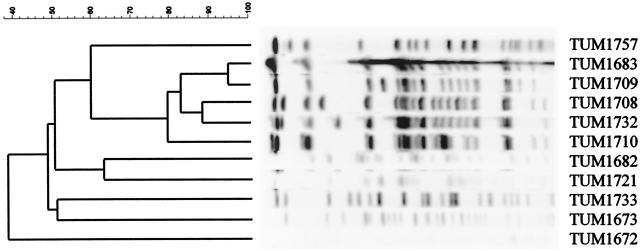
The type of metallo-β-lactamase gene was also confirmed by PCR. The genetic relatedness of these isolates was also evaluated by pulsed-field gel electrophoresis as described above (Fig. 2, Table 2). Strains TUM1683, TUM1708, TUM1709, TUM1710, and TUM1732 had related electrophoresis chromosomal DNA banding patterns, whereas other strains (TUM1672, TUM1673, TUM1682, TUM1721, TUM1733, and TUM1757) showed different banding patterns. Strain TUM1708, TUM1709, and TUM1710 were isolated from same hospital, suggesting nosocomial spread. Interestingly, although strains TUM1683, TUM1708 (or TUM1709 and TUM1710), and TUM1732 has been isolated in different hospitals, Kawasaki, Saitama, and Nara, respectively, these isolates had related patterns. Since the distance from Okayama to Saitama and from Saitama to Nara is about 800 and 400 km, respectively, the results observed suggested clonal spread of metallo-β-lactamase-producing strains.
FIG. 2.
Pulsed-field gel electrophoresis profiles obtained with SpeI chromosomal digestion of metallo-β-lactamase-carrying P. aeruginosa. The second through sixth lanes contained related strains TUM1683, TUM1709, TUM1708, TUM1732, and TUM1710, respectively. Lanes first and seventh to eleventh lanes contained unrelated strains TUM1757, TUM1682, TUM1721, TUM1733, TUM1673, and TUM1672, respectively.
TABLE 2.
Characteristics of blaIMP-containing non-imipenem-susceptible P. aeruginosa isolates
| Strain | Hospital no. | Material | Type of enzyme | Patternb | Integron structurec | MIC (μg/ml)a
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CAZ | IPM | MEM | LVX | AZT | AMK | NET | GEN | KAN | ABK | ||||||
| TUM1672 | 1 | Urine | VIM-2-like | A | I | 64 | >128 | >128 | 16 | 32 | 0.06 | 0.5 | 0.5 | 8 | 0.06 |
| TUM1673 | 1 | Sputum | IMP-1-like | B | II | >128 | 8 | 32 | 16 | 8 | 64 | >128 | 4 | >128 | 16 |
| TUM1682 | 2 | Sputum | IMP-1-like | C | III | >128 | 64 | >128 | 32 | 32 | 32 | >128 | 2 | >128 | 2 |
| TUM1683 | 2 | Sputum | IMP-1-like | D | IV | >128 | 64 | >128 | 32 | 64 | 16 | >128 | 2 | >128 | 2 |
| TUM1708 | 3 | Urine | IMP-1-like | D | IV | >128 | 64 | >128 | 32 | 32 | 32 | >128 | 4 | >128 | 4 |
| TUM1709 | 3 | Urine | IMP-1-like | D | IV | >128 | 64 | >128 | 32 | 32 | 32 | >128 | 4 | >128 | 2 |
| TUM1710 | 3 | Urine | IMP-1-like | D | IV | >128 | 64 | >128 | 32 | 64 | 32 | >128 | 2 | >128 | 4 |
| TUM1721 | 4 | Urine | IMP-1-like | E | V | >128 | 64 | >128 | 32 | 32 | 32 | >128 | >128 | >128 | 64 |
| TUM1732 | 5 | Urine | IMP-1-like | D | IV | >128 | 64 | >128 | 32 | 128 | 32 | >128 | 4 | >128 | 2 |
| TUM1733 | 5 | Pus | IMP-1-like | F | VI | >128 | 64 | >128 | 64 | 32 | 2 | >128 | >128 | >128 | 1 |
| TUM1757 | 6 | Sputum | IMP-1-like | G | VII | >128 | 64 | >128 | 16 | 16 | 32 | >128 | 1 | >128 | 16 |
CAZ, ceftazidime; IPM, imipenem; MEM, meropenem; LVX, levofloxacin; AZT, aztreonam; AMK, amikacin; NET, netilmicin; GEN, gentamicin; KAN, kanamycin; ABK, arbekacin.
PEGE profiles obtained with Spe1 chromosomal digestion of P. aeruginosa carrying a metallo-β-lactamase gene as recommended by Tenover et al. (15).
