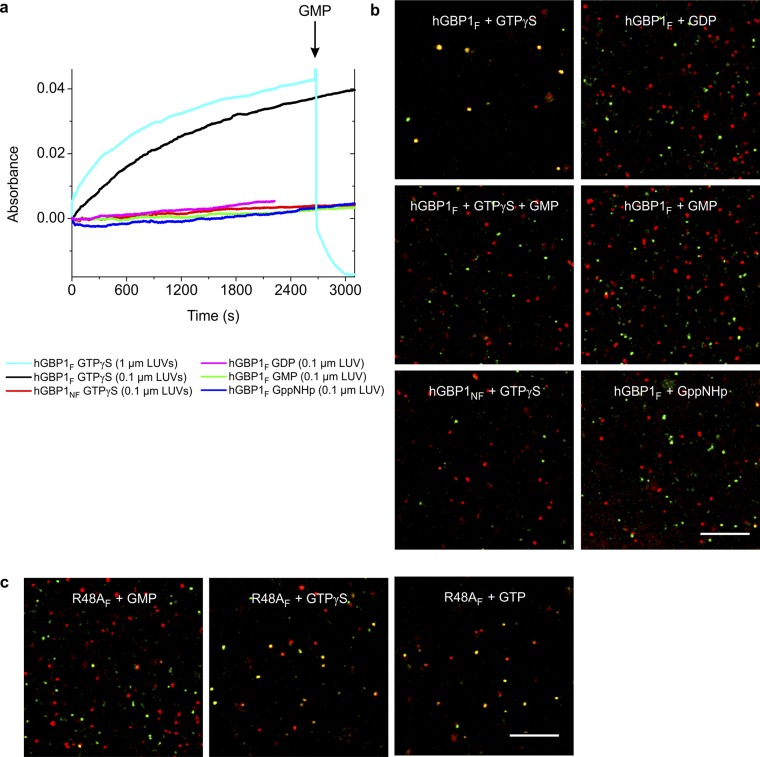
Fig. S8.
Nucleotide-dependent membrane tethering mediated by hGBP1F. (A) HGBP1-mediated tethering followed by absorbance-based tethering assay. The increase in particle size was monitored by the absorbance at 350 nm. The initial absorbance value of 0.2 was subtracted. A total of 400 µM of the corresponding nucleotide was added at t = 0 s in the presence of 30 µM of the corresponding protein. Addition of GTPγS was done for LUVs of various sizes, 0.1 µm (black) compared with 1 µm (cyan). A total of 10 mM GMP was added after 45 min (arrow) to dissociate tethered LUVs. Reactions were done at 30 °C. (B) HGBP1-mediated tethering followed by visual tethering assay. LUVs, which also contained either ATTO-488 or Rhodamine-labeled lipids, respectively, were mixed at a 1:1 ratio (total lipid concentration, 0.8 mg/mL) in the presence of 30 µM of either hGBP1F or hGBP1NF. All samples were incubated with 400 µM of the corresponding nucleotide for 45 min at 30 °C. To dissociate tethered LUVs in the presence of GTPγS, one of the reaction mixtures with GTPγS was additionally supplied with GMP after 45 min (final concentration of 36 mM) and incubated for 10 more minutes at 30 °C. All samples were diluted, loaded into the observation chamber, and visualized by confocal microscopy. (Scale bar, 10 μm.) (C) As in B but with the GTPase-deficient farnesylated mutant R48AF.