Abstract
Despite the benefit of insulin, blockade of autoimmune attack and regeneration of pancreatic islets are ultimate goals for the complete cure of type 1 diabetes (T1D). Long-term consumption of ω-3 polyunsaturated fatty acids (PUFAs) is known to suppress inflammatory processes, making these fatty acids candidates for the prevention and amelioration of autoimmune diseases. Here, we explored the preventative and therapeutic effects of ω-3 PUFAs on T1D. In NOD mice, dietary intervention with ω-3 PUFAs sharply reduced the incidence of T1D, modulated the differentiation of Th cells and Tregs, and decreased the levels of IFN-γ, IL-17, IL-6, and TNF-α. ω-3 PUFAs exerted similar effects on the differentiation of CD4+ T cells isolated from human peripheral blood mononuclear cells. The regulation of CD4+ T cell differentiation was mediated at least in part through ω-3 PUFA eicosanoid derivatives and by mTOR complex 1 (mTORC1) inhibition. Importantly, therapeutic intervention in NOD mice through nutritional supplementation or lentivirus-mediated expression of an ω-3 fatty acid desaturase, mfat-1, normalized blood glucose and insulin levels for at least 182 days, blocked the development of autoimmunity, prevented lymphocyte infiltration into regenerated islets, and sharply elevated the expression of the β cell markers pancreatic and duodenal homeobox 1 (Pdx1) and paired box 4 (Pax4). The findings suggest that ω-3 PUFAs could potentially serve as a therapeutic modality for T1D.
Introduction
Type 1 diabetes (T1D) is a polygenic and organ-specific autoimmune disease, in which a certain subclass of T lymphocytes is involved in executing autoimmune attacks that lead to the destruction of pancreatic β cells (1, 2). Although the current prevailing treatment of T1D is lifelong daily injection of recombinant insulin, the exogenously delivered insulin does not carry C-peptide, which has been known to provide protective effects on the microvasculature (3) and on neuronal (4) and renal (5) function. Even with proper blood glucose control, T1D patients are prone to complications such as retinopathy, neuropathy, and cardiovascular disease (6, 7). Thus far, there is no effective method of reversing autoimmunity once a patient enters the course of T1D. Research in the past decade has been focused on islet allografting (8) and stem cell–derived β cells (9), as well as immunotherapies using mAbs targeting CD3 (10), glutamic acid decarboxylase-65 Ig (GAD65-Ig) (11), and cytotoxic T lymphocyte–associated antigen-4–Ig (CTLA-4–Ig) (12). Although promising phase I and II trials were reported in patients with early onset of T1D, phase III trials of anti-CD3 and GAD65-Ig immunotherapy did not meet the primary endpoints (10, 11, 13). Similarly, CTLA4-Ig preserved C-peptide concentrations for only 9 months, despite continuous i.v. administration for 2 years (12). The limitations of islet transplantation include a shortage of donors, fibrosis of transplanted islets, and various side effects of immunosuppressive agents, making it unlikely to be a primary therapeutic approach for T1D (8, 14). Indeed, either a single immunosuppressive agent or islet allografting alone appears to be insufficient to completely control or reverse the autoimmune attack on β cells. A new paradigm that targets several pathogenic pathways is therefore sorely needed for a complete cure of T1D.
Both genetic susceptibility and environmental factors can trigger the onset of T1D. In particular, environmental factors such as viral infection and nutritional imbalance can cause dysregulation of CD4+ T cells (15), leading to the infiltration of autoreactive CD8+ T cells into pancreatic islets and, ultimately, the killing of β cells. Eventually, a subsequent wave of β cell killing results in a severe reduction of β cell function and mass (1, 15). Numerous studies have shown that alterations of multiple immune cells play major roles in the etiology of T1D, chief among which Th1, Th2, and Th17 cells and Tregs have been extensively elucidated (16). Th1 cells evoke cell-mediated immunity and phagocyte-dependent inflammation, whereas Th2 cells can cause strong antibody responses but inhibit phagocyte-independent inflammation (17, 18). The Th1-dominated responses dictate the pathogenesis of organ-specific autoimmune disorders (19). Th1 and Th17 cells mainly secrete inflammatory factors such as IFN-γ and IL-17, which, by acting alone or synergistically, can aggravate the progression of T1D (20). In contrast, Th2 cells and Tregs can antagonize autoimmune responses through cytokines such as IL-4 and IL-10 (21). The intervention of dysregulated Th cells is therefore critical to stopping autoimmune progression and the inflammatory attack against β cells.
Mounting evidence suggests that dietary supplementation of fish oil starting from infancy has the beneficial effect of alleviating autoimmune progress and T1D. A longitudinal DAISY (Diabetes Autoimmunity Study in the Young) study showed that long-term dietary intake of ω-3 polyunsaturated fatty acids (PUFAs) starting from 1 year of age is associated with a reduced risk of islet autoimmunity in children with familial T1D (22). A Norwegian case-control study in which cod liver oil was given, starting from 1 year of age, to infants with a high risk of T1D and to population control subjects also resulted in a much-reduced incidence of childhood-onset T1D (23). Recently, a multicenter, randomized, double-blind trial of docosahexaenoic acid (DHA) supplementation in infants at a high genetic risk of T1D, beginning either in the last trimester of pregnancy or in the first 5 months after birth, revealed significantly reduced inflammation as reflected in the lower levels of high-sensitivity C-reactive protein (24). Apart from this, our earlier studies showed that stable cellular production of ω-3 PUFAs could enhance insulin secretion and confer strong resistance to cytokine-induced β cell destruction (25). These studies indicated that an ω-3 PUFA–enriched-diet can have a strong influence on immune responses as well as on pancreatic islet function and prompted us to investigate the strategy of using ω-3 PUFAs as a preventative, or even therapeutic, modality to deter T1D. Specifically, we improvised a nutritional as well as a gene therapy approach to elevate circulating eicosapentaenoic acid (EPA) and DHA levels, with the goals of correcting the dysregulation of T cell differentiation, silencing the immune attack against pancreatic β cells, and regenerating β cells.
Results
Intervention with dietary ω-3 PUFAs reduces the incidence of T1D.
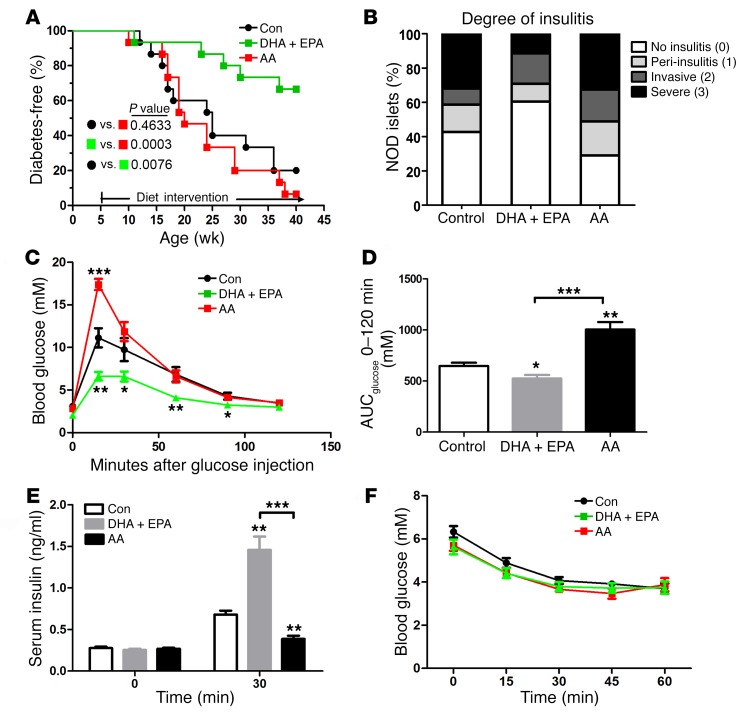
We designed a dietary intervention study, in which female NOD mice were fed a diet containing enriched EPA/DHA for 35 weeks, starting at 5 weeks of age. In addition to a control group fed a regular diet, a separate group of animals was fed a diet containing equal levels of arachidonic acid (AA, an ω-6 PUFA). The dietary intervention resulted in significant changes in fatty acid composition in serum and tissue samples (Supplemental Tables 1–3; supplemental material available online with this article; https://doi.org/10.1172/JCI87388DS1). The EPA/DHA-enriched diet resulted in a substantial increase in ω-3 PUFAs that was accompanied by a significant decrease (P < 0.0001) in ω-6 PUFAs, leading to sharply reduced ratios of ω-6/ω-3 PUFAs relative to ratios in AA diet–fed mice. Nonfasting blood glucose concentrations were used to monitor the incidence of diabetes, which was diagnosed by the presence of glucose concentrations of greater than 11.11 mmol/l for 2 consecutive weeks. No spontaneous reversal to less than 11.11 mmol/l was observed in any of the control groups (n = 15 per group). Consistent with results reported by others (26, 27), we found that 80% of female NOD mice on a regular diet developed diabetes by the age of 40 weeks. In contrast, only 33% of the mice fed an EPA/DHA-enriched diet were diabetic, which was significantly different (P = 0.0076) according to a Mantel-Cox log-rank test. Interestingly, 93% of NOD mice on the diet containing comparable levels of AA developed diabetes at the same age, although there was no significant difference between the AA intervention group and the control diet group (Figure 1A). Thus, long-term supplementation of dietary EPA/DHA reduced the incidence of T1D and delayed its onset in female NOD mice.
Figure 1. ω-3 PUFAs ameliorate the development of T1D and normalize glucose metabolism in NOD mice.
(A) Blood glucose concentrations in 3 groups of NOD mice on varied diets were monitored weekly until 40 weeks of age. Sustained hyperglycemia for 2 consecutive weeks (>11.11 mmol/l) marked the onset of disease, which was used to create a life table to determine the incidence of diabetes (n = 15/group). Statistical calculation was done using a Mantel-Cox log-rank test. (B) Sections (4-μm-thick) of pancreas from 20-week-old NOD mice were formaldehyde fixed, paraffin embedded, and stained with H&E (n =7/group). Islets were sorted into the following 4 categories on the basis of the relative degree of immune infiltration: no insulitis (0), peri-insulitis (1), invasive insulitis (2), or severe insulitis (3). Representative pancreatic sections are shown in Supplemental Figure 1. The differences in severe insulitis between DHA plus EPA group and the control group (P < 0.0001) and between the DHA plus EPA group and the AA group (P = 0.0008) were significant. The finding of no insulitis in the DHA plus EPA group was increased compared with the control (P = 0.02) and AA (P < 0.0001) groups. Statistical calculation was done using Pearson’s χ2 test. (C) Glucose tolerance tests (GTTs) in NOD mice fed a control, AA, or DHA plus EPA diet (n = 15/group) at 20 weeks of age. (D) AUC for GTTs performed in 3 groups of NOD mice fed different diets. (E) Serum insulin concentrations during the GTT at the indicated time points (n = 10/group). (F) Insulin tolerance tests (n = 10/group). (C–E) *P < 0.05, **P < 0.01, and ***P < 0.0001 versus the control group (Student’s t test). Data are representative of 2 independent experiments. All values represent the mean ± SEM.
Intervention with ω-3 PUFAs blocks the progression of immune infiltration in NOD mice.
