Abstract
Blockade of immune checkpoint proteins (e.g., CTLA-4, PD-1) improves overall survival in advanced melanoma; however, therapeutic benefit is limited to only a subset of patients. Because checkpoint blockade acts by “removing the brakes” on effector T cells, the efficacy of checkpoint blockade may be constrained by the limited pool of melanoma-reactive T cells in the periphery. In the thymus, autoimmune regulator (Aire) promotes deletion of T cells reactive against self-antigens that are also expressed by tumors. Thus, while protecting against autoimmunity, Aire also limits the generation of melanoma-reactive T cells. Here, we show that Aire deficiency in mice expands the pool of CD4+ T cells capable of melanoma cell eradication and has additive effects with anti–CTLA-4 antibody in slowing melanoma tumor growth and increasing survival. Moreover, pharmacologic blockade of central T cell tolerance and peripheral checkpoint blockade in combination enhanced antimelanoma immunity in a synergistic manner. In melanoma patients treated with anti–CTLA-4 antibody, clinical response to therapy was associated with a human Aire polymorphism. Together, these findings suggest that Aire-mediated central tolerance constrains the efficacy of peripheral checkpoint inhibition and point to simultaneous blockade of Aire and checkpoint inhibitors as a novel strategy to enhance antimelanoma immunity.
Keywords: Immunology
Keywords: Cancer immunotherapy, Cellular immune response, Melanoma
Central immune checkpoint blockade in combination with peripheral immune checkpoint disruption augments the anti-tumor immunity and improves the survival of melanoma bearing mice.
Introduction
Augmenting endogenous antimelanoma T cell responses through blockade of immune checkpoints has proven effective as a therapeutic strategy against metastatic melanoma (1). Ipilimumab, a mAb targeting the coinhibitory immune checkpoint protein CTLA-4 on T cells, was the first systemic treatment to show prolonged overall survival in patients with metastatic cutaneous melanoma (2). However, anti–CTLA-4 (aCTLA-4) antibody provides disease control in only 22% of patients (2, 3), with long-term benefit in < 10% of patients (2, 4). Thus, for most metastatic melanoma patients, the antimelanoma T cell response after CTLA-4 blockade continues to be inadequate. Since the approval of ipilimumab by the FDA in 2011, two other immune checkpoint inhibitors, which target the coinhibitory immune checkpoint protein PD-1, have been approved on the basis of randomized clinical studies (5–7). Despite improved efficacy with treatments targeting PD-1, many patients still have only transient responses or do not respond to these therapies. What constrains the antimelanoma effects of checkpoint inhibitors is currently unclear (8).
Central T cell tolerance mechanisms protect against the development of autoimmunity, but also limit antitumor immunity (9–11). A key mediator of central tolerance is the Autoimmune Regulator (Aire) gene, a transcriptional activator expressed predominantly in medullary thymic epithelial cells (mTECs). There, Aire promotes expression of tissue-restricted self-antigens (TSAs) so that self-reactive thymocytes that recognize these TSAs with high affinity undergo negative selection. A subset of Aire-regulated TSAs is expressed by both melanocytes and melanoma cells. As a consequence, while purging self-reactive T cells that recognize melanocyte antigens, Aire also removes T cells capable of recognizing and eradicating melanoma cells. In humans, protection from melanoma has been associated with distinct AIRE single nucleotide polymorphisms (SNPs), which can decrease stability of Aire mRNA (12). This protection is associated with increased frequency of T cell clones recognizing MAGE-1, a self/melanoma antigen expressed in the thymus. Together, these findings support a model in which Aire deficiency prevents deletion of T cell clones that recognize self/melanoma antigens to promote a more robust T cell–mediated antitumor response.
