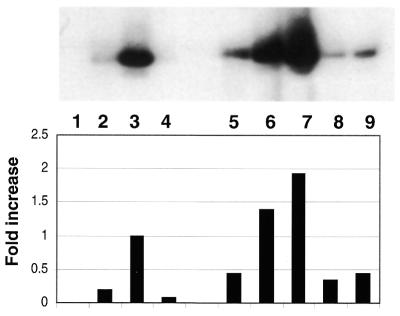
Figure 2.
In vitro phosphorylation of GST–APE/Ref-1 by PKC increases redox activity as shown by EMSA. An extract of wild-type K562 cells (10 µg) was used in combination with 1.5 µg of purified GST–APE/Ref-1 or PKC-phosphorylated GST–APE/Ref-1. ATP (Promega) was 500 µM, and where indicated 125 µM of phosphotidyl serine was used. Lane 1, oligo alone; lane 2, K562 cell lysate, which was also present in the reactions shown in lanes 3–9; lane 3, purified GST–APE/Ref-1; lane 4, purified GST–APE/Ref-1 and ATP; lane 5, phosphorylated GST–APE/Ref-1 and ATP; lane 6, dialyzed, phosphorylated GST–APE/Ref-1; lane 7, dialyzed GST–APE/Ref-1 phosporylated in the presence of phosphotidyl serine; lane 8, reaction in lane 6, plus phosphatase; lane 9, reaction in lane 7, plus phosphatase