The progression of peri-insulitis and insulitis occurs between the initiation and detection of hyperglycemia in NOD mice (28). We used H&E-stained pancreatic sections to evaluate the extent of lymphocyte infiltration into pancreatic islets isolated from 20-week-old NOD mice (16 weeks after different dietary intervention) (29) (Supplemental Figure 1). By the age of 20 weeks, the islets from the EPA/DHA-fed mice had a significantly reduced incidence of severe insulitis compared with those from mice maintained on an AA-enriched diet or a regular diet. The percentages of peri-insulitis and invasive insulitis incidence were no different among the 3 groups, suggesting that ω-3 PUFAs could not prevent the initiation of lymphocyte infiltration. Taken together, these results indicated that ω-3 PUFA supplementation in NOD mice sharply reduced the percentage of islets showing the most severe insulitis (Figure 1B).
Intervention with ω-3 PUFA can normalize glucose metabolism in NOD mice.
To study the function of β cells and the homeostasis of blood glucose, we evaluated the impact of ω-3 and ω-6 PUFAs on glucose homeostasis in NOD mice on different diets. Fasting blood glucose concentrations were indistinguishable among the 3 groups. However, during the i.p. glucose tolerance test, the NOD mice maintained on an EPA/DHA-enriched diet showed significantly lower blood glucose concentrations at all time points than did mice in the other 2 groups (Figure 1, C and D). The improved glucose tolerance in the EPA/DHA-fed mice was primarily attributed to the elevated glucose-induced insulin secretion (Figure 1E), as the insulin tolerance tests revealed no significant difference among the 3 groups of NOD mice (Figure 1F).
Modulation of CD4+ T cell differentiation by ω-3 and ω-6 PUFAs.
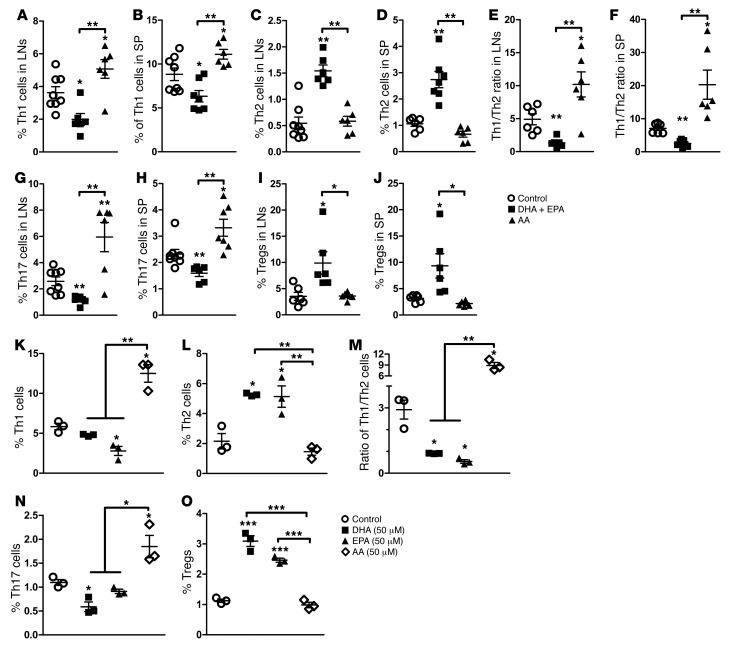
The interplay among CD4+ T cells, particularly Th1, Th2, Th17, and Tregs, as well as their secreted cytokines (30, 31) plays crucial roles in priming an immune response against pancreatic β cells (19, 32–35). The analysis of Th1 and Th2 cells in NOD mice revealed that Th1 cells significantly increased from 5.03% to 14.8%, while Th2 cells decreased from 5.65% to 2.24% after the onset of T1D in the mice fed a regular diet. Consequently, the Th1/Th2 ratio was significantly disturbed and polarized toward Th1 cells (Supplemental Figure 2, A and B). In contrast, intervention with an EPA/DHA-enriched diet reduced Th1 cell percentages and elevated the population of Th2 cells, thus rebalancing the ratio of Th1/Th2 cells (Figure 2, A–F and Supplemental Figure 3). Consistent with its role as a proinflammatory fatty acid, dietary addition of AA further boosted the population of Th1 cells, resulting in a drastically exacerbated ratio of Th1/Th2 cells (Figure 2, A–F and Supplemental Figure 3).
Figure 2. Modulation of Th cells by ω-3 and ω-6 PUFAs in vivo and in vitro.
(A–J) Percentage of IFN-γ+, IL-4+, IL-7+, and CD25+FoxP3+ Th cells in spleens and LNs of nondiabetic NOD mice fed a control, DHA plus EPA, or AA diet. NOD mice were sacrificed at 20 weeks of age, following dietary intervention, and their spleens and LNs were harvested. Each point represents an individual mouse, and the data are representative of 3 independent experiments (n =6–8/group). Representative flow cytometric images are shown in Supplemental Figure 3. (K–O) Quantification of the percentage of intracellular staining of IFN-γ+, IL-4+, IL-7+, and CD25+FoxP3+ Th cells from 10-week-old nondiabetic NOD mice. Cells were cultured for 24 hours under PMA and ionomycin stimulation in the presence of DHA, EPA, and AA (50 μM), added at the time of activation. Data are representative of 3 independent experiments (n = 3/group). Representative flow cytometric images are shown in Supplemental Figure 5. *P < 0.05, **P < 0.01, and ***P < 0.0001 versus the control group (Student’s t test). Values represent the mean ± SEM.
Dietary EPA/DHA not only corrected the imbalance of effective Th1 and Th2 cells but also reduced the proportion of Th17 cells and increased the population of Tregs. In contrast, an AA-enriched diet increased the numbers of Th17 cells and had no significant effect on Treg numbers (Figure 2, G–J and Supplemental Figure 3). We also performed a parallel analysis of some cytokines secreted by CD8+ T cells following intervention with an ω-3 or ω-6 PUFA–enriched diet. Although an ω-6 PUFA–enriched diet also markedly elevated the secretion of IFN-γ, an ω-3 PUFA–enriched diet did not induce significant changes in IFN-γ secretion from CD8+ T cells (Supplemental Figure 4). Furthermore, the levels of IL-4 secreted from CD8+ T cells were too low to be detected by FACS. Thus, CD8+ T cells were probably not the primary targets of autoimmune regulation by ω-3 PUFAs. Taken together, intervention with ω-3 PUFAs normalized the ratio of Th1/Th2 cells and had an inhibitory effect on Th1 and Th17 cells, but a stimulatory effect on Tregs in NOD mice.
Dietary ω-3 PUFAs restore cytokine secretion profiles in NOD mice.
Activated T cells produce a series of cytokines that exert many actions on T cell themselves and on other immune cells. Measurement of pro- and antiinflammatory cytokines in the blood of NOD mice showed that an EPA/DHA-enriched diet had no significant impact on proinflammatory factors such as IL-17A and TNF-α and decreased the secretion of IFN-γ and IL-6, but increased the levels of IL-4 and IL-10, which are cytokines with known immunosuppressive characteristics. In contrast, the AA-enriched diet promoted the secretion of IFN-γ, IL-17A, IL-6, and TNF-α, while it had no effect on IL-4 and IL-10 secretion (Supplemental Table 4), which was consistent with the proinflammatory nature of AA.
Direct effects of ω-3 PUFAs on CD4+ T cell differentiation in vitro.
The results described above led us to examine the direct impact of ω-3 PUFAs on CD4+ T cell differentiation in vitro. CD4+ T cells were purified from the spleens and lymph nodes (LNs) of 10-week-old female NOD mice and treated with DHA, EPA, or AA, separately. Treatment with EPA or DHA, both of which are ω-3 PUFAs, reduced the population of Th1 cells, increased Th2 and Treg numbers, and consequently balanced the Th1/Th2 ratio. However, addition of AA to the culture significantly increased Th1 and Th17 cell populations, but did not affect Th2 cell or Treg populations (Figure 2, K–O, and Supplemental Figure 5), suggesting that, although ω-6 PUFAs favored the activation of Th1 and Th17, ω-3 PUFAs could counteract the excessive production of Th1 by increasing the population of Th2 cells and Tregs. We observed similar results when we analyzed secreted cytokines in the culture media. Accordingly, EPA and DHA suppressed the production of IL-17A and significantly increased IL-4 and IL-10 secretion (Supplemental Table 5). DHA also decreased the secreted levels of IFN-γ. Incubation with AA generated the opposite effect by increasing secreted IL-17A, IFN-γ, and IL-6, while significantly reducing the amount of IL-4. Thus, ω-3 PUFAs promoted the secretion of antiinflammatory cytokines and inhibited the production of proinflammatory cytokines.
Regulation of CD4+ T cell differentiation by ω-3 PUFA metabolites.
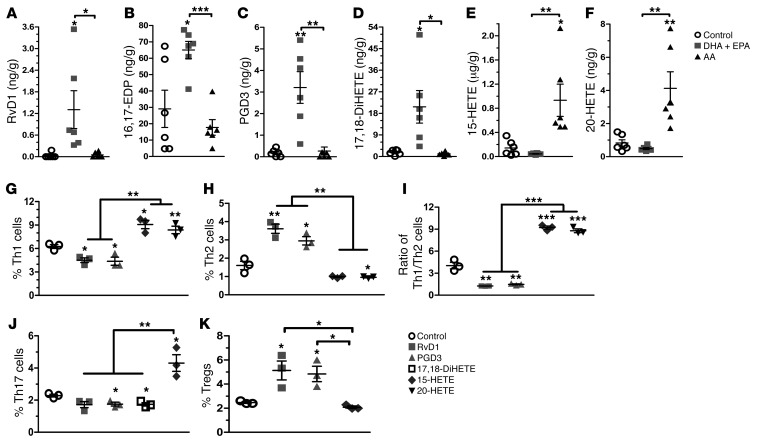
Several prominent ω-3 and ω-6 PUFA–derived eicosanoids such as prostaglandins (PGs), thromboxanes (TXs), leukotrienes (LTs), and hydroxyeicosatetraenoic acids (HETEs) are synthesized via COX and lipoxygenase (LOX) activity and can have multiple biological effects on inflammation (36, 37). Metabolomic analysis of ω-3 and ω-6 PUFA metabolites in the pancreatic samples revealed that the levels of some DHA and EPA metabolites such as resolvin D1 (RvD1); 16,17-epoxy docosapentaenoicacid (16,17-EDP); prostaglandin D3 (PGD3); and 17,18-dihydroxy-5Z,8Z,11Z,14Z-eicosatetraenoic acid (17,18-DiHETE) were significantly higher (P < 0.05) than those in mice fed a regular diet or an AA-enriched diet. On the other hand, the levels of the AA-derived metabolites 15-HETE, 20-HETE, and 15-oxo-5Z,8Z,11Z,13E-eicosatetraenoic acid (15-oxo-ETE) were higher in the samples from mice fed an AA-enriched diet (Figure 3, A–F and Supplemental Figure 6). Evaluation of the biological effects of these metabolites on CD4+ T cell differentiation indicated that DHA-derived RvD1 and EPA-derived PGD3 sharply inhibited the differentiation into Th1 cells, while promoting Th2 cell and Treg populations. In contrast, 15-HETE significantly elevated Th17 cell numbers (Figure 3, G–K and Supplemental Figure 7). The results suggested that some of the EPA- and DHA-derived metabolites were at least partly responsible for balancing CD4+ T cell differentiation.
Figure 3. Diverse metabolic production of ω-3 PUFAs regulates Th cell differentiation.