Integron structures possessed by each gene as mentioned in the text. I, blaVIM-2-like, aacA4 and aadA2; II, blaIMP-1-like, aadA1 and orfG; III, blaIMP-1-like, aadA1 and unknown gene; IV, blaIMP-1-like, aadA1 and unknown gene; V, blaIMP-1-like, aacA4, aadA1 and blaOXA-2; VI, blaIMP-1-like, aacA4; VII, only blaIMP-1-like gene.
Several researchers have reported an incidence of metallo-β-lactamase-producing P. aeruginosa of between 0.4 and 1.3% in Japan from 1992 to 2002 (5, 7, 14, 16). In this study, we isolated 1.9% metallo-β-lactamase-producing P. aeruginosa strains from geographically diverse regions in Japan. We suggest that the incidence of metallo-β-lactamase-possessing P. aeruginosa has not increased during the past decade. However, the same clone of metallo-β-lactamase-carrying P. aeruginosa has now spread throughout Japan.
It has been reported that genetic analysis of blaIMP-1 revealed features typical of an integron-located gene (9). The detection of a type 1 integron was confirmed in 11 strains. In these strains, blaIMP-1-like or blaVIM-2-like genes were located immediately downstream of the IntI1 integrase gene. However, these isolates possessed a variety of gene cassettes, such as the aacA4 aminoglycoside 6′-N-acetyltransferase gene and aadA1 and aadA2 aminoglycoside adenyltransferase genes between the metallo-β-lactamase gene and qacΔE1. Therefore, these isolates are likely resistant not only to β-lactams but also to aminoglycosides. Interestingly, strain TUM1721 possessed not only the blaIMP-1-like genes aacA4 and aadA1 but also an OXA-type β-lactamase gene on the integron gene cassette.
Little is known about optimal chemotherapy for infection due to metallo-β-lactamase-producing P. aeruginosa. To detail the antibiotic susceptibility of P. aeruginosa possessing a metallo-β-lactamase, the MICs of several antibiotics were evaluated (Table 2). All of the isolates were resistant to ceftazidime, meropenem, and levofloxacin. Ten of the 11 were resistant to imipenem and netilmicin, nine were resistant to aztreonam, and eight were not susceptible to amikacin. Bellais et al. reported that chemotherapy with high aztreonam doses effectively reduced viable cells of a metallo-β-lactamase-producing strain of P. aeruginosa in a rat pneumonia model (3). In general, although metallo-β-lactamases do not hydrolyze aztreonam, 9 of 11 isolates were resistant to aztreonam in this study (MIC ≥ 32 μg/ml). On the other hand, arbekacin was found to suppress the growth of some isolates in this study. In Japan, arbekacin, which has fewer side effects than vancomycin, has been used against methicillin-resistant Staphylococcus aureus (8). Recently, arbekacin-resistant P. aeruginosa possessing the 16S rRNA methylase gene rmtA was isolated in Japan (17). However, the incidence of these isolates is still low (0.8%, 9 of 1,113 clinical isolates). Therefore, arbekacin could be used as treatment against metallo-β-lactamase-possessing P. aeruginosa.
In conclusion, this study indicates that although the prevalence of metallo-β-lactamase-producing P. aeruginosa has not increased, this pathogen has spread from a single source to a wide geographic area of Japan. Further surveillance and monitoring of multidrug-resistant P. aeruginosa should be a high priority.
Acknowledgments
This study was supported by grants from the Ministry of Health, Labor and Welfare of Japan during 2003 (H15-Iyaku-003 and H15-shinkou-009). S.K. was supported by a grant from the Society of Japanese Pharmacopoeia.
We thank Kenneth S. Thomson, Creighton University School of Medicine, for useful advice. We also thank Kunimoto Hotta, National Institute of Infectious Diseases, for helpful discussions.
