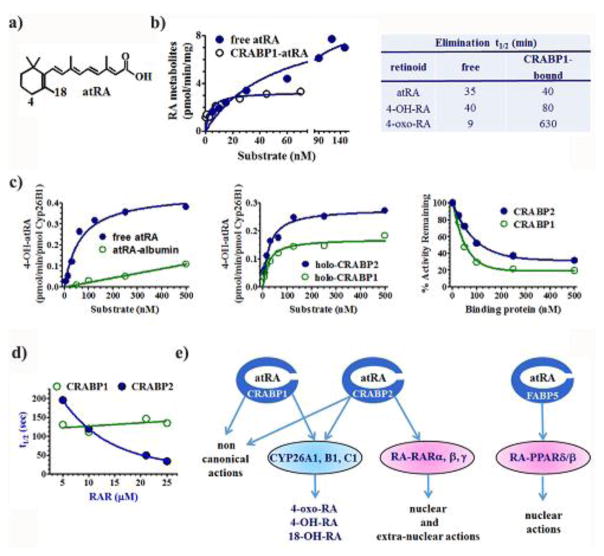
Fig. 6.
Functions of atRA BP. a) Structure of atRA designating two of the carbons oxidized by CYP26, C4 and C18. b) Michaelis-Menten kinetics revealing the differences between catabolism of free atRA vs. CRABP1-bound atRA. The reaction with BP was done with a 3/1 ratio of total CRABP1 to atRA to ensure lack of free atRA, yet enhanced the reaction rate at low concentrations of substrate. The reaction with BP-bound atRA followed typical Michaelis-Menten kinetics, whereas that of unbound atRA seemed unsaturable at concentrations that exceed the concentrations of atRA in tissues. The table shows the effects of binding atRA and two of its metabolites on their rates of catabolism, suggesting high concentrations of catabolites bound to CRABP contribute to atRA toxicity by blocking further catabolism of atRA (Fiorella & Napoli, 1991, 1994). c) Rate of 4-OH-atRA biosynthesis from free atRA or albumin bound atRA showing that not all protein-bound atRA behaves similar to CRABP-bound atRA. Michaelis-Menten kinetics illustrating protein-protein interactions between recombinant CYP26B1 and CRABP1 and 2. Impact of CRABP1 and 2 on the ability of recombinant CYP26B1 to catalyze atRA catabolism, indicating inhibition of CRP26B1 by the two atRA BP (Nelson et al., 2016). d) Effect of increasing the RAR concentration on the rate of transfer of atRA from BP. The decrease in the transfer rate indicates that only CRABP2 directly interacts with RAR (Dong et al., 1999). e) Summary of CRABP and FABP5 functions.