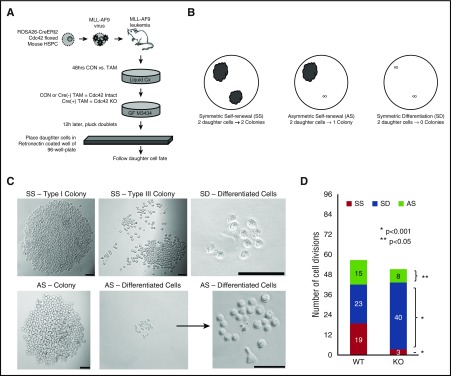
Figure 6.
Loss of Cdc42 increased the percentage of cell divisions, resulting in symmetric differentiation. (A) Murine MLL-AF9 leukemia cells were treated with 4-OHT to induce Cre-mediated deletion of Cdc42 (KO), with ethanol-treated cells serving as the vehicle control (WT). After 48 hours, the cells were plated in methylcellulose media (MethoCult GF M3434, StemCell Technologies). Sixteen hours later, doublets were identified, indicating cells at completion of the first cell division. Doublets were isolated and then transferred by pipette to a single well of RetroNectin-coated 96-well plates in IMDM + 10% FBS + 20 ng/mL recombinant rat SCF and 10 ng/mL murine granulocyte-macrophage colony-stimulating factor, murine IL-3, and human IL-6. Daughter cells were separated by gently pipetting up and down, then visualized 4 hours later to confirm 2 distinct single cells adherent to the RetroNectin. Wells were visualized over the next 48 hours to confirm subsequent divisions by each daughter cell, indicating viability. Wells in which 2 distinct, viable daughter cells could not be visualized were excluded from analysis. (B,C) Wells were then visualized after 7 days and scored as having 2 colonies (SS), 1 colony (AS), or 0 colonies (SD). Scale bar, 50 μm. (D) Data are shown as the number of each type of cell division scored per 96 wells plated with WT vs KO MA9 leukemia daughter cell pairs, and the statistical significance of differences between the 2. Data are representative of 6 independent experiments.