Abstract
Exposure to airborne influenza (or flu) from a patient's cough and exhaled air causes potential flu virus transmission to the persons located nearby. Hospital-acquired influenza is a major airborne disease that occurs to health care workers (HCW).
This paper examines the airflow patterns and influenza-infected cough aerosol transport behavior in a ceiling-ventilated mock airborne infection isolation room (AIIR) and its effectiveness in mitigating HCW's exposure to airborne infection. The computational fluid dynamics (CFD) analysis of the airflow patterns and the flu virus dispersal behavior in a mock AIIR is conducted using the room geometries and layout (room dimensions, bathroom dimensions and details, placement of vents and furniture), ventilation parameters (flow rates at the inlet and outlet vents, diffuser design, thermal sources, etc.), and pressurization corresponding to that of a traditional ceiling-mounted ventilation arrangement observed in existing hospitals. The measured data shows that ventilation rates for the AIIR are about 12 air changes per hour(ach). However, the numerical results reveals incomplete air mixing and that not all of the room air is changed 12 times per hour. Two life-sized breathing human models are used to simulate a source patient and a receiving HCW. A patient cough cycle is introduced into the simulation and the airborne infection dispersal is tracked in time using a multiphase flow simulation approach.
The results reveal air recirculation regions that diminished the effect of air filtration and prolong the presence of flu-contaminated air at the HCW's zone. Immediately after the patient coughs (0.51 s), the cough velocity from the patient's mouth drives the cough aerosols toward the HCW standing next to patient's bed. Within 0.7 s, the HCW is at risk of acquiring the infectious influenza disease, as a portion of these aerosols are inhaled by the HCW.
As time progresses (5 s), the aerosols eventually spread throughout the entire room, as they are carried by the AIIR airflow patterns. Subsequently, a portion of these aerosols are removed by the exhaust ventilation. However, the remaining cough aerosols reenter and recirculate in the HCW's zone until they are removed by the exhaust ventilation.
The infectious aerosols become diluted in the HCW's region over a period of 10 s because of the fresh air supplied into the HCW's zone. The overall duration of influenza infection in the room (until the aerosol count is reduced to less than 0.16% of the total number of aerosols ejected from the patient's mouth) is recorded as approximately 20 s. With successive coughing events, a near-continuous exposure would be possible. Hence, the ceiling-ventilation arrangement of the mock AIIR creats an unfavorable environment to the HCW throughout his stay in the room, and the modeled AIIR ventilation is not effective in protecting the HCW from infectious cough aerosols. The CFD results suggest that the AIIR ceiling ventilation arrangement has a significant role in influencing the flu virus transmission to the HCW.
Introduction
Influenza is a virulent human flu that can cause a global outbreak or serious illness. The annual number of influenza deaths is 250–500 thousand worldwide (WHO 2014; Thompson et al. 2003) and requires 114,000 hospitalizations (Bridges et al. 2002). Influenza outbreaks occur frequently in health care facilities and often more than once in a single facility during an influenza season (Patriarca et al. 1985; Drinka et al. 2000; Sugaya et al. 1996; Zadeh et al. 2000). In the case of a influenza outbreak, health care workers (HCWs) are at high risk as they are typically in close contact with influenza patients. HCWs often continue to work despite being ill (CDC 1999; Wenzel et al. 1977; Wilde et al. 1999). Influenza can then spread rapidly among other patients and HCWs, particularly in closed hospital settings (Cunney et al. 2000; Evans et al. 1997).
Relevance to National Occupational Research Agenda (NORA)
The National Institute for Occupational Safety and Health (NIOSH) developed the National Occupational Research Agenda (NORA) for improving the health of workers in indoor work environments. It identified priority research areas to substantially decrease the work-related illnesses and deaths in the upcoming years. NORA's Health Care and Social Assistance (HCSA) Agenda is one element of the larger NORA endeavor. There are over 15 million healthcare-associated employees in the U.S. that are covered by the HCSA research agenda. According to the NIOSH NORA HCSA Sector, an estimated 18.6 million people are employed within the HCSA sector (CDC 2013). About 80% of HCSA workers are in health care industries and 20% in social assistance industries. Compared to other industrial sectors, the HCSA sector had the second largest number of occupational injuries and illnesses (CDC 2009). One specific research priority within the HCSA research agenda is the prevention of occupationally-acquired transmissions of infectious disease among HCSA workers. The research discussed in this manuscript is in direct response to this HCSA research priority.
The HCSA Sector Council has developed five goals designed to address top safety and health concerns, to advance protection to HCWs and, at the same time, ensure patient safety (CDC 2009). The HCSA Sector Council five strategic goals are
Safety and health programs
Musculoskeletal disorders
Hazardous drugs and other chemicals
Sharps injuries
Infectious diseases.
Motivation
The mission of the research discussed in this manuscript is to prevent occupational transmission of infectious influenza disease to HCWs.
