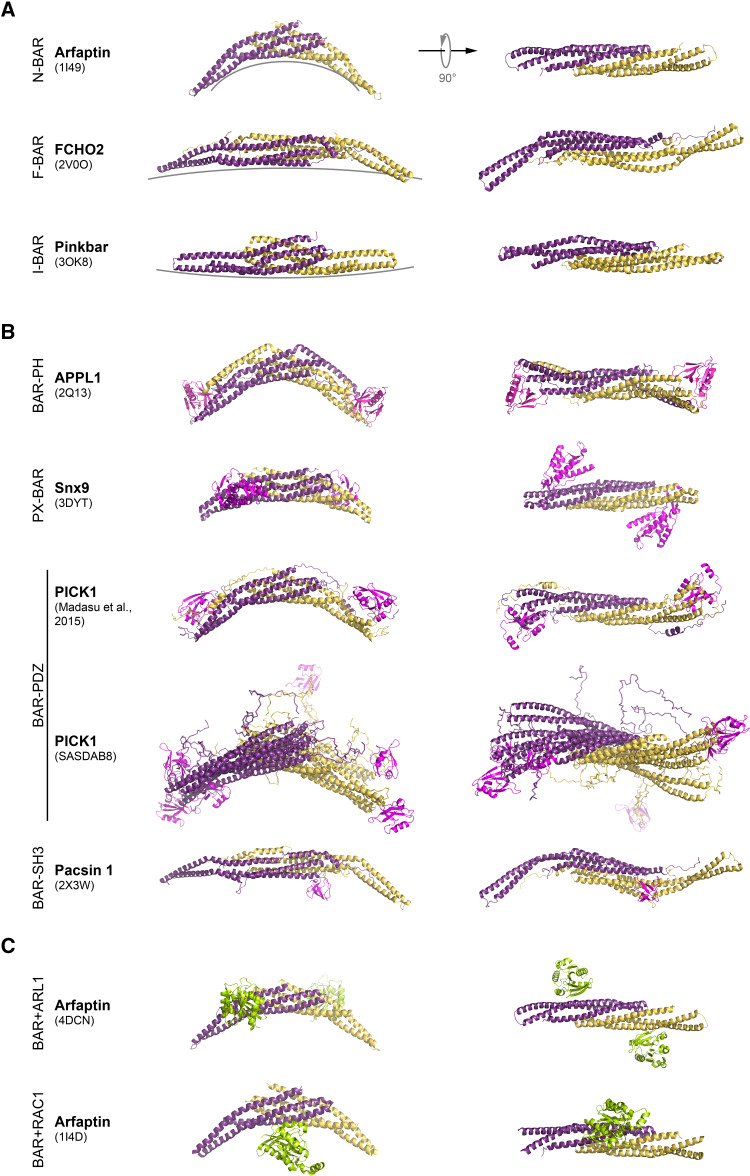
Fig. 1.
Structure of selected BAR domain dimers. The BAR domain dimers form an elongated structure with a core bundle of six α-helices generated by antiparallel dimerisation of two BAR domain monomers. 3D structures of BAR domain dimers are shown as a ribbon. Monomers are depicted in different colors (yellow and dark magenta). Side view of the each BAR dimer is shown on left, while top view is on right. a Examples of BAR domain dimers representing N-BAR, F-BAR, and I-BAR domain fold. Different degrees of curvature adopted by each class of BAR domain dimers are depicted by grey lines. b Structures of BAR domain dimers from different subfamilies with their accessory domains (PH, PX, PDZ, and SH3) shown in magenta. Note, for PICK1, that two SAXS analysis derived models are shown. In PICK1 model (SASDAB8), the PDZ domains are far apart and flexible with respect to the BAR domain. Here, overlay of three generated models is shown. In the PICK1 model of Madasu et al. [78], the position of the PDZ domain was found to be well constrained, and packed against BAR domain. c Structure of the Arfaptin-2 BAR domain dimer in complex with Arl1 GTPase, and Rac1-GDP, both shown in green