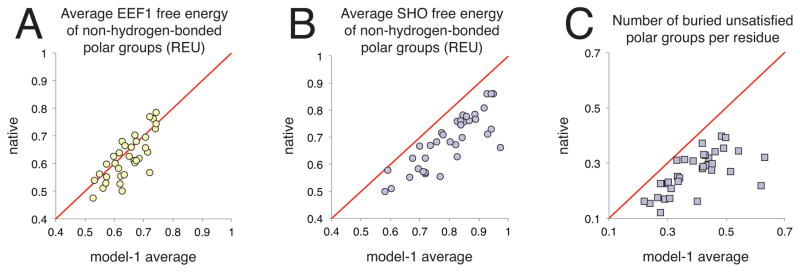
Figure 5. Applying SHO to identify native protein structures.
For each of 36 CASP11 refinement targets, quantities measured for the native structure are compared to those averaged over the “model-1” submissions from each participant. A) The average EEF1 free energy for polar groups not engaged in hydrogen bonds is lower for the native than for the average of the models in 21 of 36 cases (points below the diagonal); in the other 15 cases, the average of the models is lower than the native. B) Using SHO, the average free energy for the same set of polar groups is lower for the native structure in all 36 targets (points below the diagonal). Thus, SHO provides energetic discrimination for native versus near-native protein structures. C) Using SHO to identify buried unsatisfied polar groups, we find that these unfavorable features occur more frequently in the models than in the corresponding native structures (all points are below the diagonal): this provides the basis by which SHO discriminates energetically between native and near-native structures.