Airborne Infection Isolation Room (AIIR)
Use of an airborne infection isolation room (AIIR) is prescribed by various federal and state health organizations (Ninomura et al. 2001; FGI 2014; ASHRAE 2013) when HCWs have to conduct cough-generating procedures on influenza patients. In using an AIIR, the infectious flu virus is contained within the room and its concentration in the room is reduced via dilution ventilation. This is done by following ASHRAE and Facility Guidelines Institute (FGI) guidelines (Ninomura and Bartley 2001; FGI 2014; ASHRAE 2013; Heiselberg 1996) on the construction of AIIRs and their ventilation system design. The airflow and ventilation design parameters are controlled in AIIRs to reduce the potential for airborne migration of infectious aerosols into other areas of the hospital.
Objective
The objective of this study is the computational analysis of the airflow patterns and influenza-infected cough aerosol transport behavior in a ceiling-ventilated AIIR and determination of the ceiling ventilation's effectiveness in mitigating a HCW's exposure to airborne infectious aerosols.
Methodology
Problem Statement
The Mock AIIR layout and its ventilation parameters at NIOSH's Alice Hamilton Research Laboratories in Cincinnati, OH, is considered for the present study. Figure 1 presents the isometric view of the geometric model and the configuration details of the mock AIIR modeled at the NIOSH facility. The modeled room dimensions were: length = 170 in. (4.318 m), width = 96 in. (2.4384 m), height = 192 in. (4.879 m). The corresponding computer-aided design (CAD) model was constructed for the mock AIIR, and a computational grid was generated using the ANSYS ICEM computational fluid dynamics (CFD) software (ANSYS 2016a).
Figure 1.

Schematic layout of the mock AIIR.
This mock isolation room consists of two ceiling inlet supply vents (square and linear supply) and one ceiling exhaust grille, a bathroom with an exhaust vent, and a room entrance/exit main door for the patient and HCW. The ceiling air supply diffusers used in the mock AIIR are shown in Figure 2.
Figure 2.

(a) Linear supply slot diffuser—isometric view. (b) Linear supply slot diffuser with dimensions (in.)—front view. (c) Square supply diffuser— isometric view. (d) Square supply with dimensions (in.)—plane view.
The supply diffusers for the mock AIIR are selected to provide minimum ventilation rates and indoor air quality that is acceptable to the patient and HCW as per ANSI/ASHRAE/ ASHE Standard 170 (ASHRAE 2013).
A rectangular linear diffuser (Figure 2a and 2b) with two air slots is used for the ceiling installation. The standard diffuser module length is 48 in. (1.219 m). The length of each slot is 46.25 in. (1.17 m) and the thickness of each slot is 0.718 in. (0.018 m). The two slots are equipped with air deflectors so the direction of air discharge can be adjusted. Supply air flows at an angle of 45° from the slot closest to the window and directly downward (vertical air distribution) for the second slot furthest from the window. The total supply flow rate from the linear diffuser is 81.49 cfm (138.45 m3/h).
A square diffuser with two slots is used for the ceiling installation (Figure 2c and 2d). The standard diffuser module length is 24 in. (0.6 m). The thicknesses of the outer and inner slots are 2.7 and 2.9 in. (2.685 and 0.073 m), respectively. Supply air flows at an angle of 45° outward into the room. The total supply flow rate from the square diffuser is 66.67 cfm (113.27 m3/h). The supply air temperature from the linear and square diffusers is 69.72°F (20.96°C).
The patient and HCW are modeled using the NASA anthropometric data from the Human Integration Design Handbook (NASA 2014). Table 1 gives the patient's and HCW's dimensions. The patient is lying on the bed inclined at 30° and the HCW is facing the patient at a distance of 3.46 in. (0.087 cm) from the patient on the side of the monitoring instrumentation location.
Table 1. Patient and HCW Dimensions (NASA 2014).
| Body Part | Length, in. (m) | Width, in. (m) | Height, in. (m) |
|---|---|---|---|
| Head | 14.96 (0.38) | 6.29 (0.16) | 11.14 (0.283) |
| Trunk | 19.68 (0.5) | 9.84 (0.25) | 25.66 (0.652) |
| Hands | 3.93 (0.1) | 3.93 (0.1) | 30.82 (0.783) |
| Legs | 5.11 (0.13) | 5.11 (0.13) | 5.9 (0.15) |
| Mouth | Diameter: 1.18 in. (0.03 m) | ||
The mouth diameter is taken to be 1.18 in. (0.03 m) (VanSciver et al. 2011) created at the patient's face for cough flow from the patient's mouth. The nose opening dimensions were modeled with an area of 0.116 in.2 (7.5×10−5 m2) (Gupta et al. 2009) at the patient's and HCW's face centers for breathing. Figure 3 shows the two semicircular nose openings and mouth opening at the HCW's face.
Figure 3.

Patient and HCW's face showing nose and mouth openings.