REFERENCES
- 1.Ambler, R. P. 1980. The structure of β-lactamases. Phil. Trans R. Soc. Lond. B Biol. Sci. 289:321-331. [DOI] [PubMed] [Google Scholar]
- 2.Arakawa, Y., N. Shibata, K. Shibayama, H. Kurokawa, T. Yagi, H. Fujiwara, and M. Goto. 2000. Convenient test for screening metallo-β-lactamase-producing gram-negative bacteria by using thiol compounds. J. Clin. Microbiol. 38:40-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bellais, S., O. Mimoz, S. Leotard, A. Jacolot, O. Petitjean, and P. Nordmann. 2002. Efficacy of β-lactams for treating experimentally induced pneumonia due to a carbapenem-hydrolyzing metallo-β-lactamase-producing strain of Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 46:2032-2034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Duck, W. M., C. D. Steward, S. N. Banerjee, J. E. McGowan, Jr., and F. C. Tenover. 2003. Optimization of computer software settings improves accuracy of pulsed-field gel electrophoresis macrorestriction fragment pattern analysis. J. Clin. Microbiol. 41:3035-3042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hirakata, Y., K. Izumikawa, T. Yamaguchi, H. Takemura, H. Tanaka, R. Yoshida, J. Matsuda, M. Nakano, K. Tomono, S. Maesaki, M. Kaku, Y. Yamada, S. Kamihira, and S. Kohno. 1998. Rapid detection and evaluation of clinical characteristics of emerging multiple-drug-resistant gram-negative rods carrying the metallo-β-lactamase gene blaIMP. Antimicrob. Agents Chemother. 42:2006-2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ishii, Y., J. Alba, S. Kimura, K. Nakashima, Y. Abe, and K. Yamaguchi. 2002. Rapid pulsed-field gel electrophoresis technique for determination of genetic diversity of Serratia marcescens. J. Infect. Chemother. 8:368-370. [DOI] [PubMed] [Google Scholar]
- 7.Kurokawa, H., T. Yagi, N. Shibata, K. Shibayama, and Y. Arakawa. 1999. Worldwide proliferation of carbapenem-resistant gram-negative bacteria. Lancet 354:955. [DOI] [PubMed] [Google Scholar]
- 8.Matsuo, H., M. Kobayashi, T. Kumagai, M. Kuwabara, and M. Sugiyama. 2003. Molecular mechanism for the enhancement of arbekacin resistance in a methicillin-resistant Staphylococcus aureus. FEBS Lett. 546:401-406. [DOI] [PubMed] [Google Scholar]
- 9.Nordmann, P., and L. Poirel. 2002. Emerging carbapenemases in Gram-negative aerobes. Clin. Microbiol. Infect. 8:321-331. [DOI] [PubMed] [Google Scholar]
- 10.Osano, E., Y. Arakawa, R. Wacharotayankun, M. Ohta, T. Horii, H. Ito, F. Yoshimura, and N. Kato. 1994. Molecular characterization of an enterobacterial metallo β-lactamase found in a clinical isolate of Serratia marcescens that shows imipenem resistance. Antimicrob. Agents Chemother. 38:71-78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rosser, S. J., and H. K. Young. 1999. Identification and characterization of class 1 integrons in bacteria from an aquatic environment. J. Antimicrob. Chemother. 44:11-18. [DOI] [PubMed] [Google Scholar]
- 12.Rowe-Magnus, D. A., and D. Mazel. 2002. The role of integrons in antibiotic resistance gene capture. Int. J. Med. Microbiol. 292:115-125. [DOI] [PubMed] [Google Scholar]
- 13.Sambrook, J., and D. W. Russell. 2001. In vitro amplification of DNA by the polymerase chain reaction, p. 8.1-8.126. In J. Sambrook and D. W. Russell (ed.), Molecular cloning: a laboratory manual, 3rd ed. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.
- 14.Senda, K., Y. Arakawa, K. Nakashima, H. Ito, S. Ichiyama, K. Shimokata, N. Kato, and M. Ohta. 1996. Multifocal outbreaks of metallo-β-lactamase-producing Pseudomonas aeruginosa resistant to broad-spectrum β-lactams, including carbapenems. Antimicrob. Agents Chemother. 40:349-353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tenover, F. C., R. D. Arbeit, R. V. Goering, P. A. Mickelsen, B. E. Murray, D. H. Persing, and B. Swaminathan. 1995. Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: criteria for bacterial strain typing. J. Clin. Microbiol. 33:2233-2239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yamasaki, K., M. Komatsu, T. Yamashita, K. Shimakawa, T. Ura, H. Nishio, K. Satoh, R. Washidu, S. Kinoshita, and M. Aihara. 2003. Production of CTX-M-3 extended-spectrum β-lactamase and IMP-1 metallo β-lactamase by five Gram-negative bacilli: survey of clinical isolates from seven laboratories collected in 1998 and 2000, in the Kinki region of Japan. J. Antimicrob. Chemother. 51:631-638. [DOI] [PubMed] [Google Scholar]
- 17.Yokoyama, K., Y. Doi, K. Yamane, H. Kurokawa, N. Shibata, K. Shibayama, T. Yagi, H. Kato, and Y. Arakawa. 2003. Acquisition of 16S rRNA methylase gene in Pseudomonas aeruginosa. Lancet 362:1888-1893. [DOI] [PubMed] [Google Scholar]