While Aire limits antimelanoma immunity through its function in the thymus, CTLA-4 and other checkpoint proteins limit T cell responses through their activity in the immunologic periphery (13). Upon T cell receptor (TCR) activation, T cells upregulate checkpoint proteins that attenuate the T cell response (14). CTLA-4, for example, dampens early T cell activation by inducing inhibitory downstream TCR signaling and competitive inhibition of CD28-mediated coactivation. The distinct mechanisms of actions of Aire and checkpoint proteins led us to hypothesize that blockade of central Aire-mediated tolerance may interact with blockade of peripheral checkpoint inhibition to enhance T cell–mediated antimelanoma immunity.
We report here that Aire deficiency and aCTLA-4 antibody in combination have an additive effect in diminishing melanoma outgrowth and prolonging survival in melanoma-bearing mice. A pool of melanoma-reactive, cytolytic CD4+ T cells that escape thymic deletion in the setting of Aire deficiency are further activated by checkpoint inhibition in the periphery, leading to an enhanced antitumor effect. Additionally, combination therapy using pharmacologic depletion of Aire-expressing mTECs (15), and inhibition of CTLA-4, significantly prolonged survival in melanoma-bearing mice compared with either strategy alone. Finally, an Aire SNP (rs1055311) is associated with response to ipilimumab therapy in metastatic melanoma patients, as part of the E1608 clinical trial, a randomized phase 2 study of ipilimumab versus ipilimumab plus GM-CSF (16). These findings point to Aire-mediated central tolerance as a key mechanism limiting the efficacy of checkpoint inhibitors and provide preclinical evidence for combining central and peripheral tolerance blockade to expand the antitumor immune response.
Results
Aire deficiency enhances antimelanoma effects of CTLA-4 blockade in mice.
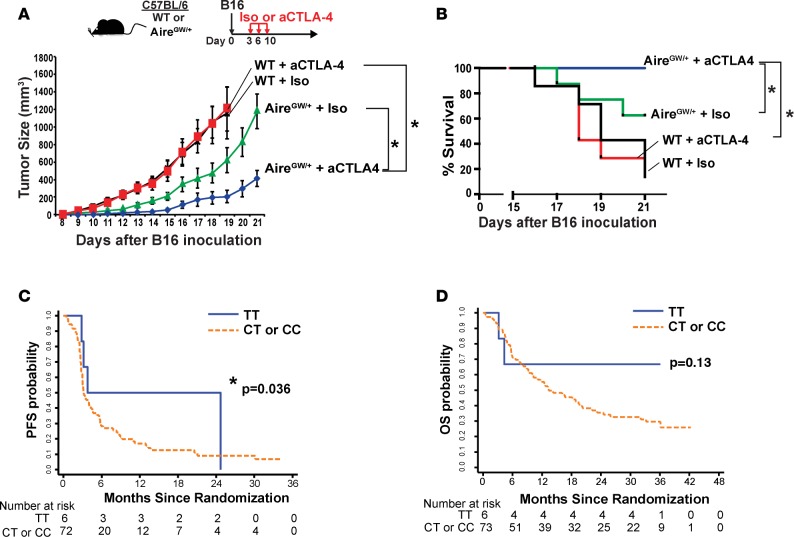
A dominant Aire G228W mutation results in partial loss of Aire function (17), and mice with one copy of this mutation (AireGW/+ mice) have increased antimelanoma immunity (11). We used AireGW/+ mice to test the hypothesis that Aire deficiency and peripheral checkpoint inhibition have additive effects in increasing antimelanoma immunity. CTLA-4 is a coinhibitory immune checkpoint protein induced by T cell activation. In Aire-sufficient (WT) mice challenged with B16 melanoma, aCTLA-4 antibody administration did not alter melanoma growth or host survival (Figure 1, A and B), a finding consistent with previous reports (18, 19). In AireGW/+ mice, on the other hand, aCTLA-4 antibody decreased melanoma growth and improved host survival compared with isotype (iso) control (Figure 1, A and B). These results suggest that Aire deficiency and aCTLA-4 antibody administration in combination have additive effects on decreasing B16 melanoma growth and improving host survival.
Figure 1. Association between Aire and antimelanoma effects of CTLA-4 blockade in mice and humans.
