Introduction
Kimura disease (KD) is a rare idiopathic condition that presents as solitary or multiple painless subcutaneous masses in the head and neck region. It may also involve extracutaneous sites, such as regional lymph nodes, major salivary glands, and the kidneys.1 Most cases reported occurred in Asian men between 20 and 30 years of age.2 Treatment is often sought for symptomatic relief and cosmetic concerns. Therapeutic modalities for KD include surgical excision, radiotherapy, and various immunomodulating agents, such as oral corticosteroids, cyclosporine, and leflunomide.1, 3, 4, 5, 6, 7 Most cases have favorable initial responses to treatment, but relapse occurs at rates as high as 60% to 100%.4, 5, 6 We report a case of KD with sustained response to mycophenolate mofetil (MMF) monotherapy.
Case report
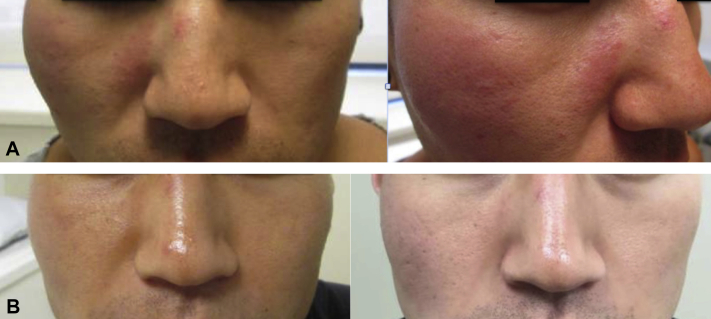
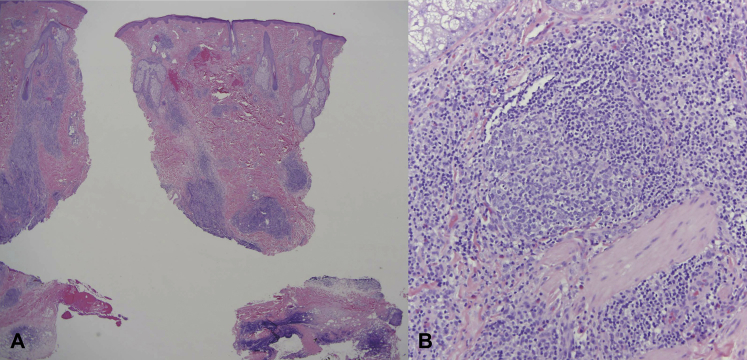
A 32-year-old Chinese man presented for a 1-year history of a pruritic, enlarging nodule on the right cheek. Physical examination found an ill-defined, erythematous, nontender nodule measuring 6 × 5 cm on the right malar cheek and a smaller 1.5- × 3-cm plaque on the right medial cheek (Fig 1, A). Computed tomography scan and magnetic resonance imaging of the head and neck found extension to the subcutaneous fat without skeletal muscle, bony, or lymph node involvement. Histopathologic examination found nodular lymphocytic aggregates characterized by centrally located germinal centers (positive for CD10 and Bcl-6) surrounded by small lymphocytes in the reticular dermis and subcutaneous fat. There were numerous blood vessels and many admixed eosinophils within the zones of nodular lymphocytic infiltrate (Fig 2). No significant fibrosis was noted. No lymphoid atypia was identified, and a polyclonal pattern was confirmed by gene rearrangement analysis. Tissue culture was negative. A diagnosis of Kimura's disease was rendered. Laboratory analyses found an elevated IgE at 315 IU/mL, normal IgG4 19 mg/dL, and a mildly elevated peripheral eosinophil percentage (5.9%). The absolute eosinophil count was within normal range. Mild thrombocytopenia (platelet count, 115,000/μL) was noted on initial workup, but platelet count normalized to 145,000/μL, and no further workup was pursued. The patient's renal function was within normal limits, and there was no significant proteinuria (108.9 mg/24 hours) on timed urine studies.
Fig 1.
Kimura disease. A, Ill-defined, erythematous deep nodule and plaque on the right malar and medial cheek. B, Significant reduction in size and erythema at 1 month of mycophenolate mofetil, 1000 mg twice daily (left). Continued clinical response at 23 months with mycophenolate mofetil at 1000 mg daily (right).
Fig 2.
Histopathologic features of KD. A, Low-power examination shows a striking nodular lymphocytic infiltrate involving the reticular dermis and subcutaneous fat. No significant fibrosis was noted. B, Higher-power magnification shows the basic composition of this infiltrative process being one of reactive germinal centers surrounded by small mature lymphocytes and eosinophils accompanied by numerous small-caliber vessels defining a form of angiomatous pseudolymphoma. (A and B, Hematoxylin-eosin stain; original magnifications: A, ×20; B, ×400.)