(A–F) Presence of different eicosanoids from ω-3 or ω-6 PUFAs in pancreas samples from NOD mice fed a control, DHA plus EPA, or AA diet (n = 6/group). (G–K) Quantification of the percentage of intracellular staining of IFN-γ+, IL-4+, IL-7+, and CD25+ FoxP3+ Th cells from 10-week-old nondiabetic NOD mice. Cells were cultured for 24 hours under PMA and ionomycin stimulation in the presence of distinctive PUFA metabolites (0.1 μg/ml), added at the time of activation (n = 3/group). Representative flow cytometric images are shown in Supplemental Figure 7. *P < 0.05, **P < 0.01, and ***P < 0.0001 versus the control group (Student’s t test). Data are representative of 3 independent experiments. All values represent the mean ± SEM.
ω-3 PUFA–regulated CD4+ T cell differentiation via mTOR complex 1 activity.
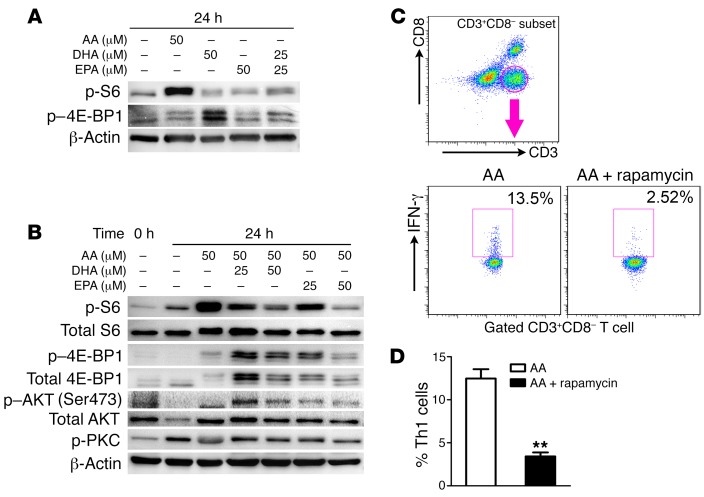
Our previous studies showed that ω-6 and ω-3 PUFAs had opposite effects on mTOR kinase (38), a critical regulator of Th cell activation and fate decision (39, 40). To test the involvement of this pathway in the regulation of T cell differentiation by PUFAs, we treated the isolated CD4+ T cells from spleens of NOD mice with AA (ω-6), DHA, or EPA (ω-3) for 24 hours. The phosphorylated form of S6 (Ser235/236) and dephosphorylated form of 4E-BP1 are indicative of the activation of mTOR complex 1 (mTORC1) (41). The treatment with AA alone strongly elevated phosphorylated S6 (p-S6) levels. However, EPA or DHA alone (or in combination) only had a small or marginal effect on S6 phosphorylation (Figure 4A). To further investigate the cross-regulation of mTORC1 activity by different PUFAs, we treated CD4+ T cells with different combinations of AA plus EPA or DHA for 24 hours. Although AA elevated p-S6 levels, the addition of EPA or DHA to the AA-containing medium ameliorated the AA-induced increase in p-S6. Consistent with this observation, p–4E-BP1 levels were higher in the medium containing AA plus EPA/DHA than the medium containing AA alone, indicative of mTORC1 inhibition (Figure 4B). The phosphorylation of PKC and AKT (Ser473), however, remained unchanged by incubation with ω-3 PUFAs (Figure 4B). Thus, ω-6 PUFAs could activate mTORC1, but not mTORC2, in CD4+ T cells, and such activation could be reversed by the addition of ω-3 PUFAs. As an additional confirmation of the importance of mTORC1 in PUFA-mediated CD4+ T cell differentiation, coincubation with rapamycin completely blocked AA-promoted differentiation into Th1 cells (Figure 4, C and D). Thus, the counterregulation of mTORC1 activity by ω-3 and ω-6 PUFAs plays a critical role in the differentiation of CD4+ T cells.
Figure 4. ω-3 PUFAs regulate Th cell differentiation through the inhibition of mTORC1.
(A and B) Immunoblot analysis of mTOR activation in lysates of naive CD4+ T cells from nondiabetic NOD mice. Cell lysates were stimulated for 24 hours with anti-CD3 and anti-CD28 Abs plus various doses of AA, DHA, and EPA (top lanes) in serum-containing medium. (C) Representative flow cytometric images, with the numbers in quadrants indicating the percentage of IFN-γ+ Th cells in splenocytes from 10-week-old nondiabetic NOD mice. Splenocytes were cultured under PMA and ionomycin stimulation in the presence of AA (50 μM) plus rapamycin (10 nM) or AA alone. (D) Quantification of Th1 cell percentages (n = 3/group). **P < 0.01 versus the AA group (Student’s t test). Data are representative of 3 independent experiments, and flow cytometric samples were gated on CD4+ T cells (CD3+CD8–). All values represent the mean ± SEM.
Reversal of diabetes development by gene therapy or nutritional supplementation of ω-3 PUFAs.
Intervention with an ω-3 PUFA–enriched diet before hyperglycemia constituted only a preventive method against T1D development in NOD mice. We speculated that there would also be a therapeutic benefit of such a modality for diabetic NOD mice. To this end, we designed a lentiviral vector carrying a modified Caenorhabditis elegans (C. elegans) cDNA, mfat-1 (referred to herein as lenti-mfat-1), that encodes an ω-3 fatty acid desaturase (42). By adding a double bond at the ω-3 position, the mFAT-1 enzyme can specifically convert ω-6 PUFAs into ω-3 PUFAs, thereby elevating endogenous levels of ω-3 PUFAs with a concomitant decrease in ω-6 PUFAs. In parallel, we also set up a group of female NOD mice that were fed an EPA/DHA-enriched diet after they became diabetic.
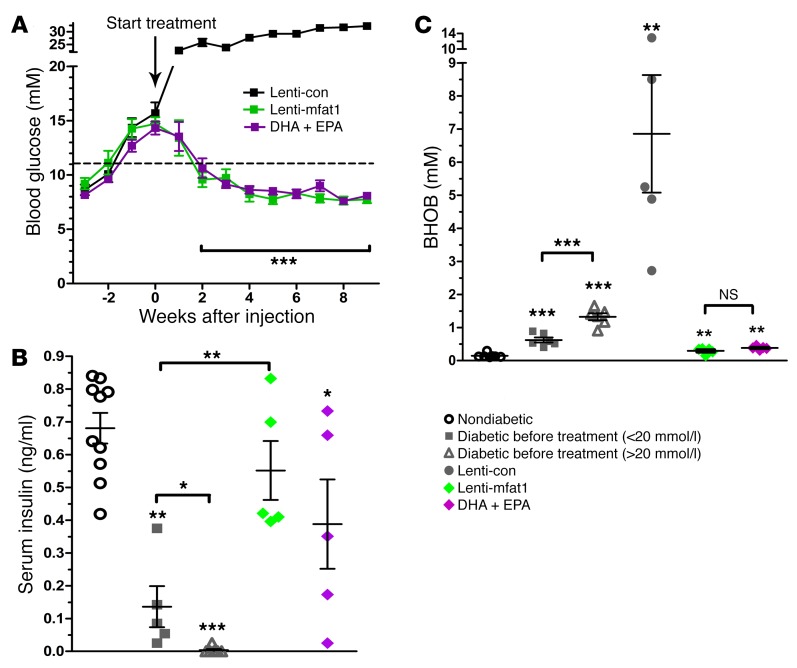
Delivery (i.v.) of lenti-mfat-1 (109 transducing units per kilogram [TU/kg]) into NOD mice increased blood concentrations of ω-3 PUFAs, primarily EPA and DHA. Because of the activity of mFAT-1, the concentrations of ω-6 PUFAs, mainly AA, were significantly reduced, and the ratio of ω-6/ω-3 was decreased from 4.2 to 1.7 (Supplemental Table 6). Approximately 3 to 4 weeks after lenti-mfat-1 treatment, nonfasting blood glucose levels had gradually dropped to 8 mmol/l or lower in 7 of 10 diabetic NOD mice. The euglycemic condition was sustained for at least 9 weeks after virus delivery. Similarly, in the dietary intervention group, 7 of 11 diabetic mice regained normal glucose concentrations 3 weeks after starting an EPA/DHA-enriched diet (Figure 5A). The treated mice maintained a euglycemic condition and survived for at least 182 days. Concomitant with the normalization of blood glucose levels, serum insulin levels in the lenti-mfat-1–treated group and the EPA/DHA-enriched diet group were completely restored, reaching 0.5 ng/ml and 0.4 ng/ml, respectively (Figure 5B). These insulin levels were similar to those observed in the prediabetic NOD mice and in sharp contrast to the almost undetectable insulin levels in the control lentivirus–treated (lenti-con–treated) diabetic NOD mice (Figure 5B).
Figure 5. ω-3 PUFAs exert a therapeutic effect on hyperglycemia in diabetic NOD mice.
(A) Nonfasting blood glucose levels in diabetic NOD mice (nonfasting blood glucose levels for 2 consecutive weeks = 11.1–20 mmol/l) after i.v. tail-vein injection of lenti-con (black, n = 7) or lenti-mfat-1 (green, n = 7), or DHA plus EPA dietary intervention (purple, n = 7). ***P < 0.0001 versus the lenti-con group (Student t test). (B and C) Concentrations of nonfasting serum insulin and BHOB, the ketone metabolite, in nondiabetic and diabetic NOD mice (nonfasting blood glucose levels <20 mmol/l for 2 consecutive weeks) before treatment; diabetic NOD mice (nonfasting blood glucose level for 2 consecutive weeks = 11.1–20 mmol/l) after lentivirus injection; or in mice that received DHA plus EPA diet intervention for 9 weeks. *P < 0.05, **P < 0.01, and ***P < 0.0001 versus the nondiabetic group (n = 5–10/group) (Student’s t test). Each point represents an individual mouse, and data are representative of 2 independent experiments. All values represent the mean ± SEM.
Interestingly, either treatment method could only rescue those diabetic NOD mice with nonfasting blood glucose levels below 20 mmol/l. A ketone test of β-hydroxybutyrate (BHOB) revealed that mice bearing blood glucose levels above 20 mmol/l had very severe diabetic ketoacidosis (DKA), with BHOB levels reaching 7.0 mmol/l, which was at least 10-fold higher than the levels seen in NOD mice with blood glucose levels below 20 mmol/l (Figure 5C).
Therapeutic supplementation of ω-3 PUFAs promotes islet and β cell regeneration.