(A and B) WT and AireGW/+ mice were s.c. injected with 1 × 105 B16 melanoma cells followed by anti–CTLA-4 (aCTLA-4) antibody or isotype control (iso) antibody treatment. Mice in each group were followed for B16 melanoma tumor growth and survival. n = 7–12 per group, cumulative of at least 2 independent experiments. Mann-Whitney U test. *P < 0.05. (C and D) Comparison of progression-free survival (PFS) and overall survival (OS) probability between patients with Aire SNP rs1055311 (TT versus CT/CC).
Human Aire polymorphism is associated with response to aCTLA-4.
We investigated whether the interaction between Aire deficiency and aCTLA-4 antibody in melanoma-bearing mice might also have relevance in melanoma patients. Monoclonal aCTLA-4 IgG1 (ipilimumab) was the first checkpoint inhibitor to obtain FDA approval for patients with advanced melanoma. However, only 10%–20% of patients respond to treatment (2, 20, 21), and the factors that determine response are unclear. Multiple Aire polymorphisms, including one that may negatively affect mRNA Aire stability (12), have been associated with protection from melanoma development. This suggested that Aire polymorphisms that disrupt Aire function may enhance antimelanoma immunity in humans. Based on our findings in mice, we sought to test whether Aire polymorphisms may be associated with response to ipilimumab.
We focused on 5 Aire polymorphisms (rs1800522, rs2075876, rs56393821, rs1800520, rs1055311) that have previously been associated with melanoma protection. Seventy-nine patients with metastatic melanoma participating in the E1608 study (16) were genotyped for these 5 Aire polymorphisms. All 79 patients included in this study were randomized to the ipilimumab-alone arm. The rs1055311 TT polymorphism, which has previously been associated with protection from melanoma development (12), was present in 6 of 79 patients (7.6%). The presence of the rs1055311 TT polymorphism was associated with increased probability of progression-free survival (Fisher’s exact test, P = 0.036; Figure 1C) and trended toward increased probability of overall survival, although this did not reach statistical significance (log rank P = 0.13; Figure 1D). A caveat of these findings is the small number of patients harboring the rs1055311 TT polymorphism, which may reflect the melanoma-protective property of this SNP. These data suggest that this Aire SNP may be associated with response to ipilimumab in metastatic melanoma and should be validated in a larger-scale study.
CD4+ T cells from Aire-deficient mice have increased cytolytic capacity.
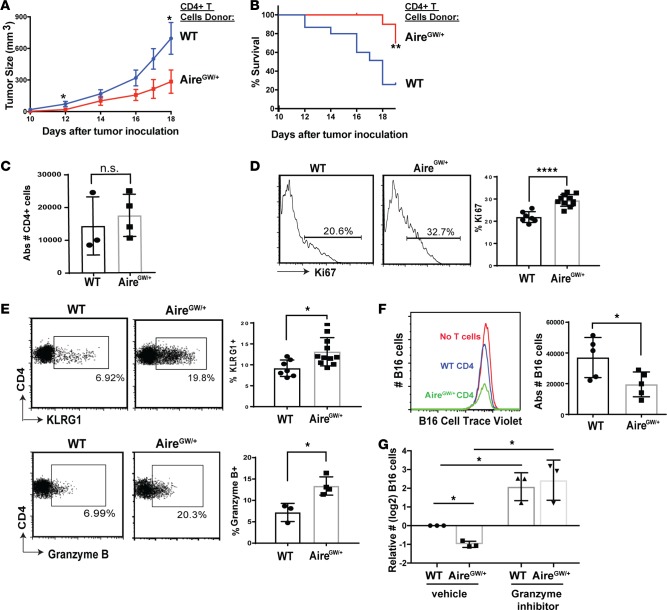
We next sought to delineate the cellular mechanism underlying enhanced antimelanoma immunity with combined Aire deficiency and CTLA-4 blockade. While it is known that Aire deficiency rescues melanoma-reactive CD4+ and CD8+ T cells from thymic deletion (10, 11), whether enhanced antitumor immunity in Aire-deficient mice is mediated by CD4+ or CD8+ T cells is unclear. To determine this, we purified CD4+ or CD8+ T cells from either AireGW/+ or WT littermates and transferred these cells into immunodeficient RAG−/− recipients. Recipients were then inoculated with syngeneic B16 melanoma cells. Reduced tumor growth and improved survival was associated with CD4+ T cells from AireGW/+ compared with WT donors (Figure 2, A and B). In contrast, CD8+ T cells from AireGW/+ and WT donors had similar effects on tumor growth and survival (Supplemental Figure 1, A and B; supplemental material available online with this article; https://doi.org/10.1172/jci.insight.93265DS1). While the absolute number of CD4+ T cells was not significantly increased (Figure 2C), Aire deficiency was associated with increased expression of Ki67, a marker of proliferation, on transferred CD4+ T cells (Figure 2D). Neither CD8+ absolute numbers nor Ki67 expression, however, was increased with Aire deficiency (Supplemental Figure 1, C and D). These data suggest the possibility that CD4+ T cells may mediate enhanced melanoma rejection in Aire deficiency.
