Abstract
Miscarriage affects ~20% of pregnancies and maternal infections account for ~15% of early miscarriages. Chlamydia trachomatis (Ct) has been associated with miscarriage but the underlying mechanisms are unknown. Successful implantation requires endometrial stromal cell (ESC) decidualisation. Maintenance of pregnancy requires angiogenesis, establishment of the correct cellular milieu and trophoblast invasion, all of which involve the action of chemokines. Our objective was to determine whether Ct infection impacts upon ESC decidualisation and chemokine secretion. Human primary ESC were decidualised in-vitro, infected with Ct serovar E, and changes in expression of genes of interest were measured using RT-PCR, proteomic array and ELISA. We demonstrate for the first time that Ct can infect and proliferate in ESC. Expression of the decidualisation marker prolactin was decreased in Ct-infected ESC at both mRNA and protein levels. Ct infection altered the chemokine profile of decidualised ESC as shown by proteomic array. Chemokines CXCL12 and CXCL16, important for trophoblast invasion, were analysed further and expression was reduced in infected decidualised cells at mRNA and protein levels. Our data indicate that Ct infection of ESC impairs decidualisation and alters chemokine release. These findings at least partially explain how Ct infection could result in adverse pregnancy outcomes.
Introduction
A miscarriage is defined as the spontaneous loss of a pregnancy during the first 24 weeks of gestation and occurs in approximately 20% of clinically recognised pregnancies1. Miscarriages are associated with considerable physical and psychological morbidity. Bleeding due to miscarriage can lead to haemodynamic shock and death and the emotional response to miscarriage can include depression and anxiety1. Approximately, 50% of early miscarriages are attributed to fetal chromosomal abnormalities, however, the underlying cause in other cases is often undefined. A number of infections have also been linked to miscarriage and infections are thought to account for 15% of early and 66% of late miscarriages (reviewed in ref. 1). Several studies have been published regarding the association of pelvic Chlamydia trachomatis (Ct) infection with miscarriage, with Ct prevalence ranging between 11–69% in miscarriages compared to 2–7% in healthy pregnant controls2–6. The mechanisms underlying this association between Ct infection and miscarriage are unknown, though a recent study suggests Ct may interfere with essential early pregnancy inflammatory processes7.
The development of a successful pregnancy depends upon maternal receptivity during the implantation window. This is largely established during decidualisation, the process whereby the stromal cells of the endometrium undergo structural and morphological changes to prepare for possible embryo implantation. Secretion of appropriate chemokine signals by decidual cells contributes to the recruitment of predominantly anti-inflammatory leukocyte subpopulations necessary for pregnancy maintenance8, and prevents recruitment of potentially damaging T lymphocytes9. The maternal immune response to miscarriage associated infections can have detrimental effects on pregnancy maintenance, a characteristic example of which is seen when malaria pathogens are detected in the placenta (reviewed in ref. 1). Furthermore, chemokines not only recruit and impact on immune cells but are also involved in trophoblast invasion and angiogenesis during early pregnancy10.
Both undifferentiated and decidualised endometrium has been shown to be altered compared to normal pregnancies11 in women with spontaneous miscarriage. Impaired decidualisation, measured by a reduction in the decidualisation marker prolactin (PRL) in the endometrium, has been associated with recurrent miscarriage12 and in rodent models decidual cell prolactin production has been shown to be critical for successful pregnancy13. Infection can markedly change the chemokine profile to recruit pro-inflammatory cell subsets.
It is well established that Ct infects endometrial epithelial cells14–16, however the effect of Ct infection on endometrial stromal cell function and decidualisation is yet undetermined and may have a role in the association of Ct infection with miscarriage. Ct is known to cause endometritis, namely inflammation of the endometrium that is often asymptomatic, in non-pregnant women17. Data from animal studies indicate that in mice, C. abortus induces the murine equivalent of miscarriage without fetal harm, likely due to decidual damage18. In cattle C. psittaci associated chronic endometritis is a recognized cause of infertility (strain now known as Chlamydia pecorum)19. To our knowledge, no study to date has identified why Ct can cause endometritis in women or how infection of the stromal compartment of the endometrium might alter the function of human endometrial stromal cells. We therefore aimed to determine whether Ct can infect human endometrial stromal cells (ESC) and examine the effect of Ct infection on decidualisation and chemokine secretion in an in-vitro model.