Boundary and Operating Conditions
The fresh air inflow of 148.16 cfm (251.72 m3/h) into the AIIR is caused by the airflow from the main supply (the linear inlet diffuser inflow rate is 81.49 cfm [138.45 m3/h] and the square supply inflow rate is 66.67 cfm [113.27 m3/h]) and the outflow of 225 cfm (382.27 m3/h) is due to the airflow through the main exhaust and the leakage into the bathroom. The boundary and operating conditions of the ceiling-ventilated mock AIIR are presented in Table 2.
Table 2. Ceiling-Ventilated Mock AIIR—Boundary and Operating Conditions.
| No. | Boundary | Boundary Condition | Boundary Value Required |
|---|---|---|---|
| 1. | Linear inlet diffuser | Number of slots in the linear diffuser | 2 |
| Flow rate | 55% of Qin = 81.49 cfm (138.45 m3/h) | ||
| Angle | 45° | ||
| Direction of flow: Toward the window (for the slot closest to window); directly downward for the second slot furthest from window. | |||
| 2. | Square supply | Flow rate | 45% of Qin = 66.67 cfm (113.27 m3/h) |
| Angle | 45° | ||
| Direction of flow: Outward, air flows into the room. | |||
| 3. | Main room exhaust | Flow rate | 225 cfm (382.27 m3/h) |
| Direction of flow: Into the exhaust vent, air extracted from the room. | |||
| 4 | Bathroom exhaust | Flow rate | 80 cfm (135.92 m3/h) |
| 5 | Main door gaps | Pressure at the main door gaps | −0.01 in. w.g. (−2.49 Pa) |
| 6. | Bathroom door gaps (assuming bathroom receives 10% of its exhaust makeup air from leaks other than the bathroom entry door) | Flow rate | 72 cfm (122.32 m3/h) |
| Direction of flow: Into bathroom, room air escapes through bathroom door gaps. | |||
| 7. | Overhead lights | Power (W) | 0 |
| 8. | HCW lower body | Temperature | 69.72°F (20.96°C), room temperature |
| 9. | HCW head and face | 97.71°F (36.51°C), normal body temperature | |
| 10 | Patient head and mouth | 100.72°F (38.18°C), patient with fever | |
| 11 | Window near patient | 61.88°C (16.6°C), outdoor temperature | |
The bathroom exhaust flow rate is 80 cfm (135.92 m3/h). It is assumed that the bathroom receives 10% of its exhaust makeup air from leaks other than the bathroom entry door. Thus, a flow rate of 72 cfm (122.32 m3/h) was specified at the gap around the bathroom door. A pressure of −0.01 in. w.g. (−2.49 Pa) was specified as the boundary condition at the gaps around the AIIR's main entry door.
The geometric model, ventilation parameters, and room pressurization of the ceiling-ventilated mock AIIR are numerically simulated along with the HCW's potential to inhale the patient's cough aerosols as follows and as indicated in Table 2.
At Patient's Mouth. Cough flow boundary condition is specified at the patient's mouth. The patient introduces cough aerosols 1 micron (μm) in diameter during coughing. This aerosol size is consistent with deep-lung penetration and airborne infectious disease transmission and thus was the focus of the model. The patient's cough cycle, taken from the research by Gupta et al. (2009), is shown in Figure 4, and the cough flow parameters are represented in Table 3.
At HCW's Mouth. A no-slip boundary condition is specified at the HCW's mouth.
At Patient's and HCW's Noses. The patient and HCW are breathing out-of-phase with a maximum volume airflow of 0.5 L (0.132 gal). Figure 5 and Table 4 show normal breathing cycle of patient and HCW and the corresponding breathing profile parameters obtained by digitizing the volume of breath curve, taken from the research by Hall (2011). It is observed that expiration takes a relatively longer time (2.29 s) than inspiration (1.71 s). Also, expiration starts at a faster pace in initial time instants (between 1.71 and 2.5 s) and slows down as it approaches 4 s.
At Patient's and HCW's Bodies. To account for a fever, it is assumed that the patient's head is at a temperature of 100.72°F (36.51°C) and the rest of the body is assumed to be covered with a sheet at room temperature. For the HCW, a normal body temperature of 97.71°F (36.51°C) is specified for the HCW's head and the rest of the body is assumed to be covered with clothes at room temperature.
For Furniture. The patient's bed is modeled as wall boundary and maintained at a normal room temperature of 69.72°F (20.96°C).
At Window. The window in the room is considered an isothermal wall. The temperature for the window facing outside is specified at outside climatic conditions of a day at 61.88°F (16.6°C).
At the Overhead Lights. A heat flux of 0 W is specified at the boundary.
Materials. The materials used in the model and their properties are listed in Table 5.
Figure 4.

Single cough flow cycle from patient's mouth.