Figure 2. Aire deficiency increases CD4+ T cell cytolytic function to reduce melanoma growth.
CD4+ splenocytes were transferred from WT and AireGW/+ donors into RAG–/– recipients, followed by s.c. B16 melanoma injection on day 7. Recipients were followed for tumor growth and survival. (A and B) B16 melanoma tumor growth and survival was measured after B16 inoculation in recipients; n = 15 per group. Mann-Whitney U test. *P < 0.05, **P < 0.01. (C–E) Tumor-infiltrating lymphocytes (TIL) were harvested on day 19 following B16 melanoma inoculation in recipients of either WT or AireGW/+ CD4+ splenocytes. Absolute numbers of CD4 tumor-infiltrating cells are shown in C. Two-tailed t test. Representative flow cytometry plots and average cumulative frequencies of Ki67+ (D) and KLRG1+ and granzyme B+ (E), among CD4+ T cells. Two-tailed t test. *P < 0.05, ****P < 0.0001. (F) Representative flow cytometry histograms and average absolute CellTrace Violet–labeled B16 cell numbers after coincubation with WT and AireGW/+ CD4+ T cells. Two-tailed t test. *P < 0.05. (G) Average relative B16 cell numbers (log2) after coincubation with CD4+ T cells from WT and AireGW/+ mice, along with pan-granzyme inhibitor (3, 4 Dichloroisocoumarin). One-way ANOVA and two-tailed t tests, with P values adjusted using Hommel’s correction for multiple comparisons. *P < 0.05.
Although CD8+ T cells are recognized as a T cell subset capable of killing cancer cells, CD4+ T cells also possess direct cytolytic capacity against tumors (22, 23). In particular, CD4+ T cells marked by killer cell lectin–like receptor subfamily G member 1 (KLRG1) express high levels of cytotoxicity-associated genes (24). Interestingly, frequency of KLRG1+ tumor infiltrating lymphocytes (TILs) was increased in recipients of CD4+ T cells from AireGW/+ compared with WT donors (Figure 2E, top). Additionally, the frequency of TILs expressing the cytolytic protein granzyme B was increased in recipients of CD4+ T cells from AireGW/+ compared with WT donors (Figure 2E, bottom). To test whether Aire-deficient CD4+ T cells have increased cytolytic capacity, we incubated purified AireGW/+ or WT CD4+ splenocytes with CellTrace Violet–labeled (CTV-labeled) B16 melanoma cell targets. A greater loss of B16 cells occurred with AireGW/+ compared with WT CD4+ T cells (Figure 2F). Similar numbers of B16 cells remained, on the other hand, after incubation with AireGW/+ and WT non-CD4+ T cells (Supplemental Figure 1E). Increased B16 cytolysis by AireGW/+ CD4+ T cells is mediated by granzyme, since addition of a granzyme inhibitor (3,4 dichloroisocoumarin) abrogated this effect (Figure 2G). Together, these findings suggest a critical role for cytolytic CD4+ T cells in mediating the enhanced melanoma rejection associated with Aire deficiency.