Results
Ct can directly infect human endometrial stromal cells (ESC)
Primary ESC (n = 4) were infected in 12 well plates with Ct serovar E at a multiplicity of infection (MOI) of 0.01, 0.1, 1, 2 and 3. No visible inclusions were present in cells at MOI 0.01 and MOI 0.1 48 hours post infection and few inclusions were observed at MOI 1 (data not shown). The non-infected ESC and ESC treated with UV-inactivated Ct at MOI 2, displayed no signs of infection (Fig. 1A–C) whereas visible chlamydial inclusions were seen in cells infected at MOI 2 (Fig. 1D,E). No copies of Ct cryptic plasmid were detected in uninfected cells, as assessed using qPCR. ESC exposed to UV-inactivated Ct (which still contained bacterial DNA) had 2.5 × 104–7.5 × 104 plasmid copies per well. Ct infected wells contained 1.2 × 106–3.8 × 106 plasmid copies indicating that significant replication had occurred (Fig. 1F). Although the number of decidualised uninfected ESC was decreased compared to non-decidualised ESC, UV-treated and Ct infected ESC samples contained similar numbers of cells compared to decidualised uninfected ESC (Fig. 1G). In infected wells, between 0.05–20% of ESC contained chlamydial inclusions (Supplementary Fig. S1). These conditions were used for all subsequent experiments. Cells infected at MOI 3 appeared similar to those infected at MOI 2 (equivalent to 4 × 105 organisms per well, Supplementary Fig. S1).
Figure 1.
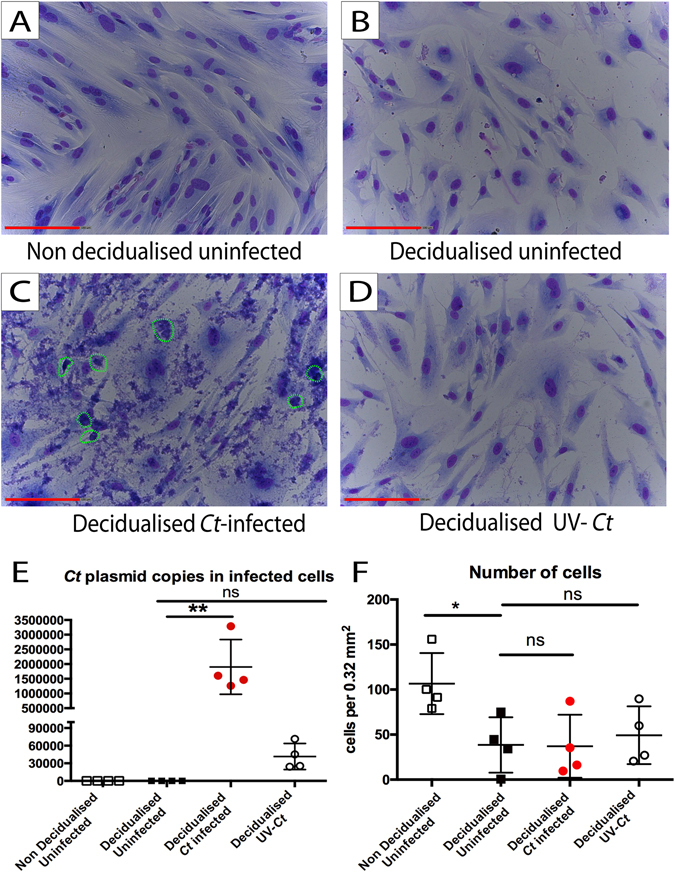
Ct infects decidualised ESC. ESC were infected with Ct MOI 2 following an in-vitro decidualisation protocol. Non decidualised uninfected ESC, uninfected decidualised ESC and UV-Ct treated ESC were used as controls. 48 hours post infection DNA was collected for qPCR and cells were stained with Giemsa. Cell counts were conducted in 15 fields of view per well measuring 0.32 mm2 each. (A) Non decidualised uninfected ESC were elongated and thin. (B) Decidualised uninfected ECS became rounder and larger compared to non decidualised ESC. (C) Ct infected decidualised ESC displayed signs of infection and contained inclusions that were stained purple by Giemsa stain (highlighted by green circles). (D) UV-Ct treated decidualised ESC did not contain chlamydial inclusions and resembled uninfected decidualised ESC in appearance. (E) 25.000–75.000 Ct plasmid DNA copies were detected in UV-Ct treated ESC. Ct infected ESC had a significantly higher number of 1.200.000–3.200.000 plasmid copies per sample, indicating proliferation of Ct only in infected cells (RM one-way ANOVA-Friedman’s test with Dunn’s multiple comparisons test, p = 0.0094, n = 4). (F) Cell counts on Giemsa stained ESC indicated that decidualised uninfected cells were significantly fewer compared to non-decidualised controls (RM one-way ANOVA-Friedman’s test with Dunn’s multiple comparisons test, p = 0.0185, n = 4). In contrast, UV-Ct treated cells and Ct infected cell numbers were similar to uninfected decidualised ESC. Scale bars equal 200 μm. Graphs show the mean and standard deviation.
Ct infection of human ESC results in defective decidualisation
Infection with Ct, but not treatment with UV-inactivated Ct, reduced the expression of the classic decidualisation marker prolactin (PRL) at both mRNA (Fig. 2A) and protein levels (Fig. 2B) in decidualised Ct infected ESC. Similarly, exposure of decidualised ESC to either 200 or 100 µg/ml lipopolysaccharide (LPS) derived from either, Escherichia coli (E. coli) or Salmonella enterica serotype Minnesota (S. Minnesota) also failed to inhibit the of prolactin secretion (Supplementary Fig. S2). Decidualised uninfected ESC had increased levels of both PRL mRNA and protein compared to non decidualised ESC.