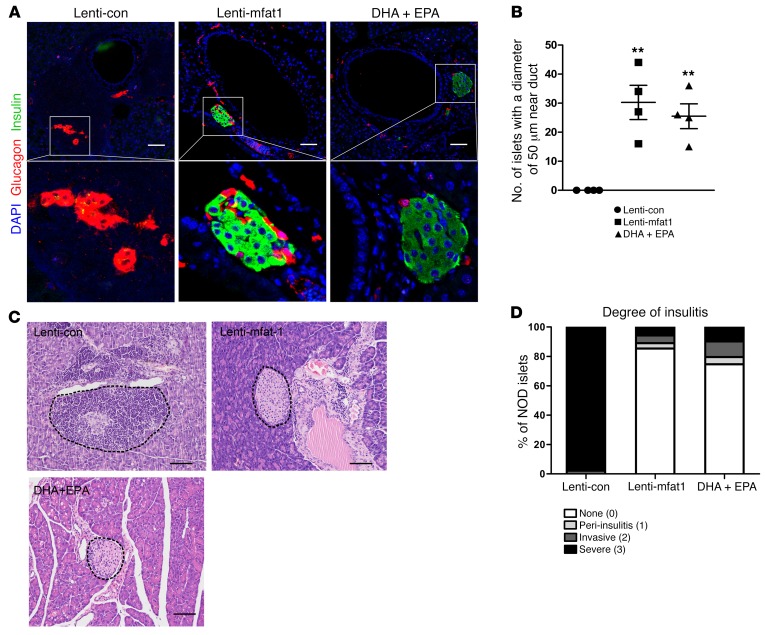
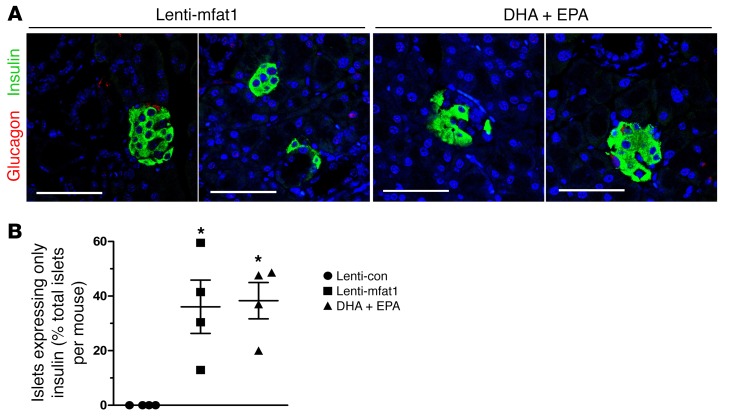
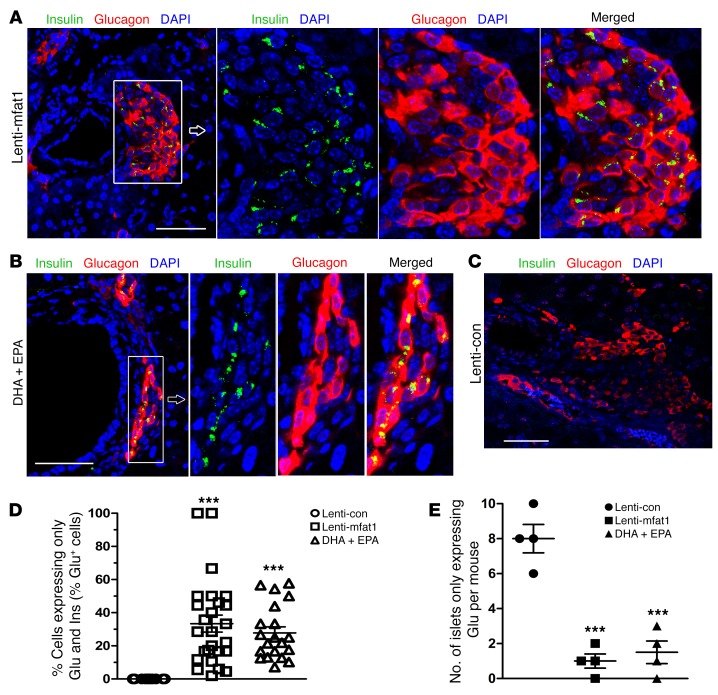
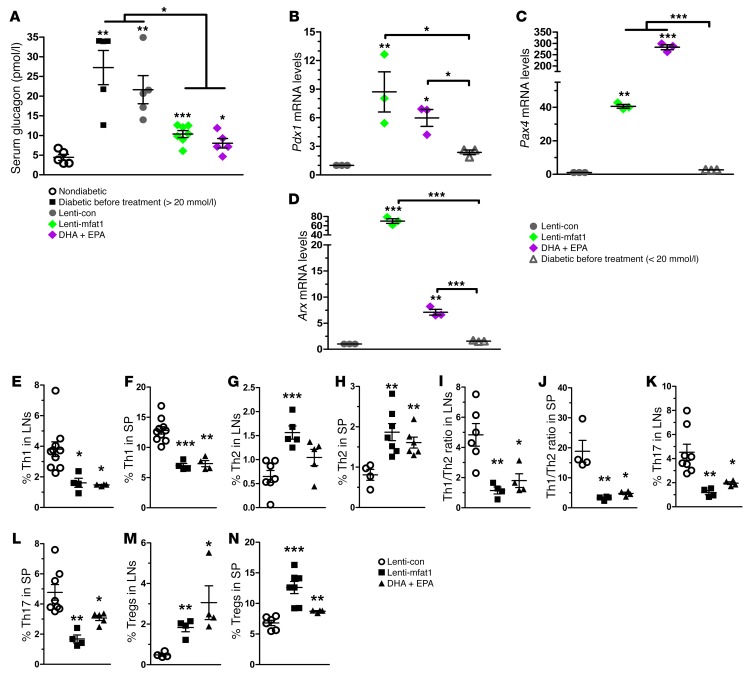
At the end of 9 weeks of therapy with the lentiviral gene or the EPA/DHA-enriched diet, confocal imaging revealed newly formed insulin-staining islets with a diameter of at least 50 μm that were located adjacent to pancreatic ducts (Figure 6, A and B). Importantly, lymphocyte infiltration of neopancreatic islets in the lenti-mfat-1–treated or EPA/DHA-enriched dietary group was much lower compared with that observed in the lenti-con–treated groups (Figure 6, C and D), suggesting that both therapeutic approaches deterred the immune attack on the pancreas. Interestingly, we found that a high percentage of regenerated islets (~40%) had essentially all β cells, with very few α cells (Figure 7, A and B). Confocal imaging revealed multiple cells with both insulin and glucagon staining (indicated by yellow fluorescence) within the regenerated islets near pancreatic ducts after either interventional approach (Figure 8, A–E), which was indicative of transdifferentiation from α cells into β cells. Consistent with these observations, blood glucagon levels in the nonfasted ω-3 PUFA intervention group were much lower than those in the nontreated diabetic mice as well as in the lenti-con–treated NOD mice (Figure 9A). Also, compared with the nontreated diabetic and lenti-con–treated groups, the transcription levels of Pdx1, Pax4, and aristaless-related homeobox (Arx) were drastically increased following treatment with either ω-3 PUFA interventional approach (Figure 9, B–D). Taken together, our data showed that the lenti-mfat-1 virus could regenerate pancreatic β cells, at least some of which came from transdifferentiated α cells.
Figure 6. ω-3 PUFAs have a therapeutic effect on immune infiltration in diabetic NOD mice.
Confocal images (A) and quantification (B) of islets with a diameter of 50 μm that appeared adjacent to pancreatic ducts in diabetic NOD mice after lentivirus treatment and DHA plus EPA dietary intervention for 9 weeks (n = 4/group). Scale bars: 50 μm. Original magnification: ×400. **P < 0.01 versus the lenti-con group (Student’s t test). Values represent the mean ± SEM. (C) H&E-stained sections of islets from pancreatic tissue obtained from diabetic NOD mice after lentivirus treatment and DHA plus EPA dietary intervention for 9 weeks. Scale bars: 50 μm. Images are representative of 3 biological replicates. (D) Quantification of the incidence of insulitis in diabetic NOD mice after lentivirus injection or DHA plus EPA dietary intervention for 9 weeks (n = 4/group). Islets were sorted into 4 categories on the basis of the relative degree of immune infiltration: no insulitis (0), peri-insulitis (1), invasive insulitis (2), and severe insulitis (3). The differences in the incidence of no insulitis or severe insulitis between the lenti-con and lenti-mfat-1 groups (P < 0.0001) and between the DHA plus EPA and lenti-con groups (P < 0.0001) were significant. Statistical significance was determined by Pearson’s χ2 test.
Figure 7. Islet and β cell regeneration in diabetic NOD mice treated with ω-3 PUFAs.
Pancreases were harvested from 9-week-old mice that had received lentivirus treatment and DHA plus EPA dietary intervention. Confocal images (A) and quantification (B) of islets expressing only insulin, without α cells. These islets were discovered next to the ductal epithelium in NOD mice treated with lenti-mfat-1 and fed a DHA plus EPA diet (n = 4/group). β cells (insulin, green), α cells (glucagon, red), and nuclei (DAPI, blue) are shown. Scale bars: 50 μm. *P < 0.05 versus the lenti-con group (ANOVA). Images are representative of 3 biological replicates. All values represent the mean ± SEM.
Figure 8. Islet and β cell regeneration and colocalization of α cells and β cells in diabetic NOD mice treated with ω-3 PUFAs.
Mice were sacrificed and pancreases harvested at 9 weeks following lentivirus treatment and DHA plus EPA dietary intervention. (A–C) Confocal images of colocalization (yellow areas) of α cells and β cells detected in islets and cells near the pancreatic duct in lenti-mfat-1–treated and DHA plus EPA dietary intervention groups of diabetic NOD mice. β cells (insulin, green), α cells (glucagon, red), and nuclei (DAPI, blue) are shown. Scale bars: 50 μm. Original magnification: ×400. Images are representative of 3 biological replicates. (D) Quantification of islets detected by glucagon and insulin (Ins and Glu) colocalization (n = 20–26 islets/group). (E) Quantification of islets expressing only glucagon (Glu) and no insulin (n = 4/group) and that appeared adjacent to pancreatic ducts. ***P < 0.0001 versus the lenti-con group (ANOVA). All values represent the mean ± SEM.
Figure 9. β Cell regeneration and modulation of Th cell subsets after lentivirus and dietary therapy with ω-3 PUFAs in diabetic NOD mice.
Mice were sacrificed at 9 weeks of age following lentivirus treatment and DHA plus EPA dietary intervention, and pancreases were harvested. (A) Concentrations of nonfasting serum glucagon levels in nondiabetic mice; diabetic NOD mice (nonfasting blood glucose level for 2 consecutive weeks <20 mmol/l) before treatment; and diabetic NOD mice (nonfasting blood glucose level for 2 consecutive weeks >11.1 mmol/l) after ω-3 PUFA therapy (n = 5–7/group). *P < 0.05, **P < 0.01, and ***P < 0.0001 versus the nondiabetic group (Student’s t test). Data are representative of 2 independent experiments. (B–D) mRNA expression of Pdx1, Pax4, and Arx measured by RT-PCR in pancreases from NOD mice that received ω-3 PUFA therapy. *P < 0.05, **P < 0.01, and ***P < 0.0001 compared with the lenti-con group (n = 3 per group) (Student’s t test). Data are representative of 3 independent experiments. (E–N) Quantification (n = 4–10 per group) of the percentage of intracellular staining of IFN-γ+, IL-4+, IL-7+, and CD25+FoxP3+ Th cells in LNs and spleens of diabetic NOD mice that received ω-3 PUFA therapy. Representative flow cytometric images are shown in Supplemental Figure 8. Data are representative of 3 independent experiments. *P < 0.05, **P < 0.01, and ***P < 0.0001 compared with the lenti-con group (Student’s t test). All values represent the mean ± SEM.
Therapeutic supplementation of ω-3 PUFAs altered CD4+ T cell differentiation profiles in diabetic NOD mice.
Implementation of either ω-3 PUFA dietary intervention had an inhibitory effect on the population of Th1 and Th17 cells but strongly promoted Th2 and Treg differentiation and activation (Figure 9, E–N and Supplemental Figure 8). Interestingly, the stimulatory effect on Th2 cells achieved through the gene therapy method was even more effective than that of dietary intervention. Thus, either ω-3 PUFA supplementation method could restore the differentiation profiles of CD4+ T cells and therefore stall the progression of autoimmunity.
Modulation of human CD4+ T cell differentiation by ω-3 PUFAs.