Additive effect of Aire deficiency and aCTLA-4 antibody on CD4+ T cell responses.
We next examined the T cell response in AireGW/+ mice treated with aCTLA-4 antibody. The frequency of Ki67+CD4+ TILs was highest in AireGW/+ mice treated with aCTLA-4 antibody, compared with iso control–treated AireGW/+ mice or aCTLA-4 antibody–treated WT mice (Figure 3A), which suggests that Aire deficiency and aCTLA-4 antibody cooperate to increase TIL proliferation. Furthermore, frequency of CD4+ T cells expressing markers associated with cytolytic activity (KLRG1, granzyme B) were also highest in AireGW/+ mice treated with aCTLA-4 antibody (Figure 3B). Among CD8+ T cells, Aire deficiency and aCTLA-4 antibody did not have additive effects on increasing the frequency of Ki67+ or KLRG1+ cells (Supplemental Figure 2). In aCTLA-4 antibody–treated mice, Aire deficiency increased the frequency of granzyme B+CD8+ T cells; notably, however, in all groups, the frequency of granzyme B+ cells among CD8+ T cells was quite low (<2% on average). In sum, these findings suggest that Aire deficiency and CTLA-4 blockade have additive effects, primarily through activating CD4+ T cells.
Figure 3. Additive effect of Aire deficiency and anti–CTLA-4 antibody on T cell responses.
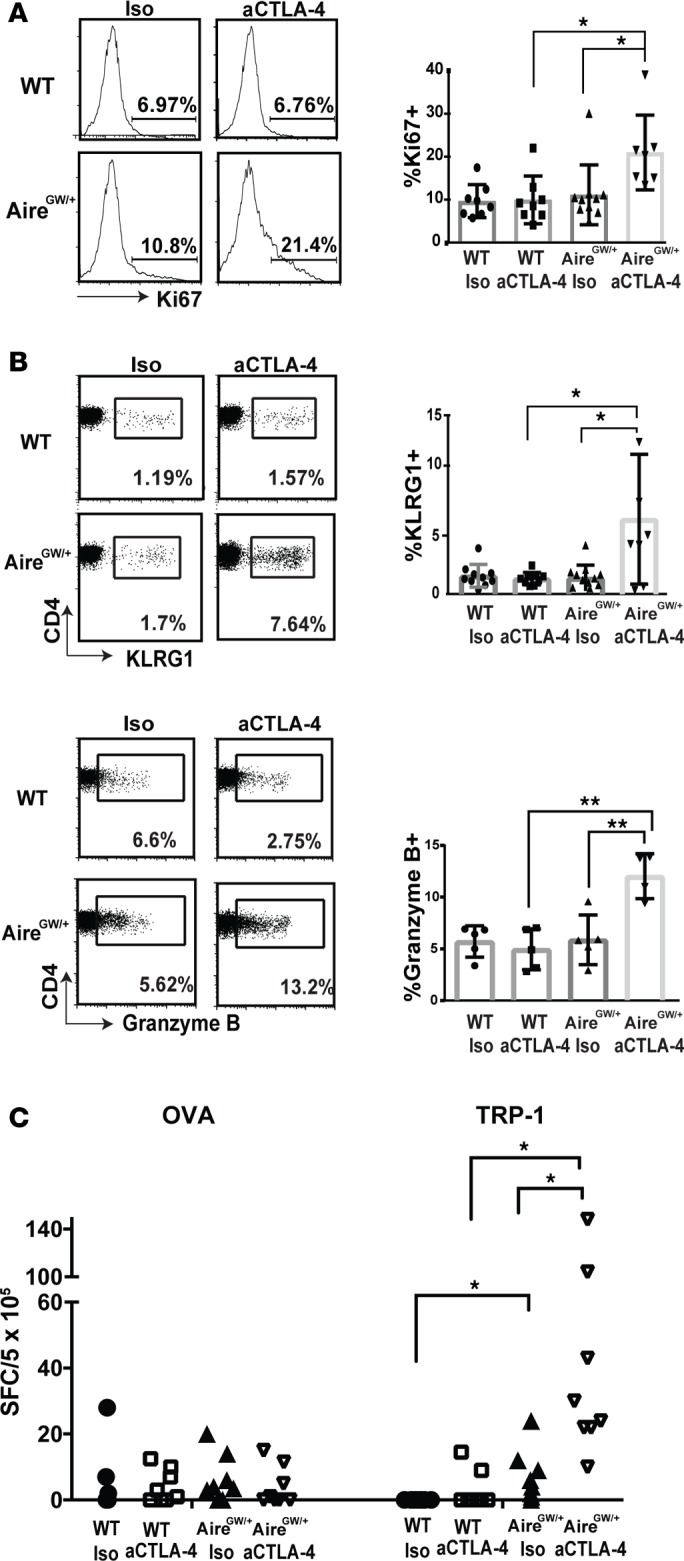
(A and B) Tumor-infiltrating lymphocytes (TIL) were harvested on day 19 following B16 melanoma inoculation in WT and AireGW/+ mice treated with aCTLA-4 or isotype control antibody (iso). Representative flow cytometry plots and average cumulative frequencies of Ki67+ (A) and KLRG1+ and granzyme B+ (B) among CD4+ T cells. n = 9–12 in each group. One-way ANOVA and two-tailed t tests with P values adjusted using Hommel’s correction for multiple comparisons. *P < 0.05, **P < 0.01. (C) ELISPOT with splenocytes harvested on day 14 following B16 melanoma inoculation in WT and AireGW/+ mice treated with anti–CTLA-4 antibody (aCTLA-4) or iso. Cumulative spot forming cells (SFC)/5×105 cells secreting IL-2 with OVA and TRP-1 was analyzed. Each data point represents an individual animal. One-way ANOVA and two-tailed t tests with P values adjusted using Hommel’s correction for multiple comparisons. *P < 0.05.