Table 3. Patient Cough Cycle Profile Parameters.
| Time Period, s | Coughing Time of Patient, s | Average Cough Volume, L (gal) | Cough Droplets Size, μm |
|---|---|---|---|
| 0.5 | between 0 and 0.5 s | 2.41 (0.636) | 1 |
Figure 5.

Normal breathing cycle.
Table 4. Breathing Profile Parameters.
| Maximum Volume Flow of Air, L (gal) | 0.5 (0.132) |
| Time Period of Breath, s | 4 |
| Time Period of Inspiration Cycle, s | 1.71 |
| Time Period of Expiration Cycle, s | 2.29 |
Table 5. Material Properties.
| Object | Material | Conductivity, W/m·K (Btu/ft·h·°F) | Specific Heat, J/kg·K (Btu/lb·°F) | Density, kg/m3 (lb/ft3) |
|---|---|---|---|---|
| Window | Window glass | 0.96 (0.55) | 840 (0.2) | 2500 (156) |
| Patient and HCW | Human skin | 0.206 (0.12) | 3558 (0.85) | 1027 (64) |
| Overhead lights | Plastic | 1.005 (0.58) | 1670 (0.39) | 1250 (78) |
| Doors | Wood | 0.173 (0.09) | 2310 (0.55) | 700 (43) |
| Room walls | Dry wall | 3.1 (1.79) | 1090 (0.26) | 600 (37.4) |
| Patient bed | Cotton fabric | 0.043 (0.025) | 1162 (0.27) | 1540 (96) |
| Floor | PVC flooring | 0.28 (0.16) | 900 (0.21) | 1450 (90.5) |
Solution Procedure
The transient, three-dimensional, incompressible Navier-Stokes equations, including gravity, were solved with the pressure-velocity coupling achieved using the Semi-Implicit Pressure-Linked Equations (SIMPLE) algorithm developed by Patankar and Spalding (1972). The energy equation was also solved to account for temperature variations. The transport equations were discretized using a second-order upwind scheme with second-order implicit discretization for the temporal terms. Turbulence was modeled using the realizable k-ε model. Numerical analysis was performed using the commercial CFD software FLUENT (ANSYS 2016b) based on the finite volume method. Over each control volume, the SIMPLE algorithm is used to iteratively solve for these governing equations.
Initially, the steady-state flow field in the AIIR was determined before the patient's coughing and the HCW's breathing were initiated. The convergence criteria for the steady-state flow field were set at 10−4 for all equations.
The patient cough cycle consists of a mixture of cough aerosol and air for a 0.5 s cough cycle. The aerosol mixture has about 6000–7000 cough aerosols contained in it, which is consistent with data showing the number of aerosols ejected from a cough is of the order of 1000–10000 (Kowalski and Bahnfleth 1998).
A Lagrangian discrete phase model is used to track cough aerosols. These cough aerosols were allowed to collide or break up. They are also allowed to coalesce or bounce based on the collision angle. Cough aerosols stick to the surface when they fall to the ground or are bounced off by the wall surface due to high-velocity collision and escape when they reach the exhaust ventilation. The patient coughs for a time period of 0.5 s (one cough cycle) while the HCW continues breathing. At the end of the cough cycle, the patient resumes a normal breathing cycle and the cough aerosols' dispersal throughout the mock AIIR is tracked in time until all the aerosols are entrained from the room by the exhaust ventilation. The results present the cough aerosol dispersal behavior, their locations at several time instants, and the analysis of the HCW's risk exposure to the infectious cough aerosols.
The time step chosen for the transient simulation was 0.001 s. The solution is assumed to be converged when the convergence residuals change by less than 10−4.
Three grid densities—Case 1: 752,437 cells, Case 2: 1,375,914 cells, and Case 3: 2,820,484 cells—were investigated to perform the gird sensitivity study. The grids are unstructured, and cells are tetrahedral. Figure 6 presents the results of the grid independence test. Here V refers to the velocity magnitude of room air monitored at a specific location close to the patient, VO is the maximum velocity magnitude of room air at the boundary condition of velocity inlet, H is the height of room air monitored at the specific location close to the patient, and HO is the room height. The dimensionless velocity at the monitored points in Case 2 was quite close to the one in Case 3. As the difference between Cases 2 and 3 was insignificant, it could be concluded that the grid system reached an independent solution. Therefore, the grid density in Case 2 was found to be sufficient and applied for this study.
Figure 6.

Grid independence test.
