Figure 7. Schema summarizing the complementary effects of BTK and BCL-2 inhibition on CLL cell mitochondria.
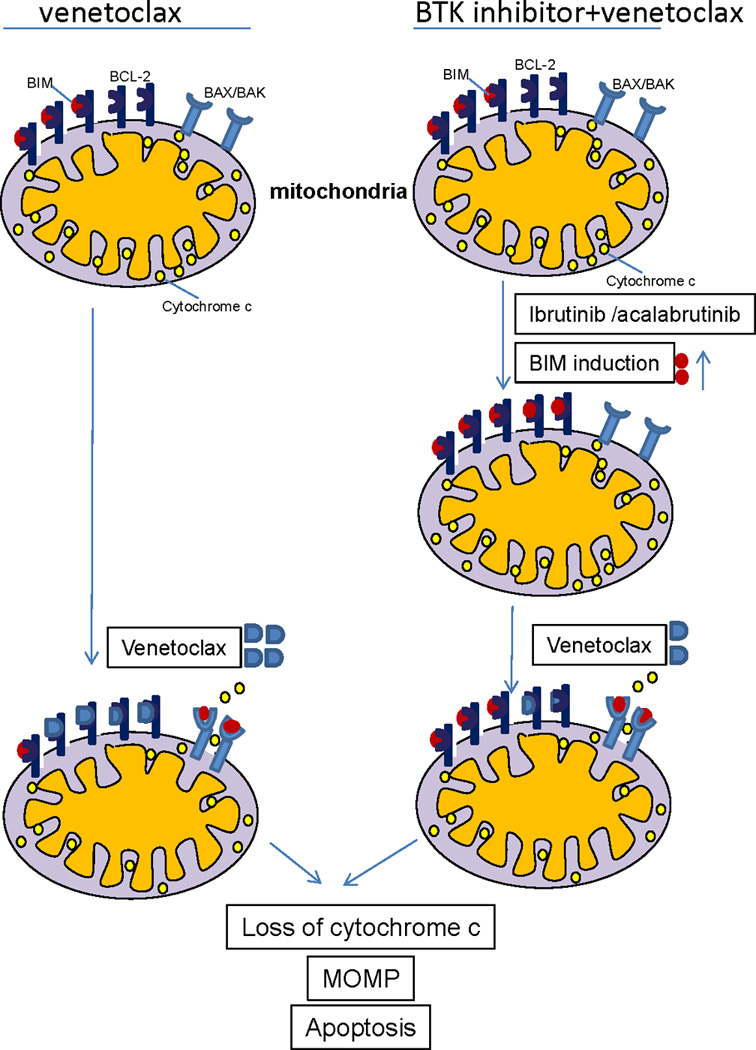
At baseline (top left), CLL cells are BCL-2 dependent and primed for apoptosis, with the pro-apoptotic protein BIM occupying some, but not all of the binding sites on BCL-2. Treatment with venetoclax (bottom left) leads to apoptosis in most CLL cells by displacing BIM from BCL-2, allowing it to bind to the pro-apoptotic effector proteins BAX/BAK, which can then homo-oligomerize leading to mitochondrial outer membrane permeabilization (MOMP), cytochrome C release, and subsequent caspase-mediated cell death. BTK inhibition with ibrutinib or acalabrutinib (right side) can increase BIM protein thereby saturating essentially all of the binding sites on BCL-2 and increasing the BCL-2 dependence of the CLL cell even further. Subsequently adding venetoclax (lower right) rapidly triggers MOMP and subsequent CLL cell death.