Interestingly, aCTLA-4 antibody administration in AireGW/+ mice increased the frequency of splenic as well as tumor-infiltrating CD4+FOXP3+ Tregs (Supplemental Figure 3, A–D). Furthermore, the ratio of effector-to-regulatory T cells (Teff/Treg) was similar, or lower, in Aire-deficient mice treated with aCTLA-4 antibody for both CD4+CD25–Teff cells and CD8+ Teff cells (Supplemental Figure 3, E and F). Thus, depletion of Tregs does not underlie the additive antimelanoma effect of Aire deficiency and CTLA-4 blockade.
Aire deficiency enhances antimelanoma effects of CTLA-4 blockade in a CD4+ TCR Tg mouse model.
We have previously reported that the self/melanoma antigen TRP-1 is expressed in the thymus under the control of Aire (11). We therefore sought to determine whether TRP-1–specific T cells might underlie the additive antimelanoma effects of Aire deficiency and CTLA-4 blockade. To determine the antigen-specificity of activated cells, we used an IL-2 ELISPOT assay to detect rare antigen-specific T cells in the spleen of melanoma-bearing mice. As expected, Aire deficiency, with or without aCTLA-4 antibody administration, did not affect the frequency of IL-2–producing T cells reactive against the irrelevant foreign antigen OVA (Figure 3C, left). In contrast, Aire-deficient mice treated with iso control antibody harbored an expanded population of IL-2–producing T cells reactive against TRP-1. Moreover, aCTLA-4 antibody treatment of Aire-deficient mice further expanded the precursor frequency of IL-2–producing T cells reactive against TRP-1 (Figure 3C, right). Thus, increased melanoma rejection in AireGW/+ mice treated with aCTLA-4 antibody is accompanied by expansion of T cells recognizing TRP-1 self/melanoma antigen.