Results and Discussion
Investigation of Airflow Patterns in the Mock AIIR
The mock AIIR design consists of two inlet supply vents and one exhaust vent, a bathroom with exhaust vent, and the room entrance/exit main door for patient and HCW transit. Figure 7 shows the airflow streamlines in the isometric view of the mock AIIR of 4.3 × 2.4 × 4.8 m (14.1 × 7.87 × 15.7 ft). The fresh air jets are ejected from the square supply inlet, at Location 2 on the room ceiling at an angle of 45° to the ceiling. The square supply vent is positioned to provide a clean-to-dirty fresh air supply from the HCW's nose and mouth zone to the contaminated patient zone. However, the fresh air from the square supply is recirculated in the patient's and HCW's regions (at Location 5) instead of being immediately removed by the exhaust vent (Location 6). The presence of the room air recirculation in front of the HCW and above the patient's body might diminish the dilution mixing effect of the mock AIIR air and could result in accumulation or prolonged presence of the influenza-virus-contaminated air, thus preventing a more rapid removal of the flu-contaminated air from the isolation room. This suggests that the HCW is at risk of exposure to the patient's infectious influenza aerosol. Nonetheless, the flu virus concentration in HCW's region is lowered because of fresh air supply from the inlet square vent.
Figure 7.

Investigation of airflow patterns in the AIIR.
In addition to the square supply vent, as shown in Figure 7, fresh air also flows downward to the floor from linear supply vents, at Location 1 on the room ceiling. The linear supply vent has two slots, one directing air at 45° toward the window and the second slot discharging supply air directly downward towards the floor. These ceiling supply vents are positioned to ensure fresh airflow throughout the AIIR. Air jets from the linear supply vent reach the floor at Locations 3 and 4 and are deflected to the other side (bathroom wall) of the room. After deflection, the rising air jet from the linear supply vent is circulated throughout the room. Room air after circulation in the room is continuously extracted by the ceiling exhaust vent (at Location 6) positioned on the room ceiling near the main door.
Tracking the Transient Cough Aerosol Dispersal Immediately After Patient's Cough in the AIIR
The patient's cough, consisting of a mixture of air and cough aerosols of 1 μ diameter, is ejected from the patient's mouth between the time interval of 0 and 0.5 seconds, i.e., one cough cycle.
Figure 8 shows the close-up side view of the transient dispersal of influenza cough aerosols immediately after the patient's cough (at 0.51 s) in the mock AIIR. The cough velocity from the patient's mouth propels the aerosols forward. The mock AIIR square and linear supplies as well as the ceiling exhaust ventilation locations and airflow rates do not have significant influence on the cough aerosol dispersal at this time instant because of their relatively low velocities compared to the patient's cough.
Figure 8.

Tracking the transient cough aerosol dispersal immediately after the patient's cough in the AIIR.
A total number of 6000–7000 cough aerosols are ejected from the patient's mouth. The cough aerosols count decreases over time because of the aerosol's coalescence right after they are expelled from the patient's cough.
Immediately after the patient's cough (at the time instant of 0.51 s), the cough aerosols remain airborne and are seen as a cloud formation rising in the air directly above the patient. The cloud of cough aerosols is well formed instead of being immediately dispersed in random directions throughout the room because of the presence of fairly low room air velocity in the patient's region. During this time, the probability of cough aerosol coalescence is high because these cough aerosols are quite close to each other.
The cough aerosols from the patient's region start to move toward the direction of the HCW at this time instant (0.51s). However, the aerosols haven't reached the HCW's nose zone. Hence, the HCW standing next to the patient's bed is unaffected by the influenza-laden aerosol at the early time instants immediately after the patient's cough.
Cough Aerosols Dispersal in the Patient's and HCW's Zones of the Mock AIIR
The cough aerosols are dispersed with in the patient's and HCW's zones till approximately 0.7 s, as shown in Figure 9. At the time instant of 0.6 s (shown in Figure 9a), a portion of cough aerosols are inhaled at the patient's nose zone, as the patient resumes a normal breathing (inhalation phase) cycle immediately after cough. This is because the cough aerosols with relatively low residence times are in the close vicinity of the patient's nose zone while the cough aerosols with high residence times, driven by the cough velocity, travel towards the HCW's direction and rise to the height of HCW's head level.
Figure 9.

Cough aerosol dispersal in the patient's zone.
However, the cough aerosols do not rise above the height of the HCW's head level. This is caused by the jets of fresh air from the ceiling square supply vents flowing in the opposite direction into the room as shown in Figure 10.
Figure 10.

Airflow pathlines from square supply vent.
At approximately 0.7 s (Figure 9b), a few cough aerosols with low residence times are present in the patient's zone. The heavier cough aerosols (>10 μ in diameter) travel only short distances and eventually fall to the ground (patient's bed or floor). Hence, these aerosols that settle on the surfaces are not inhaled by the HCW and do not cause inhalational flu infection risk to the HCW.
The cough aerosols reach the inhaling zone of the HCW and rise to the HCW's head level or reach below the HCW's lower body region. Since the HCW is in the inhalation phase of the normal breathing cycle, some of these aerosols are inhaled by the HCW. This is one of the critical stages in terms of the HCW's exposure to airborne virus: the aerosols that are in close proximity to the HCW pose a potential risk of acquiring the infectious disease.