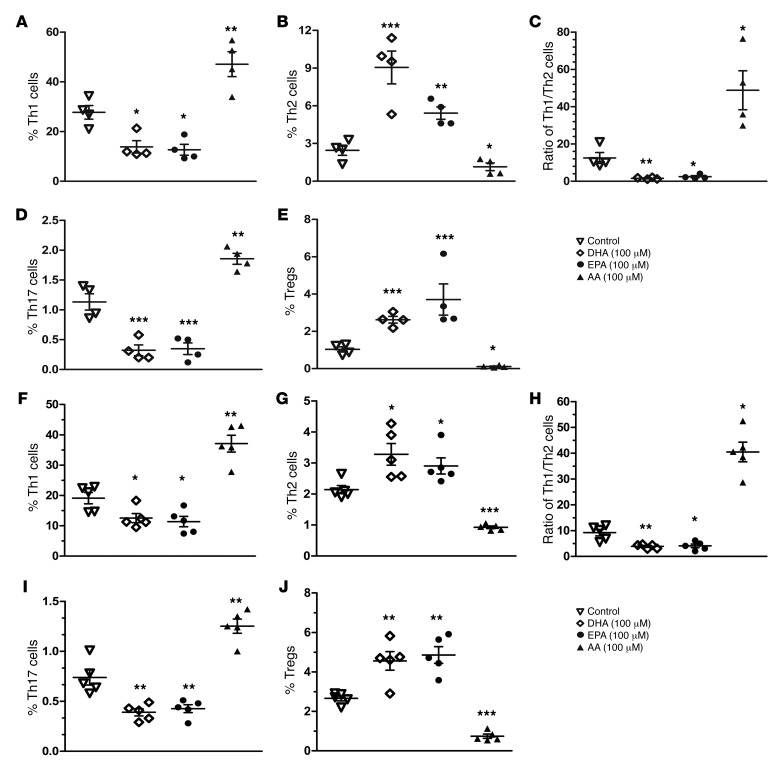
The data presented above led us to further explore whether ω-3 PUFAs have a similar impact on human CD4+ T cell differentiation. Human peripheral blood mononuclear cells (PBMCs) were isolated from 4 T1D patients and 5 nondiabetic donors (patients’ information, including A1C and autoantibodies, is provided in Supplemental Table 7) and treated with 100 μM DHA, EPA, and AA, separately, for 24 hours. CD4+ T cell differentiation was analyzed by flow cytometry. Consistent with the findings in NOD mice, we found that EPA and DHA essentially rebalanced the Th1/Th2 ratio from approximately 12 to approximately 1 by both increasing Th2 cell populations and decreasing Th1 cell populations in the samples from T1D patients (Figure 10, A–E and Supplemental Figure 9). Meanwhile, we also observed a significant reduction of Th17 cells and an elevation of Tregs. In contrast, AA, an ω-6 PUFA generally considered to be proinflammatory, exerted effects on CD4+ T cell differentiation that were completely opposite to those induced by EPA or DHA. We also obtained similar results in the T cell samples from nondiabetic donors (Figure 10, F–J and Supplemental Figure 10). To further confirm the impact of ω-3 and ω-6 PUFAs on human CD4+ T cells, we also used flow cytometry to follow the expression of transcription factors such as T-bet, GATA3, and RORγT in human PBMCs following treatment with AA, DHA, or EPA. The results were clearly consistent with the staining panels of cytokines from the nondiabetic donors (Supplemental Figure 11).
Figure 10. ω-3 and ω-6 PUFAs readjust CD4+ T cell differentiation in PBMCs from T1D patients and nondiabetic donors in vitro.
Quantification of the percentage of intracellular staining of IFN-γ+, IL-4+, IL-7+, and CD25+FoxP3+ Th cells in PBMCs from 4 T1D patients (A–E) and 5 nondiabetic donors (F–J). Cells were cultured for 24 hours under PMA and ionomycin stimulation in the presence of DHA, EPA, and AA (100 μM) added at the time of activation. Representative flow cytometric images are shown in Supplemental Figures 9 and 10. *P < 0.05, **P < 0.01, and ***P < 0.0001 compared with the control group (Student’s t test). Each point represents an individual patient or donor, and the data are representative of 3 independent experiments. All values represent the mean ± SEM.
Discussion
The deterrence of autoimmune attack and regeneration of pancreatic β cells remain daunting tasks in the pursuit of a complete cure for T1D. The study described here suggested the clinical potential of gene therapy or nutritional supplementation of ω-3 PUFAs — EPA and DHA in particular — in preventing and reversing the development of autoimmunity and T1D. Both approaches alleviated T cell–mediated autoimmunity by rebalancing the ratio of Th1 and Th2 cells, decreasing Th17 cells, and increasing Tregs, while reducing circulating levels of inflammatory cytokines. Importantly, we observed a full recovery of pancreatic islet function and a near-complete absence of any lymphocyte infiltration into regenerated islets in the diseased NOD mice following the nutritional or gene therapeutic intervention. To our knowledge, this represents the first report of a single treatment method that can both stall autoimmunity and, concomitantly, fully restore pancreatic β cell function. Three recent clinical trials in independent cohorts have convincingly demonstrated that long-term dietary intake of ω-3 PUFAs, starting from infancy, could significantly decrease the incidence of islet autoimmunity and T1D (22–24). The results from our studies are entirely consistent with the findings revealed in these clinical studies.
A targeted autoimmune attack on islets involves dysregulation and dysfunction of both T cells and B cells (16). T cell–mediated immunity is primarily responsible for the direct damage of β cells and inflammatory injury (16). The titers of autoantibodies secreted by B cells change dynamically or even become undetectable in some patients (43). In addition, the strong association between T1D and the HLA class II locus can trigger the activation of CD4+ T cells (44). Thus, our studies have primarily focused on autoimmune responses driven by CD4+ T cells in NOD mice. Elevated populations of Th1 cells, the main effectors of diabetes in NOD mice, are a hallmark of T cell–mediated autoimmunity that in turn can further stimulate CD8+ T cells and macrophages to promote inflammatory and immune attacks on pancreatic β cells (45). CD4+ T cells preferentially differentiate into Th1 cells, with a concomitant decrease in Th2 cells in NOD mice, as indicated by the elevated ratio of IFN-γ/IL-4 (46, 47). In addition to dysregulated Th1/Th2 ratios, T1D patients have an elevated percentage of activated Th17 cells and decreased numbers of Tregs. Specific anti–IL-17 antibodies have been shown to significantly increase the proportion of Tregs (48). Mesenchymal stem cell–based cellular therapy has already shown promise in elevating Tregs, which then suppress immune attack in a T1D model (49). In our study, supplementation of ω-3 PUFAs, provided as either a preventative or therapeutic modality, significantly suppressed Th1 and Th17 cells, reduced secreted IFN-γ and IL-17 levels, and increased Th2 cells and Tregs. Meanwhile, the regulatory effects of ω-3 and ω-6 PUFAs on IFN-γ secretion by CD8+ T cells were consistent but somewhat minor when compared with the effects on IFN-γ secretion by CD4+ T cells, and IL-4 secretion by CD8+ T cells was undetectable. Thus, CD8+ T cells participate in the pathogenesis of T1D mainly through their cytotoxic effects on islet cells, but not through cytokine secretion (50, 51). In further extending this concept to application in humans, we found that DHA and EPA had the same effects on the differentiation of CD4+ T cells isolated from T1D donors as those observed in NOD mice. Thus, ω-3 PUFA–initiated global changes in CD4+ T cells are likely the primary cellular mechanisms underlying the blockade of autoimmune development.
Multiple mechanisms may have contributed to the effects of ω-3 PUFAs on CD4+ T cell differentiation. EPA/DHA-derived eicosanoids, synthesized through the activities of enzymes such as LOX and COX, tend to be antiinflammatory, whereas those derived from ω-6 PUFAs are proinflammatory (37). For example, AA-derived eicosanoids play a role in the development of arthritis, lupus, and asthma (52). Some of the eicosanoids that showed drastic changes in our metabolomics analysis of pancreatic samples played distinct roles in CD4+ T cell differentiation. For example, PGD3, an EPA-derived metabolite, had a strong inhibitory effect on Th1 and Th17 cell differentiation and elevated Th2 and Treg populations. On the other hand, 17,18-DiHETE, another EPA-derived metabolite, decreased only Th17 cell populations. RvD1, a DHA-derived metabolite, had a strong inhibitory effect on Th1 cell differentiation and strongly promoted Th2 and Treg differentiation. As one might expect, AA-derived proinflammatory eicosanoids, such as 15-HETE and 20-HETE, had effects on naive CD4+ T cell differentiation that were exactly opposite to those of EPA/DHA-derived metabolites, further confirming the specificity of the effects of these eicosanoids.
Regulation of mTOR activity plays a critical role in the differentiation of CD4+ T cells (40). Application of rapamycin can inhibit the proliferation of T cells and the differentiation into Th1 and Th17 cells, even in the presence of T cell receptor (TCR) costimulatory molecules such as CD28 (53, 54). T cell–specific mutation of mTOR suppresses differentiation of Th17 cells (54) and promotes naive CD4+ T cell differentiation into Tregs (54). In our studies, EPA/DHA strongly inhibited the activation of mTORC1 by AA. Similar to the effect of EPA and DHA, rapamycin also blocked the Th1 differentiation induced by AA. Such results are also reminiscent of the anti-diabetes and autoimmunity prevention effects of rapamycin in NOD mice that were reported by several groups (55–59).
Increased intake of ω-6 PUFAs has been found to amplify immune response and the inflammatory events in mice with allergic diseases (60). Moreover, oleic acid and linoleic acid diets have been reported to elevate the development of autoimmunity and intestinal inflammation in mice (61, 62). Although the conclusions from these studies were generally consistent with our findings, one study also found the beneficial effect of an ω-6 PUFA–rich diet in alleviating inflammation in drug-induced mucosal damage (63). Thus, the effects of ω-6 PUFAs on inflammatory diseases should be carefully considered in different immune disease models.
Arguably, the most striking finding from this study is the reversal of autoimmune development and stable normalization of blood glucose levels following gene therapeutic or nutritional supplementation of ω-3 PUFAs in diabetic NOD mice. We believe these findings have strong clinical implications for the treatment of T1D. First, both intervention methods successfully readjusted the differentiation of CD4+ T cells, including the rebalancing of the Th1/Th2 ratio, the reduction of Th17 cells, and the elevation of Treg numbers, thus causing a nearly complete absence of lymphocyte infiltration into regenerated islets. Second, both interventions regenerated pancreatic islets and fully restored blood insulin levels. In the absence of a meaningful number of preexisting β cells (as is the case in diabetic NOD mice), neogenesis can be achieved via either differentiation of progenitors within the ductal epithelium (64) or transdifferentiation of acinar (65) or α cells (66) into β cells. Although the underlying mechanisms remain to be defined, neogenesis is probably the primary event, as we found expression levels of Pdx1 (67) and Pax4 (68) to be sharply elevated. We also discovered the phenomenon of α cell transdifferentiation, as a significant percentage of newly generated islets often had very few α cells and glucagon/insulin double staining following the therapeutic interventions. For reasons that are not clear, we were only able to rescue a subset of diabetic mice with our therapeutic interventions, particularly those mice without overt conditions (nonfasting blood glucose ≤20 mmol/l and only mild ketoacidosis), suggesting an irreversible damage to the neogenesis mechanisms after prolonged exposure to the severe DKA condition.
Most of the preventative and therapeutic modalities developed in NOD mice have not been successfully realized in humans with T1D (69, 70). Part of this failure may reside in the complexity of the pathogenesis of T1D. On this note, we were encouraged to observe the same effects of EPA/DHA on human CD4+ T cells as those on murine T cells. In addition, the results from our studies are entirely consistent with the conclusions derived from the DAISY and Norwegian clinical studies (22, 23).