Our patient began taking MMF, 1000 mg twice daily, and topical timolol solution. He discontinued topical timolol after 2 weeks. Clinical response was seen within 1 month of treatment (Fig 1, B). His treatment course was complicated by mild neutropenia at 4 months (absolute neutrophil count, 1,600/μL), which resolved shortly after dosage adjustment of MMF to 1000 mg daily. The patient continued to have sustained response with MMF, 1000 mg daily at 23 months of follow-up (Fig 1, B). His peripheral eosinophilia was noted to persist (9.6%; absolute number, 500/μL) at 23 months.
Discussion
KD is a chronic inflammatory disease that manifests as a triad of subcutaneous nodules in the head and neck region, peripheral blood eosinophilia, and elevated serum IgE. Renal involvement is not uncommon and most frequently results in nephrotic syndrome.1 The pathogenesis remains unknown, but allergy, atopy, autoimmunity, and parasite infestation are considered possible risk factors.4, 5, 6 Previous studies have found increased levels of interleukin-4, interleukin-5, and interleukin-13 in the peripheral blood of affected individuals, suggesting a role for T-helper 2 cytokines.8
KD is often compared with angiolymphoid hyperplasia with eosinophilia (ALHE) because of several overlapping clinical and histologic features. The question of whether KD and ALHE represent 2 ends of 1 disease spectrum or 2 separate entities remains controversial. The current understanding is that they are distinct processes. KD represents a lymphoproliferative condition, whereas ALHE is a neoplastic disorder of vascular endothelial cells with a secondary inflammatory component.2 KD most frequently affects young Asian men, whereas ALHE tends to develop in middle-age women. Lesions can be differentiated clinically, as KD lesions are deep nodules with ill-defined margins that may have overlying skin changes. In contrast, ALHE lesions are typically well-circumscribed papules or nodules that are red, brown, or purple in color.2 Lymphadenopathy, peripheral blood eosinophilia, and an elevated serum IgE occur more consistently in KD. Additionally, there are several histopathologic features that distinguish KD from ALHE, namely, the presence of numerous lymphoid follicles and the absence of irregular, dilated blood vessels in the former.2
Clinicopathologic resemblance of KD and IgG4-related skin disease has been suggested by Hattori et al.9 However, our patient lacked characteristic histopathologic features of IgG4-related disease, such as storiform fibrosis or obliterative phlebitis.10 His serum IgG4 level was normal. Taken together, his overall clinical presentation, including histopathology, presence of peripheral eosinophilia, and elevated serum IgE, was the most consistent with KD.
Many factors were considered when deciding on the treatment plan in our case: the poorly circumscribed margins, the presence of a vascular component, and the desire for optimal cosmesis. Treatment modalities reported in the literature are heterogeneous, but surgical excision and oral corticosteroids represent the most frequently used strategies.3, 5 Recurrence is common, especially when tapering steroids.4, 6 Leflunomide in combination with oral corticosteroids has shown promise in treating cases of KD refractory to steroids alone.6, 7, 11 Leflunomide works by inhibiting pyrimidine synthesis in activated lymphocytes, decreasing the proliferation of activated T- and B-lymphocytes and downregulating immunoglobulin production. MMF exhibits immunomodulatory effects in a similar fashion by inhibiting de novo purine synthesis via inosine monophosphate dehydrogenase. Additionally, it has anti-inflammatory effects via prevention of lymphocyte and monocyte adhesion to endothelial cells.
Based on this assessment, we chose a 2-pronged approach with MMF for its immunomodulatory and anti-inflammatory properties and topical timolol for its antiangiogenic function. The patient was compliant with MMF but stopped using topical timolol after 2 weeks of inconsistent use. We therefore attribute results to MMF alone. There are 5 previous reports of MMF1, 5, 12, 13, 14 used in the treatment of KD, 2 in which MMF was used as monotherapy with conflicting results (Table I).5, 12 One of the major advantages of MMF is its superior safety profile when compared with other systemic immunosuppressive agents. In this case, we observed a positive response to the treatment regimen—disease control within 1 month of treatment with MMF and sustained response at 23 months with 1000 mg daily. No adverse events were noted with the exception of transient mild neutropenia. We feel that MMF may be a promising therapy for KD.
Table I.
Reported cases of Kimura Disease treated with mycophenolate mofetil
| Study, year | Clinical features | Prior treatment | MMF dose | Concurrent treatment | Duration of treatment | Response to treatment |
|---|---|---|---|---|---|---|
| Wang et al, 201612 | 68-year-old woman with pulmonary KD and concurrent pulmonary tuberculosis | Oral steroids, cyclosporine, isoniazid, and rifampin | Unknown | Isoniazid, rifampin, pyrazinamide, and streptomycin for tuberculosis | Unknown | Alleviated cough and albuminuria; improved chest CT |
| Chen et al, 20161 | 29 patients with KD and secondary renal involvement (93% male; average age, 35.5 y) | None | 250 mg twice daily | Tripterygium wilfordii, prednisone, leflunomide, tacrolimus, and renin-angiotensin system blockers for patients with hypertension | Unknown | Complete remission in 22 patients (10 with subsequent relapse); partial remission in 4 patients |
| Kottler et al, 20155 | 1 patient with KD∗ | Unknown∗ | Unknown∗ | Unknown∗ | Unknown∗ | Relapse |
| Fouda et al, 201113 | 50-year-old Egyptian man with KD and secondary membranous nephropathy | None | 2000 mg daily | IV cyclophosphamide, high-dose prednisone, Chlorpheniramine, and ranitidine | Unknown | Complete disappearance of skin lesions and normalization of kidney function |
| Noel et al, 200914 | 29-year-old white man with KD, secondary FSGS, and immune thrombocytopenia | Prednisone and ramipril | 1000 mg twice daily | Prednisone and ramipril | Unknown | Remission of nephrotic syndrome within 1 month of treatment followed by relapse after 6 months of treatment |
CT, Computed topography; FSGS, focal segmental glomerulosclerosis; IV, intravenous; KD, Kimura disease.
