Figure 1.
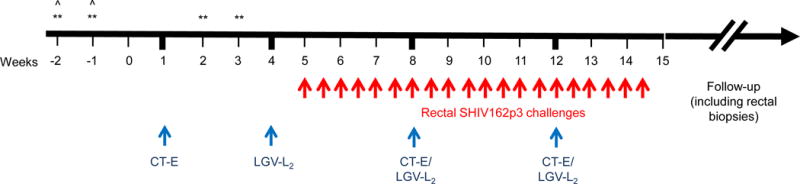
Study design. Evaluation of risk of HIV acquisition in CT co-infected animals. The design (in weeks) shows baseline collections followed by inoculation (blue arrows) with CT serovar E (CT-E) at week 1, and serovar LGV-L2 at week 4. The animals were boosted with CT (serovar E and LGV-L2) every 4 weeks to maintain infection. Starting week 5, we rectally challenged (red arrows) the animals with SHIV162p3 (10TCID50) twice a week up to 20 challenges; **=cytokines measured at these points to compare local inflammation before and after STI inoculations; ˆ=baseline rectal biopsy collections.