In this study, the daily intake of EPA/DHA applied to NOD mice was approximately 3.6 g/kg BW, which is high by clinical standards. Such a dose could very well be a saturating amount in these mice, and a dose well below this level may only be needed to achieve a therapeutic effect. We do not yet know whether the same dose for mice can simply be mathematically translated into a dose for humans to achieve the same results, given the very different metabolic profiles of mice and humans. However, the DAISY study showed that daily supplementation of a mere 150-mg dose of EPA/DHA in infants would have a strong preventative effect in children (22). For infants, starting at 1 year of age, the Norwegian study suggested that daily supplementation (1.0 g DHA/EPA) could reduce their risk of developing T1D (23). These EPA/DHA levels were far below those applied here in the NOD mice, further raising the hope of using such an approach to alleviate T1D in the clinic. Unlike the nutritional supplementation approach, gene therapy led not only to an increase of ω-3 PUFAs but also a significant decrease of ω-6 PUFAs, particularly the proinflammatory AA and its derivatives, which might have also contributed to the suppression of autoimmunity and the restoration of blood glucose levels. The dose of EPA/DHA needed to reverse T1D in the clinic will require a very thorough trial.
Our observations may also offer clinical guidance, in that those patients who are either at the early-onset stage of T1D or have consistently had good management of their blood glucose levels may benefit the most from these interventions. These treatment modalities, if cleared in safety evaluations, may potentially be helpful in the treatment of other types of autoimmune diseases as well.
Methods
Mice and diets.
The NOD (J001976) mouse colony, which was the same colony used in most of the other published studies (71–73), was purchased from The Jackson Laboratory and maintained in a strict specific pathogen–free (SPF) barrier facility at the Model Animal Research Center of Nanjing University. Both male and female NOD mice were tested regularly (at least every 3 months) for microbiological screening when housed at the barrier facility. All microbial screening test results were negative. Mice had ad libitum access to water and food. The 3 groups of mice in this study were fed a semi-purified control diet or the same control diet supplemented with either 10% AA or 10% EPA/DHA (by weight). The diets met National Research Council (NRC) nutrition requirements for mice and varied only in PUFA lipid content (74). The source of ω-3 PUFAs was fish oil (ShangHai HOPE Industry Co., Ltd.). Analysis of other fatty acids and vitamine D3 is shown in Supplemental Table 8. The SPF-level regular mouse diet and the PUFA-enriched diet were manufactured by XIETONG Bioengineering Co., Ltd. Details of the sources of protein, other fats, and other additives are provided in Supplemental Table 9. The distribution of fatty acid species in the fish oil and DHA/EPA diets was analyzed by gas chromatography–mass spectrometry (GC-MS) and is detailed in Supplemental Table 8. All PUFA-enriched diets were vacuum sealed and stored at –20°C before thawing. The PUFA-enriched diets were replenished for the mice every 48 hours. The same lot of PUFA-enriched diets was used throughout this study. Diabetes was confirmed by the presence of blood glucose concentrations above 11.11 mmol/l for 2 consecutive weeks, with the first week of hyperglycemia considered the age of disease onset.
Glucose tolerance tests and insulin tolerance tests.
All 3 groups of mice were subjected to i.p. glucose tolerance tests (GTTs). Briefly, after 16 hours (from 5:00 pm to 9:00 am) of fasting, with free access to water, unanesthetized mice were i.p. injected with a 20% glucose solution (2 g/kg BW). Plasma glucose concentrations were measured at baseline and 15, 30, 60, 90, and 120 minutes after glucose injection. Blood glucose levels were measured from tail-vein blood samples using a glucometer (Accu-Chek; Roche). For insulin tolerance tests, mice were fasted for a 4-hour period during the light cycle before i.p. injection of regular human insulin (0.8 U/kg; Novo Nordisk) diluted in sterile saline. Blood glucose levels were measured before insulin administration and then every 15 minutes afterwards, for 60 minutes (26).
Histological studies and insulitis scoring.
After the mice were sacrificed, pancreases were harvested and fixed with 4% paraformaldehyde overnight at 4°C. The pancreases were paraffin embedded and sectioned (4 μm thickness). To assess insulitis, islets from H&E-stained sections were scored using standard methods and placed into 1 of 4 categories: 1) no insulitis; 2) peri-insulitis (leukocytes in the periphery of the islet); 3) invasive insulitis (25%–50% coverage of the islet); and 4) severe insulitis (>50% infiltration). Paraffin sections of mouse pancreases were collected at intervals of 100 μM. Mouse islets (20–50 per pancreata) were scored for the evaluation of insulitis. This procedure was used for 1 paraffin cross section of pancreatic tissue placed on individual slides from each of the 7 animals included in the histopathological study, and all slides were evaluated by a pathologist and scored for degree of perivascular and islet-associated monocyte infiltration. In addition, for each section included, a standard H&E-stained slide was prepared and submitted for pathological review.
Confocal microscopy and immunofluorescence.
For immunofluorescence staining, 5-μm-thick paraffin sections were assayed with DAPI as a counterstain. The primary antibodies used were: rabbit anti-mouse insulin (1:100; Cell Signaling Technology) and monoclonal anti-mouse glucagon (1:1,000; Sigma-Aldrich). The secondary antibodies (1:1,000; Molecular Probes, Thermo Fisher Scientific) used were: Alexa Fluor 555 donkey anti-mouse and Alexa Fluor 488 donkey anti-rabbit. Sections were examined under a Zeiss LSM 710 confocal microscope.
Serum analysis.
Blood was collected when the NOD mice were sacrificed. Serum insulin levels were measured using an insulin ELISA Kit (EMD Millipore). Plasma glucagon and BHOB levels were measured using an ELISA Kit (Mercodia) and a Colorimetric Assay Kit (Cayman Chemical).
Human PBMC isolation and flow cytometry.
Human blood samples were obtained from T1D donors. The resulting PBMCs were prepared by differential density gradient separation (Lymphoprep; AXIS-SHIELD). The following anti-human surface antibodies were purchased from BD Biosciences: CD3 (UCHT1) and CD8 (RPA-T8). The following anti-mouse surface antibodies were purchased: CD3 (145-2C11; BD Biosciences) and CD8 (53-6.7; eBioscience). For intracellular cytokine FACS, we stimulated cells with PMA (50 ng/ml; Sigma-Aldrich); ionomycin (500 ng/ml; Sigma-Aldrich); and brefeldin A (5 μg/ml; Sigma-Aldrich) for 5 hours. For the detection of CD4+ T cells, CD3+ cells were gated as whole T lymphocytes with CD3-PerCP Cy5.5 mAb. In this gated CD3+ cell population, CD8 was labeled with CD8-FITC mAb. The CD8– cells were selected as the targeted CD4+ T cells (see Figure 4C and Supplemental Figures 2, 3, and 5 and 7–11). Cells were fixed and permeabilized with Fixation/Permeabilization Diluent (eBioscience) and then stained for IFN-γ, IL-4, and IL-17 (Miltenyi Biotec for mouse samples; BD Biosciences for human samples). For Treg staining, we used human and mouse Treg-staining kits (catalogs 88-8999 and 88-8111; eBioscience) according to the manufacturer’s protocol. Cells (105 per sample) were collected on an Accuri-C6 (BD Biosciences) and analyzed using FlowJo software (Tree Star). Each test was repeated 3 times. Analysis of mouse Th1, Th2, and Th17 cytokines in culture supernatants was performed with a BD Cytometric Bead Array.
CD4+ T cell isolation, cell culture, and Western blot analysis.
Naive CD4+ T cells from spleens were obtained by negative selection using the Miltenyi Biotec CD4+ Purification Kit II, according to the manufacturer’s recommendations. RPMI 1640 (Gibco, Thermo Fisher Scientific) supplemented with 10% FBS (HyClone; GE Healthcare), 50 mM β-mercaptoethanol, 100 U/ml penicillin, 100 mg/ml streptomycin, nonessential amino acids, 1 mM sodium pyruvate, and 10 mM HEPES was used as a complete culture medium. Magnetic-activated cell sorter–purified (MACS-purified) CD4+ T cells (purity >96%) were cultured in 12-well plates (2.0 × 106 cells per well) and stimulated with 2 μg/ml soluble anti-CD3 Ab and 2 μg/ml soluble anti-CD28 Ab (both from BD Biosciences). For each analysis, 8 × 106 CD4+ T cells were lysed and the aliquots analyzed by Western blotting using phospho-specific Abs or total protein (Cell Signaling Technology), with β-actin (Sigma-Aldrich) as the control.
RNA isolation, cDNA synthesis, and real-time PCR.
RNA from NOD mice pancreas was prepared using an RNeasy Micro Kit (QIAGEN). One microgram of RNA was used for the preparation of single-stranded cDNA using a QuantiTect Reverse Transcription Kit (QIAGEN). Real-time PCRs were performed using the QuantiNova SYBR Green PCR Kit (QIAGEN) on Applied Biosystems StepOne equipment (Applied Biosystems). Reactions were performed at least in triplicate, and specificity of the amplified products was determined by melting peak analysis. Quantification for each gene of interest was performed using the 2–ΔΔCt method. Quantified values were normalized against the housekeeping gene Actb. The primers used are listed in Supplemental Table 10.
Gas chromatographic analysis of fatty acids.
Lipids were extracted from mouse food and whole-blood samples collected from mouse tail veins according to the method of Bligh and Dyer (75). Gas chromatography was performed on a Model 6890 N Network Gas Chromatograph (Agilent Technologies). Identification of components was done by comparison of retention times with those of authentic standards (Sigma-Aldrich).
Lentiviral vector design, production, and administration.
In this study, the lentiviral transfer vector pLJM1-EGFP (Addgene) containing mfat-1 cDNA encoding a ω-3 fatty acid desaturase under the control of the CMV promoter was designed on the basis of a design that was previously detailed and proven successful for converting ω-6 PUFAs into the corresponding ω-3 forms in transgenic mice (25). Vesicular stomatitis virus glycoprotein–pseudotyped lentiviral supernatant was produced by transient transfection of HEK293FT cells (Life Technologies, Thermo Fisher Scientific) with the 3-plasmid system, including the lentiviral transfer vector pLJM1-EGFP, and the packaging plasmids psPAX2 and pMD2.G. The lentiviral vectors were produced in 293FT cells and concentrated by ultracentrifugation to 108 TU/ml, as described previously (76). Transducing units per milliliter (TU/ml) were determined for each vector stock by assessing EGFP+ cells, using limiting dilution on 293FT cells. Both lenti-mfat-1 and lenti-con at 109 TU/kg were injected into the tail vein of diabetic NOD mice.
Metabolomic analysis.
Pancreatic tissues from female NOD mice fed different PUFA-enriched diets were subject to eicosanoid extraction according to a previously reported method (77). Chromatographic separation involved an ACQUITY UPLC BEH C18 Column (Waters) consisting of ethylene-bridged hybrid particles. The metabolites were quantified using a 5500 QTRAP hybrid triple quadrupole linear ion trap mass spectrometer (SCIEX) equipped with a turbo ion spray electrospray ionization (ESI) source.
Statistics.
All data are presented as the mean ± SEM. P values were calculated using a 2-tailed Student’s t test or 1-way ANOVA with a Tukey’s post-hoc multiple comparisons test. A P value of less than 0.05 was considered statistically significant. Survival plot analyses were used to evaluate the difference in the incidence of diabetes onset between the different groups of NOD mice, with the differences being determined using a Mantel-Cox log-rank test. All statistic analyses (Student’s t tests, ANOVA, and survival analyses) were performed using GraphPad Prism 5.0 software (GraphPad Software).