Figure 2.
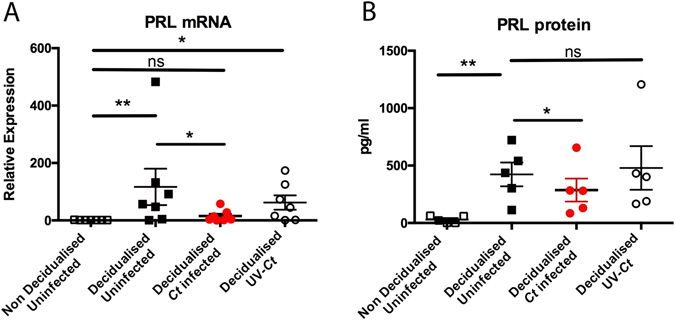
Ct serovar E infection of ESC results in suboptimal decidualisation of ESC. (A) PRL mRNA is upregulated in response to decidualisation stimulus, whereas it is downregulated in infected decidualised cells compared to uninfected controls (n = 5, One way ANOVA Friedman’s test with Dunn’s multiple comparisons test, p = 0.0009 and 0.0151 respectively). (B) PRL protein levels are increased in decidualised non infected cells compared to non decidualised controls as expected (n = 5, One way ANOVA Friedman’s test with Dunn’s multiple comparisons test, p = 0.0044). Reduction of PRL is observed only in cells infected by Ct compared to decidualised controls, indicating the adverse effect of Ct infection on decidualisation (n = 5, One way ANOVA Friedman’s test with Dunn’s multiple comparisons test, p = 0.028). Each circle/square represents an individual patient.
Ct-infected human ESC have an altered chemokine profile
To investigate the effect of Ct infection on chemokine secretion by decidualised ESC, the levels of 31 secreted chemokines were examined by proteomic array in supernatants from decidualised uninfected and infected ESC. All of the chemokines on the array were detected and a pattern of preferential expression between infected and uninfected cells was seen (Fig. 3). Supplementary Table S3 shows that four chemokines were upregulated and 17 downregulated in Ct infected decidualised compared to non-infected ESC (cutoff 5%). Of the chemokines known to be involved with early pregnancy, trophoblast invasion and/or genital infection CXCL12 and CXCL16 were both strongly downregulated in infected decidualised ESC, as were chemokines CCL7, CCL12 and Midkine whilst CXCL7, CXCL1 and XCL1 were strongly upregulated (see Supplementary Table S3 for further details). To validate the array results, qPCR and ELISA were used to measure changes in mRNA and protein levels of chemokines CXCL12 and CXCL16 as a result of infection with Ct. As seen in Fig. 4A and C, there were no significant differences in expression of mRNA encoding CXCL12 and CXCL16. In contrast, infected ESC secreted significantly less CXCL12 and CXCL16 (Fig. 4B,D). Expression of mRNA encoding CXR4 (the receptor for CXCL12) was significantly decreased in infected decidualised ESC compared to uninfected or (Fig. 4E). In contrast, expression of CXCR6 (the receptor for CXCL16) mRNA was not affected by Ct infection (Fig. 4F).
Figure 3.
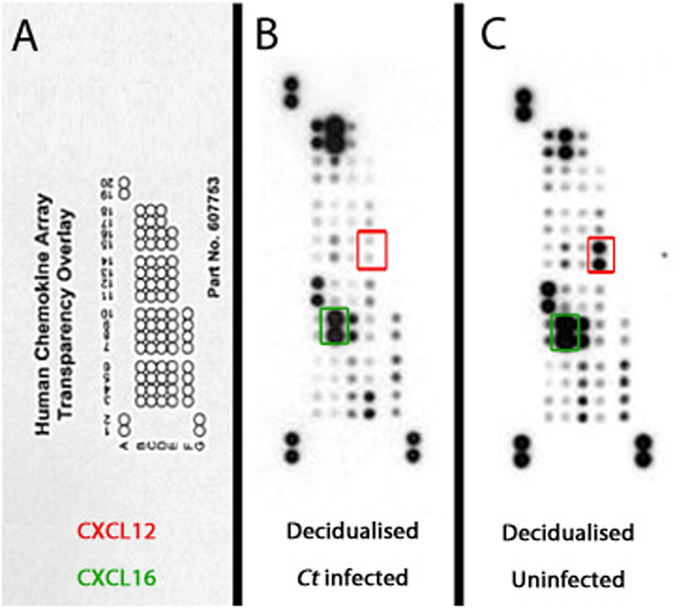
Chemokine proteomic array of 31 chemokines on pooled cell supernatants from Ct infected and uninfected decidualised cells. Each chemokine is represented by a dot. (A) Ct infected decidualised ESC secreted chemokine profile, demonstrating ESC secrete all 31 chemokines. (B) Uninfected decidualised ESC secreted chemokine profile that is altered compared to Ct infected ESC (n = 3).
