Table 1.
Patient Demographic Information and the Number of Pain Reports Supplied by Patients Across the Entire Study
| Demographic characteristics | |
|---|---|
| Institution, N (%) | |
| A | 14 (35.9) |
| B | 17 (43.6) |
| C | 8 (20.5) |
| Gender, N (%) | |
| Male | 16 (41.0) |
| Female | 23 (59.0) |
| Age at baseline (years), N (%) | |
| 18–34 | 24 (61.5) |
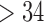
|
15 (38.5) |
| SCD type, N (%) | |
| Hemoglobin SC | 8 (20.5) |
| Hemoglobin SS | 22 (56.4) |
Hemoglobin SB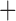 (Beta) Thalassemia (Beta) Thalassemia |
5 (12.8) |
| Beta-Zero Thalassemia | 3 (7.7) |
| SOArab | 1 (2.6) |
| Hydroxyurea user, N (%) | 27 (69.2) |
| Folic acid vitamin user, N (%) | 26 (66.7) |
| Long-acting opioid user, N (%) | 29 (74.4) |
| Short-acting opioid user, N (%) | 35 (89.7) |
| Nonopioid user, N (%) | 29 (74.4) |
| Mean | SD | (Min, Max) | |
|---|---|---|---|
| Number of pain reports | 67.2 | 60.4 | (9.0, 257.0) |
| Days of pain reports | 164.6 | 109.6 | (10.3, 435.1) |
| Within-patient average VAS score | 4.7 | 2.1 | (0.3, 9.4) |
| Mean | SD | (Min, Max) | |
|---|---|---|---|
| Number of pain reports (first 2 weeks) | 13.2 | 9.6 | (2.0, 45.0) |
| Number of long-acting opioid doses (first 2 weeks) | 6.0 | 8.4 | (0.0, 35.0) |
| Number of short-acting opioid doses (first 2 weeks) | 7.2 | 7.5 | (0.0, 35.0) |
| Number of nonopioid doses (first 2 weeks) | 2.1 | 3.1 | (0.0, 12.0) |
SCD, sickle cell disease; VAS, visual analog scale.