Cough Aerosol Dispersal Throughout the Isolation Room
The cough aerosols tend to disperse away from the patient's and HCW's regions as shown in Figure 11a. At 0.8 s, the aerosols have traveled far away, toward the opposite wall of the patient's bed (front wall) of the room. By this time, coalescence losses have decreased because the aerosols are no longer in very close proximity to each other, the cough aerosols are dispersed, and their movement is primarily influenced by the AIIR airflow patterns.
Figure 11.
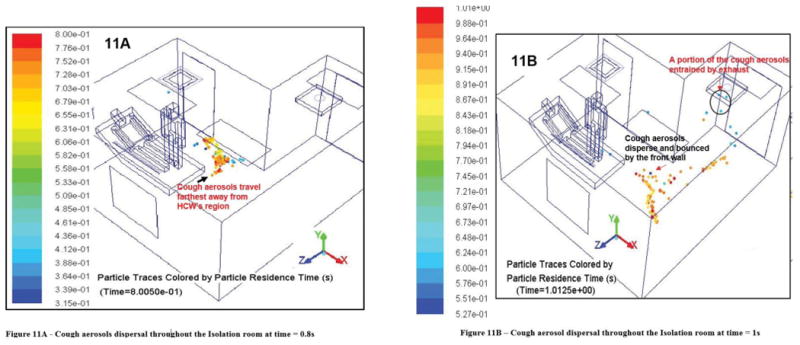
Cough aerosols dispersal throughout the isolation room at times (a) 0.8 s and (b) 1 s.
The cough aerosol cluster is also deflected by the front wall of the room at the time instant of 1 s, as shown in Figure 11b. Subsequently, a part of the air that is contaminated by the flu virus aerosols in the region near the room front wall is pulled into the ceiling exhaust vent. The cough aerosol entrainment from the AIIR starts at 1 s. This results in a portion of the aerosols gradually beginning to move toward the main exhaust without encountering the HCW. While most of the aerosols are located close to the front wall region, the remaining cough aerosols are observed moving towards the patient's and HCW's zones after being deflected by the front wall.
Therefore, even though a portion of the cough aerosols are entrained by the exhaust ventilation and the cough aerosols traveled away from the HCW's region between the time intervals 0.8 and 1 s, the AIIR ceiling ventilation is not effective in preventing the cough aerosols from reentering the HCW's inhaling zone.
Evaluation of Cough Aerosol Dispersal Behavior and AIIR Ventilation Effectiveness in Preventing Influenza Infection to the HCW
Figure 12 presents the investigation of cough aerosol dispersal behavior and the AIIR ventilation effectiveness in preventing patient cough aerosols from entering the HCW's breathing zone. It can be observed that as the cough aerosols are deflected by the front wall of the room within the first second after the patient's cough, they disperse into the HCW's zone and various locations throughout the room.
Figure 12.

Evaluation of cough aerosol dispersal behavior and AIIR ventilation effectiveness in preventing influenza infection to the HCW.
During the first second after the patient's cough is initiated, as the cough aerosols are in close proximity, coalescence is prominent, and the aerosols count drastically decreases within 5 s. Coalescence also results in heavier aerosols (>1 μ diameter) that tend to settle on surfaces. However, at later time instants (between 5 s and 20 s), as the distance between aerosols increases, the occurrence of coalescence decreases and finally stops. This results in the smaller aerosols (<1 μ diameter) to be carried by the room airflow pattern and remain airborne throughout their presence in the room. Because of this, these aerosols have the potential to travel longer distances until they are eventually pulled into the AIIR exhaust vent.
As time progresses (at time = 10 s), the cough aerosols become slightly diluted in the HCW's region because of the presence of ceiling square vent continuously providing fresh air to the HCW's location. Even though the supply vents plays an important role in diluting these infectious aerosols in the HCW's zone, the AIIR supply ventilation is not sufficient to be effective in preventing the aerosols from entering the HCW's zone.
The cough aerosols remain airborne for an extended time period (approximately 20 s) in the room instead of being immediately removed by the exhaust vent. Therefore, the overall duration of influenza infection in the room (until the aerosol count is reduced to less than 0.16% of the total number of aerosols ejected from the patient's mouth) is recorded as approximately 20 s. This creates an unfavorable environment for the HCW throughout his stay in the room. These airborne cough aerosols that are in the breathing vicinity of the HCW are potentially inhaled by the HCW for prolonged periods.
Because of the incomplete air mixing and the presence of recirculation zones where the air is merely circulated instead of being exhausted, the cough aerosols tend to circulate throughout the room for a prolonged time period of at least 20 s.
The cough aerosols that are in the vicinity of the main exhaust vent are exhausted from the room. This results in the decrease of the aerosol cough over time. However, the exhaust vent can not immediately remove the cough aerosols from the room. Therefore, the AIIR exhaust ventilation is not effective in immediately entraining the aerosols in such a way as to prevent flu virus transmission to the HCW.
