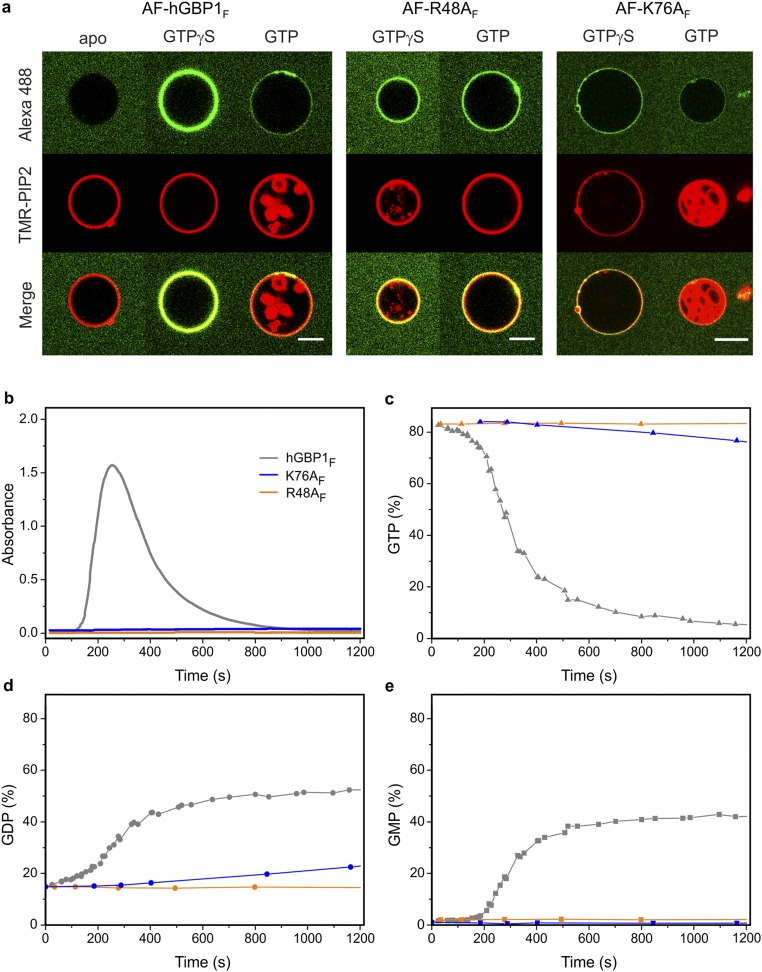
Fig. S2.
Membrane binding and polymerization properties of hGBP1 mutants. Farnesylated GTPase-deficient mutant R48AF (orange) and mutant K76AF (blue), which has a slower GTPase activity and a deficient second hydrolysis step leading to a lack of GMP formation, were investigated for their capability for GUV binding and polymerization. (A) Comparison of GTP- (10 min) and GTPγS-dependent GUV binding of Alexa-fluorescent R48AF (AF-R48AF) and K76AF (AF-K76AF) to AF-hGBP1F. Apo state of AF-hGBP1F is illustrated as representative for all mutants, which likewise failed to associate with membranes in the absence of nucleotide. (Scale bar, 5 µm.) (B–E) A total of 10 µM of hGBP1F wild type (gray), R48AF (orange), and K76AF (blue) were investigated for their capability to polymerize. Polymerization was triggered by addition of 1 mM GTP (t = 0 s) in the absence of liposomes. Time courses of absorbance (B) and the fractions of nucleotides GTP (C), GDP (D), and GMP (E) analyzed by HPLC at different time points along the turbidity assay.