Figure 4.
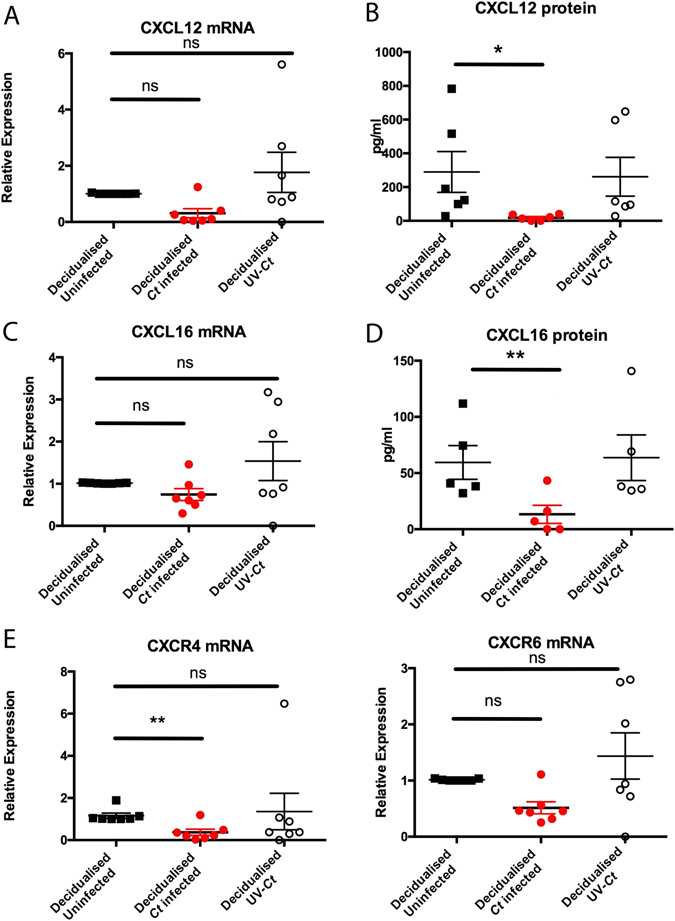
Innate immune response of decidualised ESC to Ct infection. The innate response was determined by measurement (at an mRNA and protein level) of trophoblast invasion-associated chemokines CXCL12 and CXCL16. (A,B) CXCL12 mRNA was not altered in response to infection, however there was a reduction in protein levels in cell supernatants (n = 4, One way ANOVA Friedman’s test with Dunn’s multiple comparisons test p = 0.0267). (C,D) Similarly, there was less secreted CXCL16 protein observed only in infected cells however no change in mRNA levels (n = 4, One way ANOVA Friedman’s test with Dunn’s multiple comparisons test, p = 0.0078). (E) CXR4 mRNA, receptor of CXCL12, was increased only in infected cells (n = 7, One way ANOVA Friedman’s test with Dunn’s multiple comparisons test, p = 0.0267). (F) CXCR6, receptor of CXCL16 does not seem to be affected by Ct infection in mRNA level. Each circle/square represents an individual patient.
Discussion
Herein, we demonstrate for the first time that Ct can infect and proliferate in human endometrial stromal cells (ESC) and that active infection impairs decidualisation and alters the secretion of chemokines.
To our knowledge, all previous studies have only explored the effect of Ct on epithelial cells of the female reproductive tract20–22, however as we have demonstrated here, Ct can also infect and affect the function of ESC. In light of this novel finding, we propose that ascending genital Ct infection might be a more complicated process than previously thought, as Ct could breach the epithelial barrier of the endometrium and infect other cell types such as stromal cells, endothelial cells or glandular epithelial cells (endometrial structure reviewed in ref. 1) causing extended inflammation. Infection with Ct has been reported to cause endometritis and some studies have suggested an association of chronic endometritis with miscarriage and implantation failure23, 24. The effect of a Ct infected endometrial stromal compartment on gestation-related processes, such as decidualisation, might at least partially explain infection related adverse pregnancy outcomes.
Using an in-vitro model, we demonstrated that Ct infection attenuates ESC decidualisation. Furthermore, in the current model active Ct infection, but not exposure to either UV-inactivated organisms or to purified E. coli or S. Minnesota LPS, caused a reduction in mRNA and protein levels of the widely used phenotypic decidualisation marker, PRL. PRL is a key factor in the process of decidualisation and is thought to also be involved in epithelial cell differentiation, implantation, angiogenesis, trophoblast cell growth and immune regulation during early pregnancy25. We believe that this observation is important because a reduction of endometrial PRL has been linked recently to recurrent miscarriage12.
Furthermore, we show that Ct infection changes the chemokine secretion profile in decidualised ESC. Interestingly, we observed a reduction in the levels of CXCL12 and CXCL16. The reduction in protein expression in the culture supernatants cannot be attributed to increased receptor-ligand interactions as the receptor for neither chemokine showed increased expression. We believe that this may have relevance to the role of Ct in miscarriage because these cytokines are known to promote trophoblast migration26–28. It is possible that that infection could therefore lead to defective trophoblast invasion due to lack of essential attractant signalling molecules secreted from maternal decidua, such as CXCL12 and CXCL16.