Conclusion
This study simulated influenza cough aerosol dispersal in the NIOSH mock AIIR. The study intent was to evaluate the effectiveness of the ceiling ventilation configuration to protect an attending HCW from patient-generated cough aerosol.
In the mock AIIR, the air recirculation regions present in the HCW's vicinity diminished the contaminant removal efficiency and prolonged the presence of the influenza contaminated air within the HCW's zone. Immediately after the patient's cough is initiated, the cough velocity from the patient's mouth propels the aerosols to the HCW standing next to the patient's bed. The AIIR ventilation locations and airflow rates do not have significant influence on the cough aerosol dispersal because of their relatively low velocities compared to the patient's cough. Within 0.7 s, the cough aerosols reach the HCW's breathing zone and some of these aerosols are assumed to be inhaled by the HCW. Thus, the HCW is exposed to the infectious influenza aerosol within a short time interval after the patient's cough.
During the time interval of 1 s and 5 s, the aerosols disperse away from the patient's and HCW's regions, traveling longer distances as they are carried by the AIIR airflow patterns. Subsequently, a part of these aerosols are pulled towards the exhaust vent. However, the remaining cough aerosols enter and recirculate within the HCW's zone. Therefore, the evaluated AIIR ceiling ventilation design is not effective in completely removing the aerosols immediately after the patient's cough.
As the ceiling supply ventilation continuously provides fresh air into the HCW's zone, the aerosols become diluted in the HCW's region over time (10 s). Even though the supply vents play an important role in diluting these infectious aerosols in the HCW's zone, the AIIR supply ventilation is insufficient to prevent the aerosols from entering into the HCW's zone. The overall duration of influenza infection in the room (until the aerosol count is reduced to less than 0.16% of the total number of aerosols ejected from the patient's mouth) is recorded as approximately 20 s. With successive coughing events, a near-continuous exposure would be possible. Thus the ceiling ventilation arrangement of the mock AIIR creates an unfavorable environment for the HCW throughout his stay within the room.
For the tested scenario on the NIOSH mock AIIR, alternative ceiling ventilation arrangements and/or a direct-control ventilation configuration might mitigate the exposure risk of the HCW. Hence, further simulations are planned to evaluate additional ventilation design configurations with the intended goal to better protect the HCW.
Acknowledgments
We express sincere gratitude for NIOSH sponsorship of this research; ASHRAE 2015-2016 Graduate Student Grant-In-Aid award support; the high-performance computing resources of the Ohio Supercomputer Center; colleagues Santosh Roopak Dungi, Santosh Konangi, and Dr. Arvind Kishore; the assistance of NIOSH employee Dylan Neu; and all the members of the Computational Fluid Dynamics Research Laboratory, University of Cincinnati.
Footnotes
Publisher's Disclaimer: Disclaimer: The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the National Institute for Occupational Safety and Health (NIOSH). Mention of a product or company name does not constitute endorsement by the Centers for Disease Control and Prevention or NIOSH.
This paper is based on findings resulting from an ASHRAE 2015-2016 Graduate Student Grant-In-Aid award.
Contributor Information
Deepthi Sharan Thatiparti, Doctoral candidate, Department of Mechanical Engineering, University of Cincinnati, Cincinnati, OH.
Urmila Ghia, Department of Mechanical Engineering, University of Cincinnati, Cincinnati, OH.
Kenneth R. Mead, Mechanical engineer, Center for Disease Control and Prevention, National Institute for Occupational Safety and Health (NIOSH), Cincinnati, OH.
References
- ANSYS. ANSYS ICEM CFD. Canonsburg, PA: ANSYS; 2016a. http://resource.ansys.com/Products/Other+Products/ANSYS+ICEM+CFD. [Google Scholar]
- ANSYS. ANSYS Fluent. Canonsburg, PA: ANSYS; 2016b. www.ansys.com/products/fluids/ansys-fluent. [Google Scholar]
- ASHRAE. Ventilation of health care facilities. Atlanta, GA: ASHRAE; 2013. ANSI/ASHRAE/ASHE Standard 170-2013. [Google Scholar]
- ASHRAE. Ventilation for acceptable indoor air quality. Atlanta: ASHRAE; 2016. ANSI/ASHRAE Standard 62.1-2016. [Google Scholar]