Study approval.
All experimental protocols were approved by the Research Ethics Committee of Nanjing Medical University, and all experiments were conducted in compliance with guidelines for the care and use of laboratory animals and approved by the IACUC of Nanjing Medical University. PBMCs from T1D patients and nondiabetic donors were provided with prior informed consent, and blood samples were collected with IRB approval of the First Affiliated Hospital of Nanjing Medical University.
Author contributions
XB, SL, XZ, YD, and XL designed and performed experiments, analyzed data, and contributed to the writing of the first draft of the manuscript. FL and YJ supervised the histopathological studies and revised the manuscript. XB and TY recruited patients and performed experiments with human PBMCs. AZZ designed experiments, supervised the work, and wrote and revised the manuscript. All authors reviewed the manuscript.
Supplementary Material
Acknowledgments
We thank Yu Jiang (Department of Pharmacology and Chemical Biology, University of Pittsburgh School of Medicine, Pittsburgh, Pennsylvania, USA) and Xiaochun Bai (Department of Cell Biology, School of Basic Medical Sciences, Southern Medical University, Guangzhou, China) for their helpful scientific discussions and technical assistance. This work was supported by grants from the National Program on Key Basic Research Project of China (973 Program, 2013CB945202, to AZZ and FL); the National Natural Science Foundation of China (NSFC) (81170780 and 81630021, to AZZ; 81372798, to FL; 81200570, to XL); the Natural Science Foundation of Jiangsu Province of China (JSNFC) (BK20130059, to AZZ; 2011766, to XL); and the Key University Science Research Project of Jiangsu Province (12KJB310006, to XL).
Footnotes
Conflict of interest: The authors have declared that no conflict of interest exists.
Reference information:J Clin Invest. 2017;127(5):1757–1771.https://doi.org/10.1172/JCI87388.
Contributor Information
Xinyun Bi, Email: bixinyun@outlook.com.
Fanghong Li, Email: lifanghong2006@yahoo.com.
Shanshan Liu, Email: liushanshan44@163.com.
Yan Jin, Email: jinyan_freedom@126.com.
Xin Zhang, Email: zhanglin890826@sina.com.
Tao Yang, Email: yangt@njmu.edu.cn.
Yifan Dai, Email: daiyifan@njmu.edu.cn.
Xiaoxi Li, Email: lxx@njmu.edu.cn.
References
- 1.Wållberg M, Cooke A. Immune mechanisms in type 1 diabetes. Trends Immunol. 2013;34(12):583–591. doi: 10.1016/j.it.2013.08.005. [DOI] [PubMed] [Google Scholar]
- 2.Atkinson MA, et al. How does type 1 diabetes develop?: the notion of homicide or β-cell suicide revisited. Diabetes. 2011;60(5):1370–1379. doi: 10.2337/db10-1797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Panero F, et al. Fasting plasma C-peptide and micro- and macrovascular complications in a large clinic-based cohort of type 1 diabetic patients. Diabetes Care. 2009;32(2):301–305. doi: 10.2337/dc08-1241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sima AA, et al. C-peptide prevents and improves chronic Type I diabetic polyneuropathy in the BB/Wor rat. Diabetologia. 2001;44(7):889–897. doi: 10.1007/s001250100570. [DOI] [PubMed] [Google Scholar]
- 5.Johansson BL, Sjöberg S, Wahren J. The influence of human C-peptide on renal function and glucose utilization in type 1 (insulin-dependent) diabetic patients. Diabetologia. 1992;35(2):121–128. doi: 10.1007/BF00402543. [DOI] [PubMed] [Google Scholar]
- 6.Johansson BL, Linde B, Wahren J. Effects of C-peptide on blood flow, capillary diffusion capacity and glucose utilization in the exercising forearm of type 1 (insulin-dependent) diabetic patients. Diabetologia. 1992;35(12):1151–1158. doi: 10.1007/BF00401369. [DOI] [PubMed] [Google Scholar]
- 7.Bhatt MP, Lim YC, Kim YM, Ha KS. C-peptide activates AMPKα and prevents ROS-mediated mitochondrial fission and endothelial apoptosis in diabetes. Diabetes. 2013;62(11):3851–3862. doi: 10.2337/db13-0039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Shapiro AM, et al. Islet transplantation in seven patients with type 1 diabetes mellitus using a glucocorticoid-free immunosuppressive regimen. N Engl J Med. 2000;343(4):230–238. doi: 10.1056/NEJM200007273430401. [DOI] [PubMed] [Google Scholar]
- 9.Kroon E, et al. Pancreatic endoderm derived from human embryonic stem cells generates glucose-responsive insulin-secreting cells in vivo. Nat Biotechnol. 2008;26(4):443–452. doi: 10.1038/nbt1393. [DOI] [PubMed] [Google Scholar]
- 10.Bach JF. Anti-CD3 antibodies for type 1 diabetes: beyond expectations. Lancet. 2011;378(9790):459–460. doi: 10.1016/S0140-6736(11)60980-X. [DOI] [PubMed] [Google Scholar]
- 11.Ludvigsson J, et al. GAD65 antigen therapy in recently diagnosed type 1 diabetes mellitus. N Engl J Med. 2012;366(5):433–442. doi: 10.1056/NEJMoa1107096. [DOI] [PubMed] [Google Scholar]
- 12.Orban T, et al. Co-stimulation modulation with abatacept in patients with recent-onset type 1 diabetes: a randomised, double-blind, placebo-controlled trial. Lancet. 2011;378(9789):412–419. doi: 10.1016/S0140-6736(11)60886-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wherrett DK, et al. Antigen-based therapy with glutamic acid decarboxylase (GAD) vaccine in patients with recent-onset type 1 diabetes: a randomised double-blind trial. Lancet. 2011;378(9788):319–327. doi: 10.1016/S0140-6736(11)60895-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Shapiro AM, et al. International trial of the Edmonton protocol for islet transplantation. N Engl J Med. 2006;355(13):1318–1330. doi: 10.1056/NEJMoa061267. [DOI] [PubMed] [Google Scholar]
- 15.van Belle TL, Coppieters KT, von Herrath MG. Type 1 diabetes: etiology, immunology, and therapeutic strategies. Physiol Rev. 2011;91(1):79–118. doi: 10.1152/physrev.00003.2010. [DOI] [PubMed] [Google Scholar]
- 16.Lehuen A, Diana J, Zaccone P, Cooke A. Immune cell crosstalk in type 1 diabetes. Nat Rev Immunol. 2010;10(7):501–513. doi: 10.1038/nri2787. [DOI] [PubMed] [Google Scholar]
- 17.Mosmann TR, Cherwinski H, Bond MW, Giedlin MA, Coffman RL. Two types of murine helper T cell clone. I. Definition according to profiles of lymphokine activities and secreted proteins. J Immunol. 1986;136(7):2348–2357. [PubMed] [Google Scholar]
- 18.Abbas AK, Murphy KM, Sher A. Functional diversity of helper T lymphocytes. Nature. 1996;383(6603):787–793. doi: 10.1038/383787a0. [DOI] [PubMed] [Google Scholar]
- 19.Stechova K, et al. High T-helper-1 cytokines but low T-helper-3 cytokines, inflammatory cytokines and chemokines in children with high risk of developing type 1 diabetes. Diabetes Metab Res Rev. 2007;23(6):462–471. doi: 10.1002/dmrr.718. [DOI] [PubMed] [Google Scholar]
- 20.Luger D, et al. Either a Th17 or a Th1 effector response can drive autoimmunity: conditions of disease induction affect dominant effector category. J Exp Med. 2008;205(4):799–810. doi: 10.1084/jem.20071258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tang Q, et al. Visualizing regulatory T cell control of autoimmune responses in nonobese diabetic mice. Nat Immunol. 2006;7(1):83–92. doi: 10.1038/ni1289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Norris JM, et al. Omega-3 polyunsaturated fatty acid intake and islet autoimmunity in children at increased risk for type 1 diabetes. JAMA. 2007;298(12):1420–1428. doi: 10.1001/jama.298.12.1420. [DOI] [PubMed] [Google Scholar]
- 23.Stene LC, Joner G, Norwegian Childhood Diabetes Study Group Use of cod liver oil during the first year of life is associated with lower risk of childhood-onset type 1 diabetes: a large, population-based, case-control study. Am J Clin Nutr. 2003;78(6):1128–1134. doi: 10.1093/ajcn/78.6.1128. [DOI] [PubMed] [Google Scholar]
- 24.Chase HP, et al. Effect of docosahexaenoic acid supplementation on inflammatory cytokine levels in infants at high genetic risk for type 1 diabetes. Pediatr Diabetes. 2015;16(4):271–279. doi: 10.1111/pedi.12170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wei D, et al. Cellular production of n-3 PUFAs and reduction of n-6-to-n-3 ratios in the pancreatic beta-cells and islets enhance insulin secretion and confer protection against cytokine-induced cell death. Diabetes. 2010;59(2):471–478. doi: 10.2337/db09-0284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Norquay LD, et al. Insulin receptor substrate-2 in beta-cells decreases diabetes in nonobese diabetic mice. Endocrinology. 2009;150(10):4531–4540. doi: 10.1210/en.2009-0395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ferris ST, Carrero JA, Mohan JF, Calderon B, Murphy KM, Unanue ER. A minor subset of Batf3-dependent antigen-presenting cells in islets of Langerhans is essential for the development of autoimmune diabetes. Immunity. 2014;41(4):657–669. doi: 10.1016/j.immuni.2014.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Anderson MS, Bluestone JA. The NOD mouse: a model of immune dysregulation. Annu Rev Immunol. 2005;23:447–485. doi: 10.1146/annurev.immunol.23.021704.115643. [DOI] [PubMed] [Google Scholar]
- 29.Fox CJ, Danska JS. Independent genetic regulation of T-cell and antigen-presenting cell participation in autoimmune islet inflammation. Diabetes. 1998;47(3):331–338. doi: 10.2337/diabetes.47.3.331. [DOI] [PubMed] [Google Scholar]
- 30.Makhlouf L, et al. Depleting anti-CD4 monoclonal antibody cures new-onset diabetes, prevents recurrent autoimmune diabetes, and delays allograft rejection in nonobese diabetic mice. Transplantation. 2004;77(7):990–997. doi: 10.1097/01.TP.0000118410.61419.59. [DOI] [PubMed] [Google Scholar]
- 31.Pfleger C, Meierhoff G, Kolb H, Schloot NC, p520/521 Study Group Association of T-cell reactivity with beta-cell function in recent onset type 1 diabetes patients. J Autoimmun. 2010;34(2):127–135. doi: 10.1016/j.jaut.2009.08.004. [DOI] [PubMed] [Google Scholar]
- 32.Feuerer M, Shen Y, Littman DR, Benoist C, Mathis D. How punctual ablation of regulatory T cells unleashes an autoimmune lesion within the pancreatic islets. Immunity. 2009;31(4):654–664. doi: 10.1016/j.immuni.2009.08.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lindley S, Dayan CM, Bishop A, Roep BO, Peakman M, Tree TI. Defective suppressor function in CD4(+)CD25(+) T-cells from patients with type 1 diabetes. Diabetes. 2005;54(1):92–99. doi: 10.2337/diabetes.54.1.92. [DOI] [PubMed] [Google Scholar]