Our findings are also important because decidualised endometrium from spontaneous and recurrent miscarriages have been reported to have different immune cell profiles compared to viable pregnancies, including increased levels of uterine natural killer cells (uNK) and macrophages29, 30. Dysregulated chemokines due to Ct infection, as indicated in our array, could therefore also impact on the population of immune cells at the feto-maternal interface by altering immune cell recruitment.
In summary, our data suggest a novel mechanism through which infection leads to defective endometrial stromal cell decidualisation, resulting in an altered immune response that could impact upon trophoblast migration and immune cell recruitment. Future work to clarify the potential role of Ct and other bacterial infections upon trophoblast invasion via CXCL12 and CXCL16 dysregulation and immune cell recruitment in the endometrium due to altered chemokine profile could further our understanding of this potential mechanism of infection associated miscarriage.
Materials and Methods
Subjects
Ethical approval for this study was obtained from the Lothian Research Ethics Committee (LREC10/S1402/59). Informed written consent obtained from all patients and all of the methods were carried out in accordance with the approved guidelines. Human endometrial stromal cells (ESC) were collected after informed written consent from women undergoing hysterectomy for benign gynaecological conditions. Only samples from women in the proliferative phase of the menstrual cycle (cycle staging determined by the last menstrual period and measurement of serum estradiol and progesterone levels) were selected for further experiments. The tissue was dissociated using enzymatic digestion with 1 mg/ml of collagenase type IV for two hours at 37 °C, (Sigma C5138), followed by mechanical breakdown using a 70 μm and subsequently 40 μm filter (Falcon, Corning, Cat. Nos 352350 and 352340 respectively). Single cells were plated in RPMI 1640 media (Sigma, Cat. No. R0883) supplemented with 10% heat inactivated foetal calf serum (HIFCS, Gibco, Cat. No. 10082139), 1% L-glutamine (Sigma, Cat. No. G-7513) and 1% Penicillin/Streptomycin (Sigma, Cat. No. P4333).
Primary human endometrial stromal cell (ESC) culture
Prior to any treatments, cells were transferred to phenol red-free RPMI 1640 (Sigma, Cat. No. R7509) containing 10% charcoal stripped fetal calf serum (CSFCS, prepared in house from FCS), 1% L-glutamine and 0.5% gentamycin (Sigma, Cat. No. G1272) for 48 hours prior to use. The antibiotic gentamycin was used instead of the standard penicillin/streptomycin regimen, because it does not inhibit Ct growth31. Cells were maintained at 37 °C and 5% CO2. Treatments were completed in duplicate per experiment and were repeated on 4 (for initial chlamydial growth experiments) or 6 (prolactin/chemokine expression) occasions using cells derived from different patients on each occasion.
In-vitro decidualisation
To assess the effects of chlamydial infection on the decidualisation of primary endometrial stromal cells, an in-vitro decidualisation method previously described was used32. Cells were trypsinised, spun for 3.5 minutes at 800 rpm and resuspended phenol red-free RPMI 1640 with 10% CSFCS. They were then plated at 105 cells per well of a 12-well plate for a minimum of 24 hours. Before the decidualisation protocol commenced, cells were serum starved in 2% CSFCS medium for 24 hours, supplemented as previously described. The cells were treated with 10–6 M progesterone (Sigma, Cat. No. P0130) and 8-Bromo-adenosine 3′-5′-cyclic monophosphate (cAMP, Sigma, Cat. No. A9501) at a final concentration of 0.1 mg/ml. The medium was changed every 48 hours over a six-day period (Supplementary Table S4).
Ct elementary bodies (EBs) growth and purification Chlamydia trachomatis
(Ct) serotype E stock was produced in HEp2 cells using well established protocols. Sub-confluent flasks of HEp2’s were inoculated with Ct. Infected cells were cultured for 48–72 hours in Iscove’s Modified Dulbecco’s Medium (IMDM, Life Technologies, Paisley, UK) supplemented with 2% heat inactivated fetal calf serum (FBS) and 1 µg/ml cycloheximide (Sigma, Cat. No. C4859) (PAA laboratories Ltd, Yeovil, Somerset, UK), until high numbers of mature inclusions were observed by optical microscopy. The cell monolayers were disrupted with glass beads and the medium containing cell debris was briefly sonicated (Vibracell, Sonics & Materials, Connecticut, USA) and centrifuged at 50 × g for 5 minutes at 4 °C to remove intact cells. The supernatant was removed and centrifuged at 12,000 × g using a J-LITE JLA-16.250 rotor (Beckman Coulter Ltd. High Wycombe, UK). The pellet was resuspended by sonication in Tris/KCL and 25 ml of inoculum were layered onto 10 ml 40% Gastrografin® (Bayer plc, Berkshire, UK) and centrifuged at 20,000 rpm at 4 °C for 45 minutes SW 32Ti rotor on Optima L-90K ultracentrifuge (Beckman Coulter Ltd). The pellet was re-suspended into 1 ml Tris/KCL and layered onto discontinuous gradient (54%, 44%, 34% Gastrografin®) and centrifuged at 20, 000 rpm at 4 °C for two hours. The interface between the 44% and 54% layers, containing the EBs was carefully removed. The EBs were washed by resuspension in 10 ml Tris KCL and centrifugation at 20, 000 rpm at 4 °C for a further 45 minutes, to remove residual traces of Gastrografin®. The final pellet was resuspended by sonication in 10 ml of Chlamydia Transport Medium (218 mM Sucrose, 3.76 mM KH2P04, 7.1 mM K2HP04, 4.9 mM L-glutamic acid, 10% FBS, 0.05 mg/ml Gentamycin, 0.1 mg/ml Streptomycin, 150 U/ml Nystatin). and the aliquots stored at −80 °C.