Retrospective study, individual case details not discussed.
Footnotes
Funding sources: None.
Conflicts of interest: None declared.
References
- 1.Chen Y., Wang J., Xu F., Zeng C., Liu Z. Clinicopathological features and prognosis of Kimura's disease with renal involvement in Chinese patients. Clin Nephrol. 2016;85(6):332–339. doi: 10.5414/CN108749. [DOI] [PubMed] [Google Scholar]
- 2.Buder K., Ruppert S., Trautmann A., Brocker E.B., Goebeler M., Kerstan A. Angiolymphoid hyperplasia with eosinophilia and Kimura's disease - a clinical and histopathological comparison. J Dtsch Dermatol Ges. 2014;12(3):224–228. doi: 10.1111/ddg.12257_suppl. [DOI] [PubMed] [Google Scholar]
- 3.Luo G., Gu F., Liu T., Huang Y. Kimura's disease of the right cheek: a case report. Exp Ther Med. 2016;11(1):218–220. doi: 10.3892/etm.2015.2901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chen Q.L., Dwa S., Gong Z.C. Kimura's disease: risk factors of recurrence and prognosis. Int J Clin Exp Med. 2015;8(11):21414–21420. [PMC free article] [PubMed] [Google Scholar]
- 5.Kottler D., Barete S., Quereux G. Retrospective multicentric study of 25 Kimura disease patients: emphasis on therapeutics and shared features with cutaneous IgG4-related disease. Dermatology. 2015;231(4):367–377. doi: 10.1159/000439346. [DOI] [PubMed] [Google Scholar]
- 6.Dai L., Wei X.N., Zheng D.H., Mo Y.Q., Pessler F., Zhang B.Y. Effective treatment of Kimura's disease with leflunomide in combination with glucocorticoids. Clin Rheumatol. 2011;30(6):859–865. doi: 10.1007/s10067-011-1689-2. [DOI] [PubMed] [Google Scholar]
- 7.Ma X.R., Xin S.J., Ouyang T.X., Ma Y.T., Chen W.Y., Chang M.L. Successful treatment of Kimura's disease with leflunomide and methylprednisolone: a case report. Int J Clin Exp Med. 2014;7(8):2219–2222. [PMC free article] [PubMed] [Google Scholar]
- 8.Katagiri K., Itami S., Hatano Y., Yamaguchi T., Takayasu S. In vivo expression of IL-4, IL-5, IL-13 and IFN-gamma mRNAs in peripheral blood mononuclear cells and effect of cyclosporin A in a patient with Kimura's disease. The Br J Dermatol. 1997;137(6):972–977. [PubMed] [Google Scholar]
- 9.Hattori T., Miyanaga T., Tago O. Isolated cutaneous manifestation of IgG4-related disease. J Clin Pathol. 2012;65(9):815–818. doi: 10.1136/jclinpath-2012-200749. [DOI] [PubMed] [Google Scholar]
- 10.Deshpande V., Zen Y., Chan J.K. Consensus statement on the pathology of IgG4-related disease. Mod Pathol. 2012;25(9):1181–1192. doi: 10.1038/modpathol.2012.72. [DOI] [PubMed] [Google Scholar]
- 11.Liu C., Hu W., Chen H. Clinical and pathological study of Kimura's disease with renal involvement. J Nephrol. 2008;21(4):517–525. [PubMed] [Google Scholar]
- 12.Wang Z., Wan J., Gong J., Zhang Y. Kimura's disease in respiratory department: two case reports. Chest. 2016;149(4):A470. [Google Scholar]
- 13.Fouda M.A., Gheith O., Refaie A. Kimura disease: a case report and review of the literature with a new management protocol. Int J Nephrol. 2011;2010:673908. doi: 10.4061/2010/673908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Noel N., Meignin V., Rosenstingl S., Ronco P., Boffa J.J. Nephrotic syndrome associated with immune thrombocytopenia revealing Kimura's disease in a non-Asian male. NDT Plus. 2009;2(6):452–454. doi: 10.1093/ndtplus/sfp099. [DOI] [PMC free article] [PubMed] [Google Scholar]