- 34.Park H, et al. A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat Immunol. 2005;6(11):1133–1141. doi: 10.1038/ni1261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Emamaullee JA, et al. Inhibition of Th17 cells regulates autoimmune diabetes in NOD mice. Diabetes. 2009;58(6):1302–1311. doi: 10.2337/db08-1113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Weylandt KH, Chiu CY, Gomolka B, Waechter SF, Wiedenmann B. Omega-3 fatty acids and their lipid mediators: towards an understanding of resolvin and protectin formation. Prostaglandins Other Lipid Mediat. 2012;97(3–4):73–82. doi: 10.1016/j.prostaglandins.2012.01.005. [DOI] [PubMed] [Google Scholar]
- 37.Schmitz G, Ecker J. The opposing effects of n-3 and n-6 fatty acids. Prog Lipid Res. 2008;47(2):147–155. doi: 10.1016/j.plipres.2007.12.004. [DOI] [PubMed] [Google Scholar]
- 38.Chen Z, et al. mTORC1/2 targeted by n-3 polyunsaturated fatty acids in the prevention of mammary tumorigenesis and tumor progression. Oncogene. 2014;33(37):4548–4557. doi: 10.1038/onc.2013.402. [DOI] [PubMed] [Google Scholar]
- 39.Yang K, et al. T cell exit from quiescence and differentiation into Th2 cells depend on Raptor-mTORC1-mediated metabolic reprogramming. Immunity. 2013;39(6):1043–1056. doi: 10.1016/j.immuni.2013.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Delgoffe GM, et al. The kinase mTOR regulates the differentiation of helper T cells through the selective activation of signaling by mTORC1 and mTORC2. Nat Immunol. 2011;12(4):295–303. doi: 10.1038/ni.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chi H. Regulation and function of mTOR signalling in T cell fate decisions. Nat Rev Immunol. 2012;12(5):325–338. doi: 10.1038/nri3198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Spychalla JP, Kinney AJ, Browse J. Identification of an animal omega-3 fatty acid desaturase by heterologous expression in Arabidopsis. Proc Natl Acad Sci U S A. 1997;94(4):1142–1147. doi: 10.1073/pnas.94.4.1142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Steck AK, et al. Predictors of Progression From the Appearance of Islet Autoantibodies to Early Childhood Diabetes: The Environmental Determinants of Diabetes in the Young (TEDDY) Diabetes Care. 2015;38(5):808–813. doi: 10.2337/dc14-2426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Nepom GT. Class II antigens and disease susceptibility. Annu Rev Med. 1995;46:17–25. doi: 10.1146/annurev.med.46.1.17. [DOI] [PubMed] [Google Scholar]
- 45.Boitard C. T-lymphocyte recognition of beta cells in type 1 diabetes: clinical perspectives. Diabetes Metab. 2013;39(6):459–466. doi: 10.1016/j.diabet.2013.08.001. [DOI] [PubMed] [Google Scholar]
- 46.Charlton B, Lafferty KJ. The Th1/Th2 balance in autoimmunity. Curr Opin Immunol. 1995;7(6):793–798. doi: 10.1016/0952-7915(95)80050-6. [DOI] [PubMed] [Google Scholar]
- 47.Delovitch TL, Singh B. The nonobese diabetic mouse as a model of autoimmune diabetes: immune dysregulation gets the NOD. Immunity. 1997;7(6):727–738. doi: 10.1016/S1074-7613(00)80392-1. [DOI] [PubMed] [Google Scholar]
- 48.Ferraro A, et al. Expansion of Th17 cells and functional defects in T regulatory cells are key features of the pancreatic lymph nodes in patients with type 1 diabetes. Diabetes. 2011;60(11):2903–2913. doi: 10.2337/db11-0090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Vija L, et al. Mesenchymal stem cells: Stem cell therapy perspectives for type 1 diabetes. Diabetes Metab. 2009;35(2):85–93. doi: 10.1016/j.diabet.2008.10.003. [DOI] [PubMed] [Google Scholar]
- 50.Skowera A, et al. β-cell-specific CD8 T cell phenotype in type 1 diabetes reflects chronic autoantigen exposure. Diabetes. 2015;64(3):916–925. doi: 10.2337/db14-0332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Jörns A, et al. Islet infiltration, cytokine expression and beta cell death in the NOD mouse, BB rat, Komeda rat, LEW.1AR1-iddm rat and humans with type 1 diabetes. Diabetologia. 2014;57(3):512–521. doi: 10.1007/s00125-013-3125-4. [DOI] [PubMed] [Google Scholar]
- 52.Patterson E, Wall R, Fitzgerald GF, Ross RP, Stanton C. Health implications of high dietary omega-6 polyunsaturated Fatty acids. J Nutr Metab. 2012;2012:539426. doi: 10.1155/2012/539426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lee K, et al. Mammalian target of rapamycin protein complex 2 regulates differentiation of Th1 and Th2 cell subsets via distinct signaling pathways. Immunity. 2010;32(6):743–753. doi: 10.1016/j.immuni.2010.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Delgoffe GM, et al. The mTOR kinase differentially regulates effector and regulatory T cell lineage commitment. Immunity. 2009;30(6):832–844. doi: 10.1016/j.immuni.2009.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Baeder WL, Sredy J, Sehgal SN, Chang JY, Adams LM. Rapamycin prevents the onset of insulin-dependent diabetes mellitus (IDDM) in NOD mice. Clin Exp Immunol. 1992;89(2):174–178. doi: 10.1111/j.1365-2249.1992.tb06928.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Rabinovitch A, Suarez-Pinzon WL, Shapiro AM, Rajotte RV, Power R. Combination therapy with sirolimus and interleukin-2 prevents spontaneous and recurrent autoimmune diabetes in NOD mice. Diabetes. 2002;51(3):638–645. doi: 10.2337/diabetes.51.3.638. [DOI] [PubMed] [Google Scholar]
- 57.Battaglia M, et al. Induction of tolerance in type 1 diabetes via both CD4+CD25+ T regulatory cells and T regulatory type 1 cells. Diabetes. 2006;55(6):1571–1580. doi: 10.2337/db05-1576. [DOI] [PubMed] [Google Scholar]
- 58.Battaglia M, Stabilini A, Migliavacca B, Horejs-Hoeck J, Kaupper T, Roncarolo MG. Rapamycin promotes expansion of functional CD4+CD25+FOXP3+ regulatory T cells of both healthy subjects and type 1 diabetic patients. J Immunol. 2006;177(12):8338–8347. doi: 10.4049/jimmunol.177.12.8338. [DOI] [PubMed] [Google Scholar]
- 59.Monti P, et al. Rapamycin monotherapy in patients with type 1 diabetes modifies CD4+CD25+FOXP3+ regulatory T-cells. Diabetes. 2008;57(9):2341–2347. doi: 10.2337/db08-0138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.van den Elsen LW, van Esch BC, Dingjan GM, Hofman GA, Garssen J, Willemsen LE. Increased intake of vegetable oil rich in n-6 PUFA enhances allergic symptoms and prevents oral tolerance induction in whey-allergic mice. Br J Nutr. 2015;114(4):577–585. doi: 10.1017/S0007114515002007. [DOI] [PubMed] [Google Scholar]
- 61.Pestka JJ, Vines LL, Bates MA, He K, Langohr I. Comparative effects of n-3, n-6 and n-9 unsaturated fatty acid-rich diet consumption on lupus nephritis, autoantibody production and CD4+ T cell-related gene responses in the autoimmune NZBWF1 mouse. PLoS ONE. 2014;9(6):e100255. doi: 10.1371/journal.pone.0100255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ghosh S, Molcan E, DeCoffe D, Dai C, Gibson DL. Diets rich in n-6 PUFA induce intestinal microbial dysbiosis in aged mice. Br J Nutr. 2013;110(3):515–523. doi: 10.1017/S0007114512005326. [DOI] [PubMed] [Google Scholar]
- 63.Ueda T, et al. Beneficial effect of an omega-6 PUFA-rich diet in non-steroidal anti-inflammatory drug-induced mucosal damage in the murine small intestine. World J Gastroenterol. 2015;21(1):177–186. doi: 10.3748/wjg.v21.i1.177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Xu X, et al. Beta cells can be generated from endogenous progenitors in injured adult mouse pancreas. Cell. 2008;132(2):197–207. doi: 10.1016/j.cell.2007.12.015. [DOI] [PubMed] [Google Scholar]
- 65.Minami K, et al. Lineage tracing and characterization of insulin-secreting cells generated from adult pancreatic acinar cells. Proc Natl Acad Sci U S A. 2005;102(42):15116–15121. doi: 10.1073/pnas.0507567102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Thorel F, et al. Conversion of adult pancreatic alpha-cells to beta-cells after extreme beta-cell loss. Nature. 2010;464(7292):1149–1154. doi: 10.1038/nature08894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Jonsson J, Carlsson L, Edlund T, Edlund H. Insulin-promoter-factor 1 is required for pancreas development in mice. Nature. 1994;371(6498):606–609. doi: 10.1038/371606a0. [DOI] [PubMed] [Google Scholar]
- 68.Sosa-Pineda B, Chowdhury K, Torres M, Oliver G, Gruss P. The Pax4 gene is essential for differentiation of insulin-producing beta cells in the mammalian pancreas. Nature. 1997;386(6623):399–402. doi: 10.1038/386399a0. [DOI] [PubMed] [Google Scholar]
- 69.von Herrath M, Nepom GT. Animal models of human type 1 diabetes. Nat Immunol. 2009;10(2):129–132. doi: 10.1038/ni0209-129. [DOI] [PubMed] [Google Scholar]
- 70.Gill RG, et al. A preclinical consortium approach for assessing the efficacy of combined anti-CD3 plus IL-1 blockade in reversing new-onset autoimmune diabetes in NOD mice. Diabetes. 2016;65(5):1310–1316. doi: 10.2337/db15-0492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Zhang L, et al. Monoclonal antibody blocking the recognition of an insulin peptide-MHC complex modulates type 1 diabetes. Proc Natl Acad Sci U S A. 2014;111(7):2656–2661. doi: 10.1073/pnas.1323436111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Karumuthil-Melethil S, Gudi R, Johnson BM, Perez N, Vasu C. Fungal β-glucan, a Dectin-1 ligand, promotes protection from type 1 diabetes by inducing regulatory innate immune response. J Immunol. 2014;193(7):3308–3321. doi: 10.4049/jimmunol.1400186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Ablamunits V, et al. Synergistic reversal of type 1 diabetes in NOD mice with anti-CD3 and interleukin-1 blockade: evidence of improved immune regulation. Diabetes. 2012;61(1):145–154. doi: 10.2337/db11-1033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. National Research Council (US) Subcommittee on Laboratory Animal Nutrition. Nutrient Requirements of Laboratory Animals: Fourth Revised Edition, 1995. Washington (DC): National Academies Press (US); 1995. [PubMed] [Google Scholar]
- 75.Bligh EG, Dyer WJ. A rapid method of total lipid extraction and purification. Can J Biochem Physiol. 1959;37(8):911–917. doi: 10.1139/o59-099. [DOI] [PubMed] [Google Scholar]
- 76.Tiscornia G, Singer O, Verma IM. Production and purification of lentiviral vectors. Nat Protoc. 2006;1(1):241–245. doi: 10.1038/nprot.2006.37. [DOI] [PubMed] [Google Scholar]
- 77.Li L, et al. Opposite effects of gene deficiency and pharmacological inhibition of soluble epoxide hydrolase on cardiac fibrosis. PLoS ONE. 2014;9(4):e94092. doi: 10.1371/journal.pone.0094092. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.