UV inactivation of Ct elementary bodies
To UV inactivate Ct EB’s, 500 μl of inoculum was exposed to 2MJ UV-C. The successful inactivation of Ct was confirmed by cell culture of HeLa cells with UV inactivated bacteria for 96 hours without the presence of any inclusions.
Titration of Ct stock
To determine the titre of the Ct stock, HEp2 cells were plated at 105 per well of 8 well glass chamber slides, infected with serial tenfold dilutions of Ct inoculum (Multiplicity of infection [MOI] 1–1 × 10−8) and cultured for 48 hours. The cells were fixed in ice-cold acetone for 5 minutes, left to air dry and stored at −20 °C. After thawing, the slides were rehydrated in PBS and incubated with the primary antibody (C. abortus LPS, Santa Cruz Biotechnology, Cat. No. 13/4) for 1 hour at room temperature. Following PBS washes, the slides were incubated with a fluorescein isothiocyanate (FITC)-conjugated anti-mouse antibody (Sigma) for 60 minutes at room temperature in a light-tight humidity chamber. Slides were mounted using ProLong® Gold Antifade Mountant with DAPI (lifeTechonologies, Cat. No. P36930). The total number of inclusions per well was counted at serial dilutions 10−5–10−7 and the titre of stock was calculated.
Ct infection of decidualised ESC
ESC were seeded at 12-well plates and decidualised as described above. On day 6, ESC were infected with Ct MOI 0.01, 0.1, 1, 2 and 3 or UV-Ct at the same MOI for 48 hours prior to sample collection. To determine whether decidualisation of ESC was affected by exposure to lipopolysaccharide (LPS) from other bacterial species, decidualised ESC isolated from a further five subjects were exposed to LPS from either E. coli or S. Minnesota (both Invivogen) (each at 200 μg/ml and 100 μg/ml) for 48 hours prior to sample collection.
RNA extraction, quantification and quality assessment
To assess changes in expression levels of genes of interest, total RNA extraction was performed Using the RNeasy® Micro kit (Qiagen, Crawley, UK, Cat. No. 74004) following the manufacturer’s protocol.
Real time PCR
All reactions were carried out in 384 well plates at a final reaction volume of 10 μl per sample, as shown in Supplementary Table S5. The samples were mixed by repeat pipetting and centrifuged in a mini plate spinner (Applied Biosystems, Warrington, UK) at 1000 g for 20 seconds to remove any bubbles. A standard curve of cDNA made from standardised placenta RNA (Ambion, Cat. No. AM7950) was used on every plate to ensure comparability among all plates. All primers were predesigned KiCqStart® SYBR® Green Primers (Cat. No. KSPQ12012).
PCR conditions were an initial step of 3 min at 95 °C, followed by 40 cycles of 5 s at 95 °C, 15 s at 60 °C and a final disassociation step consisting of 15 s at 95 °C, 15 s at 60 °C and finally 15 s at 95 °C.
DNA extraction from ESC
To extract DNA from infected cells, the MagMAX™ Total Nucleic Acid Isolation Kit (Ambion, Cat. No. AM1840) was used according to the manufacturers’ protocol.
Ct plasmid copy assay
To assess whether Ct could proliferate in ESC the ‘Chlamydia trachomatis Genesig Standard Kit’ (PrimerDesign, Cat. No Path-C.trachomatis-standard) was used. A standard curve of known concentration provided by the kit was used to absolutely quantify Ct plasmid copy numbers in DNA extracts from ESC cells using qPCR.
Human Chemokine Array
Proteome Profiler™ Human Chemokine Array Kit (R&D Systems, Abingdon, UK, Cat. No. ARY017) was used. Samples were pooled supernatants from ESC cultures and the manufacturer’s protocol was followed without deviation. The data are expressed as percentage of positive control of each membrane and pixel density was measured using ImageJ.
Enzyme-linked Immunoabsorbant Assay (ELISA)
The sandwich ELISA was used to detect PRL (R&D Systems, Cat. No. DY682) protein concentrations as per manufacturer’s instructions. Sample concentrations were calculated using MasterPlex® QT (Hitachi) software.
