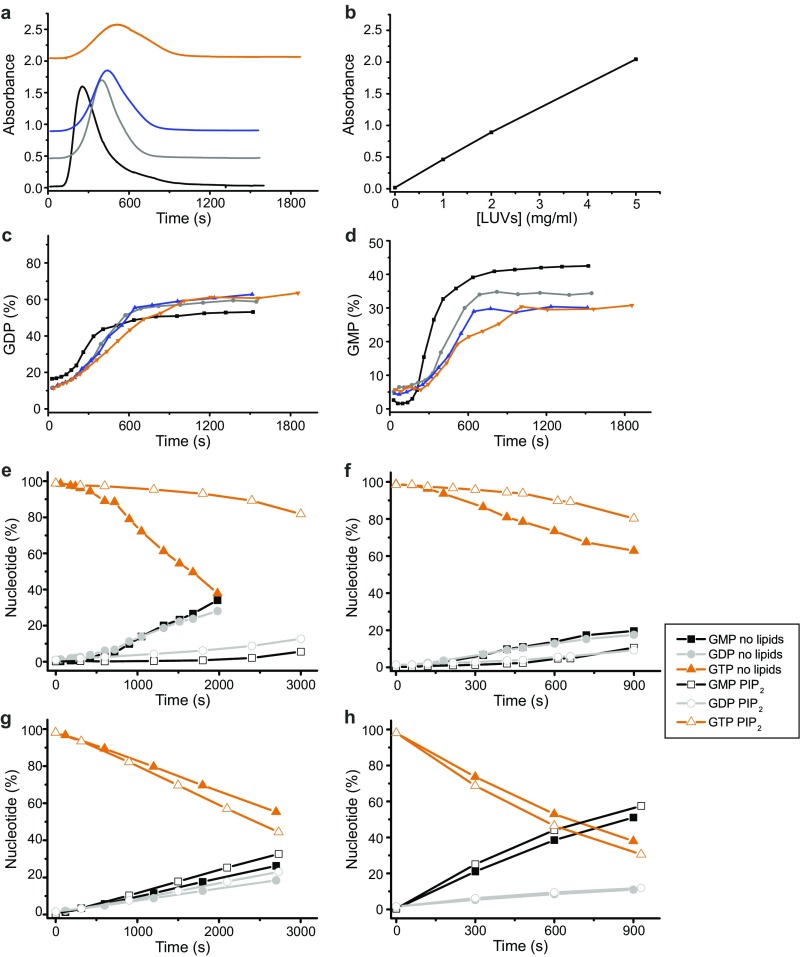
Fig. S6.
Polymerization and GTPase activity of hGBP1 under various lipid and temperature conditions. (A and B) Time courses of hGBP1F polymer absorbance (A) and the obtained background absorbance of liposomes as a function of lipid concentration (B). Experiments were performed with 10 µM hGBP1F in the absence of liposomes (black) and in the presence of 1 mg/mL (gray), 2 mg/mL (blue), and 5 mg/mL (orange) of final lipid concentration. Polymerization was triggered upon addition of 1 mM GTP (t = 0 s). (C and D) Analysis of hGBP1F catalyzed GTP hydrolysis products GDP (C) and GMP (D) for same experiments. (E–H) Nucleotide composition of the GTPase reaction performed either with 2 μM hGBP1F at 25 °C (E) and 37 °C (F) or with 2 μM hGBP1NF at 25 °C (G) and 37 °C (H) in the absence of liposomes (filled symbols) or in the presence of 0.67 mg/mL PIP2 liposomes (open symbols).