- Bridges CB, Fukuda K, Uyeki TM, Cox NJ, Singleton JA. Prevention and control of influenza: Recommendations of the Advisory Committee on Immunization Practices (ACIP) Morbidity and Mortality Weekly Report Recommendations and Reports. 2002;51(RR-3):1–31. [PubMed] [Google Scholar]
- CDC. Update: Influenza activity—United States, 1998–99 season. Morbidty and Mortality Weekly Report. 1999;48(2):25–27. [PubMed] [Google Scholar]
- CDC. State of the sector—Healthcare and social assistance identification of research opportunities for the next decade of NORA. Atlanta: Centers for Disease Control and Prevention; 2009. http://www.cdc.gov/niosh/docs/2009-139. [Google Scholar]
- CDC. NIOSH progarm portfolio: Healthcare and social assistance. Atlanta: Centers for Disease Control and Prevention; 2013. www.cdc.gov/niosh/programs/hcsa/ [Google Scholar]
- Cunney RJ, Bialachowski A, Thornley D, Smaill FM, Pennie RA. An outbreak of influenza A in a neonatal intensive care unit. Infection Control and Hospital Epidemiology. 2000;21(7):449–54. doi: 10.1086/501786. [DOI] [PubMed] [Google Scholar]
- Drinka PJ, Gravenstein S, Krause P, Nest L, Dissing M, Shult P. Reintroduction of influenza A to a nursing building. Infection Control and Hospital Epidemiology. 2000;21(11):732–35. doi: 10.1086/501715. [DOI] [PubMed] [Google Scholar]
- Evans ME, Hall KL, Berry SE. Influenza control in acute care hospitals. American Journal of Infection Control. 1997;25(4):357–62. doi: 10.1016/s0196-6553(97)90029-8. [DOI] [PubMed] [Google Scholar]
- FGI. 2014 FGI guidelines for design and construction of hospitals and outpatient facilities. Dallas, TX: Facility Guidelines Institute; 2014. [Google Scholar]
- Gupta JK, Lin CH, Chen Q. Flow dynamics and characterization of a cough. Indoor Air. 2009;19(6):517–25. doi: 10.1111/j.1600-0668.2009.00619.x. [DOI] [PubMed] [Google Scholar]
- Hall JE. Guyton and Hall textbook of medical physiology. 12. Amsterdam: Elsevier Health Sciences; 2011. [Google Scholar]
- Heiselberg PK. Room air and contaminant distribution in mixing ventilation. ASHRAE Transactions. 1996;102(2):332–39. [Google Scholar]
- Kowalski WJ, Bahnfleth WP. Airborne respiratory diseases and mechanical systems for control of microbes. HPAC: Heating, Piping, Air Conditioning. 1998;70(7):34–48. [Google Scholar]
- NASA. Human integration design handbook (HIDH) Washington, DC: National Aeronautics and Space Administration; 2014. NASA/SP-2010-3407. http://ston.jsc.nasa.gov/collections/trs/_techrep/SP-2010-3407REV1.pdf. [Google Scholar]
- Ninomura P, Bartley J. New ventilation guidelines for health care facilities. ASHRAE Journal. 2001;43(6):29–33. [Google Scholar]
- Patankar SV, Spalding DB. A calculation procedure for heat, mass and momentum transfer in three-dimensional parabolic flows. International Journal of Heat and Mass Transfer. 1972;15(10):1787–806. [Google Scholar]
- Patriarca PA, Weber JA, Parker RA, Hall WN, Kendal AP, Bregman DJ, Schonberger LB. Efficacy of influenza vaccine in nursing homes: Reduction in illness and complications during an influenza A (H3N2) epidemic. JAMA. 1985;253(8):1136–39. [PubMed] [Google Scholar]
- Sugaya N, Kusumoto N, Suzuki Y, Nerome R, Nerome K. Large sequential outbreaks caused by influenza A (H3N2) and B viruses in an institution for the mentally handicapped. Journal of Medical Virology. 1996;50(2):120–25. doi: 10.1002/(SICI)1096-9071(199610)50:2<120::AID-JMV4>3.0.CO;2-C. [DOI] [PubMed] [Google Scholar]
- Thompson WW, Shay DK, Weintraub E, Brammer L, Cox N, Anderson LJ, Fukuda K. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA. 2003;289(2):179–86. doi: 10.1001/jama.289.2.179. [DOI] [PubMed] [Google Scholar]
- VanSciver M, Miller S, Hertzberg J. Particle image velocimetry of human cough. Aerosol Science and Technology. 2011;45(3):415–22. [Google Scholar]
- Wenzel RP, Deal EC, Hendley JO. Hospital-acquired viral respiratory illness on a pediatric ward. Pediatrics. 1977;60(3):367–71. [PubMed] [Google Scholar]
- WHO. Influenza (seasonal) Geneva, Switzerland: World Health Organization; 2014. Fact sheet No. 211. www.who.int/mediacentre/factsheets/fs211/en/ [Google Scholar]
- Wilde JA, McMillan JA, Serwint J, Butta J, O'Riordan MA, Steinhoff MC. Effectiveness of influenza vaccine in health care professionals: A randomized trial. JAMA. 1999;281(10):908–13. doi: 10.1001/jama.281.10.908. [DOI] [PubMed] [Google Scholar]
- Zadeh MM, Bridges CB, Thompson WW, Arden NH, Fukuda K. Influenza outbreak detection and control measures in nursing homes in the United States. Journal of the American Geriatrics Society. 2000;48(10):1310–15. doi: 10.1111/j.1532-5415.2000.tb02606.x. [DOI] [PubMed] [Google Scholar]