Magnetic Luminex®Screening Assay
The Luminex®Screening Assay (R&D Systems, Cat. No. LXSAHM) was used to detect CXCL12 and CXCL16 as per manufacturer’s instructions.
Statistical analysis
Statistical analysis was carried out using GraphPad Prism 6. Friedman’s non parametric test was used for all sample sets. The “n” number corresponds to individual patients.
Electronic supplementary material
Acknowledgements
We would like to thank Professor Hilary Critchley for kindly donating the primary endometrial stromal cells used for this study. This work was funded by an MRC Centre Grant (MRC G1002033) and Tommy’s.
Author Contributions
A.H., G.E., and S.H. conceived the project. S.G., N.W., J.W., I.S., D.G., P.S. performed experimental work. S.G., S.H. and A.H. analysed the data. S.G., S.H., J.B., P.H. and G.E. and A.H. wrote the manuscript. S.G., S.H., N.W. and A.H. prepared the figures and the supplementary file. All authors reviewed the manuscript.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Electronic supplementary material
Supplementary information accompanies this paper at doi:10.1038/s41598-017-02223-z
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Giakoumelou S, et al. The role of infection in miscarriage. Hum. Reprod. Update. 2016;22:116–33. doi: 10.1093/humupd/dmv041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Arsovic A, Nikolov A, Sazdanovic P, Popovic S, Baskic D. Prevalence and diagnostic significance of specific IgA and anti-heat shock protein 60 Chlamydia trachomatis antibodies in subfertile women. Eur. J. Clin. Microbiol. Infect. Dis. 2014;33:761–766. doi: 10.1007/s10096-013-2008-4. [DOI] [PubMed] [Google Scholar]
- 3.Baud D, et al. Role of Chlamydia trachomatis in miscarriage. Emerg. Infect. Dis. 2011;17:1630–5. doi: 10.3201/eid1709.100865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kortekangas-Savolainen O, Mäkinen J, Koivusalo K, Mattila K. Hospital-diagnosed late sequelae after female Chlamydia trachomatis infections in 1990–2006 in Turku, Finland. Gynecol. Obstet. Invest. 2012;73:299–303. doi: 10.1159/000334822. [DOI] [PubMed] [Google Scholar]
- 5.Wilkowska-Trojniel M, et al. The influence of Chlamydia trachomatis infection on spontaneous abortions. Adv. Med. Sci. 2009;54:86–90. doi: 10.2478/v10039-009-0008-5. [DOI] [PubMed] [Google Scholar]
- 6.Licciardi F, Grifo J. Relation between antibodies to Chlamydia trachomatis and spontaneous abortion following in vitro fertilization. J. Assist. …. 1992;9:207–10. doi: 10.1007/BF01203814. [DOI] [PubMed] [Google Scholar]
- 7.Singh, N. et al. Does aberrant expression of cyclooxygenase-2 and prostaglandin-E2 receptor genes lead to abortion in Chlamydia trachomatis-infected women. J. Matern. Neonatal Med. 1–6, 10.3109/14767058.2015.1031741 (2015). [DOI] [PubMed]
- 8.He Y-Y, et al. The decidual stromal cells-secreted CCL2 induces and maintains decidual leukocytes into Th2 bias in human early pregnancy. Clin. Immunol. 2012;145:161–73. doi: 10.1016/j.clim.2012.07.017. [DOI] [PubMed] [Google Scholar]
- 9.Nancy P, et al. Chemokine gene silencing in decidual stromal cells limits T cell access to the maternal-fetal interface. Science. 2012;336:1317–21. doi: 10.1126/science.1220030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Du M-R, Wang S-C, Li D-J. The integrative roles of chemokines at the maternal-fetal interface in early pregnancy. Cell. Mol. Immunol. 2014;5:438–448. doi: 10.1038/cmi.2014.68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Plaisier M, et al. Decidual vascularization and the expression of angiogenic growth factors and proteases in first trimester spontaneous abortions. Hum. Reprod. 2009;24:185–197. doi: 10.1093/humrep/den296. [DOI] [PubMed] [Google Scholar]
- 12.Salker M, et al. Natural selection of human embryos: Impaired decidualization of endometrium disables embryo-maternal interactions and causes recurrent pregnancy loss. PLoS One. 2010;5:e10287. doi: 10.1371/journal.pone.0010287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bao L, et al. Decidual prolactin silences the expression of genes detrimental to pregnancy. Endocrinology. 2007;148:2326–34. doi: 10.1210/en.2006-1643. [DOI] [PubMed] [Google Scholar]
- 14.Rasmussen SJ, et al. Secretion of proinflammatory cytokines by epithelial cells in response to Chlamydia infection suggests a central role for epithelial cells in chlamydial pathogenesis. J. Clin. Invest. 1997;99:77–87. doi: 10.1172/JCI119136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Buchholz KR, Stephens RS. Activation of the host cell proinflammatory interleukin-8 response by Chlamydia trachomatis. Cell. Microbiol. 2006;8:1768–79. doi: 10.1111/j.1462-5822.2006.00747.x. [DOI] [PubMed] [Google Scholar]
- 16.Rödel J, et al. Persistent Chlamydia trachomatis infection of HeLa cells mediates apoptosis resistance through a Chlamydia protease-like activity factor-independent mechanism and induces high mobility group box 1 release. Infect. Immun. 2011;80:195–205. doi: 10.1128/IAI.05619-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tait IA, Duthie SJ, Taylor-Robinson D. Silent upper genital tract chlamydial infection and disease in women. Int J STD AIDS. 1997;8:329–331. doi: 10.1258/0956462971920037. [DOI] [PubMed] [Google Scholar]
- 18.Buendía AJ, et al. Kinetics of Infection and Effects on Placental Cell Populations in a Murine Model of Chlamydia psittaci -Induced Abortion. Infect. Immun. 1998;66:5–12. doi: 10.1128/iai.66.5.2128-2134.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wittenbrink MM, Schoon HA, Schoon D, Mansfeld R, Bisping W. Endometritis in cattle experimentally induced by Chlamydia psittaci. Zentralbl Vet. B. 1993;40:437–450. doi: 10.1111/j.1439-0442.1993.tb00650.x. [DOI] [PubMed] [Google Scholar]
- 20.Kintner J, Schoborg RV, Wyrick PB, Hall JV. Progesterone antagonizes the positive influence of estrogen on Chlamydia trachomatis serovar E in an Ishikawa/SHT-290 co-culture model. Pathog. Dis. 2015;73:ftv015–ftv015. doi: 10.1093/femspd/ftv015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kaushic C, Grant K, Crane M, Wira CR. Infection of polarized primary epithelial cells from rat uterus with Chlamydia trachomatis: cell-cell interaction and cytokine secretion. Am. J. Reprod. Immunol. 2000;44:73–79. doi: 10.1111/j.8755-8920.2000.440202.x. [DOI] [PubMed] [Google Scholar]
- 22.Mackern Oberti JP, et al. Chemokine response induced by Chlamydia trachomatis in prostate derived CD45+ and CD45− cells. Reproduction. 2011;142:427–437. doi: 10.1530/REP-11-0163. [DOI] [PubMed] [Google Scholar]
- 23.McQueen DB, Bernardi LA, Stephenson MD. Chronic endometritis in women with recurrent early pregnancy loss and/or fetal demise. Fertil. Steril. 2014;101:1026–30. doi: 10.1016/j.fertnstert.2013.12.031. [DOI] [PubMed] [Google Scholar]
- 24.Cicinelli E, et al. Prevalence of chronic endometritis in repeated unexplained implantation failure and the IVF success rate after antibiotic therapy. Hum. Reprod. 2014;0:1–8. doi: 10.1093/humrep/deu292. [DOI] [PubMed] [Google Scholar]
- 25.Jabbour HN, Critchley HOD. Potential roles of decidual prolactin in early pregnancy. Reproduction. 2001;121:197–205. doi: 10.1530/rep.0.1210197. [DOI] [PubMed] [Google Scholar]
- 26.Warner JA, et al. Human cytomegalovirus infection inhibits CXCL12- mediated migration and invasion of human extravillous cytotrophoblasts. Virol. J. 2012;9:255. doi: 10.1186/1743-422X-9-255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Huang Y, et al. Chemokine CXCL16, a scavenger receptor, induces proliferation and invasion of first-trimester human trophoblast cells in an autocrine manner. Hum. Reprod. 2006;21:1083–91. doi: 10.1093/humrep/dei436. [DOI] [PubMed] [Google Scholar]
- 28.Isozaki T, Arbab A, Haas C. Evidence that CXCL16 is a potent mediator of angiogenesis and is involved in endothelial progenitor cell chemotaxis: studies in mice with K/BxN serum-induced arthritis. Arthritis …. 2013;65:1–20. doi: 10.1002/art.37715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Quenby S, et al. Uterine natural killer cells and angiogenesis in recurrent reproductive failure. Hum. Reprod. 2009;24:45–54. doi: 10.1093/humrep/den348. [DOI] [PubMed] [Google Scholar]
- 30.Helige C, et al. Distribution of decidual natural killer cells and macrophages in the neighbourhood of the trophoblast invasion front: A quantitative evaluation. Hum. Reprod. 2014;29:8–17. doi: 10.1093/humrep/det353. [DOI] [PubMed] [Google Scholar]
- 31.Bowie WR, Lee CK, Alexander ER. Prediction of efficacy of antimicrobial agents in treatment of infections due to Chlamydia trachomatis. J InfectDis. 1978;138:655–659. doi: 10.1093/infdis/138.5.655. [DOI] [PubMed] [Google Scholar]
- 32.Brosens JJ, et al. Human endometrial fibroblasts immortalized by simian virus 40 large T antigen differentiate in response to a decidualization stimulus. Endocrinology. 1996;137:2225–2231. doi: 10.1210/endo.137.6.8641169. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