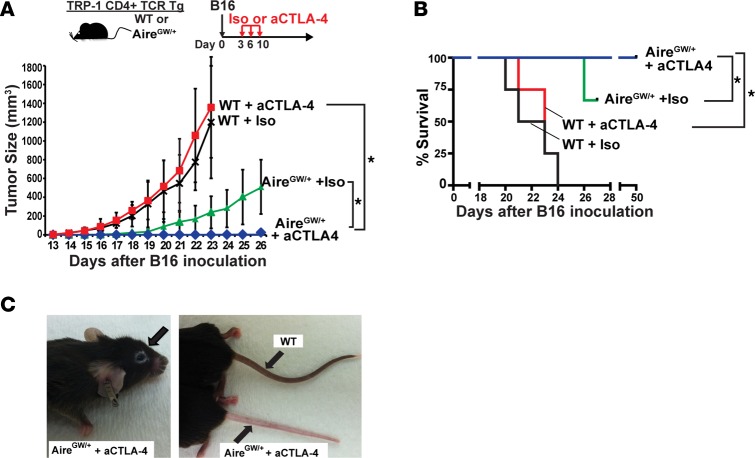
Given this finding, we used a CD4+ TCR Tg model to test whether CD4+ T cells reactive against TRP-1 may mediate the additive effects of blocking Aire and CTLA-4 in combination. TRP-1 TCR Tg mice are an MHCII-restricted mouse model in which CD4+ T cells recognize the melanoma antigen TRP-1 (25). In Aire sufficient (WT) TRP1-TCR Tg mice challenged with B16 melanoma, aCTLA-4 antibody administration did not affect melanoma growth or host survival (Figure 4, A and B). In AireGW/+ TRP1-TCR Tg littermates, on the other hand, aCTLA-4 antibody completely prevented tumor growth, with survival of all mice up to 50 days after tumor inoculation. These findings suggest that TRP-1–specific CD4+ T cells may mediate the additive antimelanoma effects of Aire deficiency and aCTLA-4 antibody administration in combination.
Figure 4. Additive antimelanoma effect of Aire deficiency and anti–CTLA-4 antibody in TRP-1 CD4+ TCR Tg mice.
(A and B) Aire sufficient (WT) and AireGW/+ TRP-1 TCR Tg mice were s.c. injected with 1 × 105 B16 melanoma cells followed by anti–CTLA-4 antibody (aCTLA-4) or isotype control antibody (Iso) treatment, as outlined. Mice in each group were followed for B16 melanoma tumor growth (A) and survival (B). n = 7–12 per group, cumulative of at least 2 independent experiments. Mann-Whitney U test. *P < 0.05. (C) Examples of periporbital and tail vitiligo in AireGW/+ mice treated with aCTLA-4 antibody.
Vitiligo is a T cell–mediated autoimmune condition associated with effective melanoma immunotherapy (26). Notably, periorbital and tail vitiligo (Figure 4C) was observed in AireGW/+ TRP-1 TCR Tg mice treated with aCTLA-4 antibody, reminiscent of epidermal vitiligo described in ref. 27. These findings provide evidence that Aire deficiency and CTLA-4 blockade also have additive effects in autoimmune destruction of melanocytes.
aCTLA-4 antibody does not impair thymic negative selection of TRP-1 TCR Tg T cells.
In our working model, we hypothesized that Aire deficiency and aCTLA-4 antibody have additive antimelanoma effects due to their function at distinct sites. Whereas Aire functions in the thymus, aCTLA-4 antibody functions in the immune periphery. However, it is possible that this model is incorrect and that aCTLA-4 may also function through altering thymocyte development. To test this possibility, we treated TRP-1 TCR Tg mice with either iso control or aCTLA-4 antibody. TRP-1 TCR Tg CD4 single positive (CD4SP) T cells undergo negative selection in an Aire-dependent manner (11) and therefore can be used to assess aCTLA-4 antibody effects. As expected, Aire deficiency results in defective negative selection, as demonstrated by the increased percent of CD4SP (%CD4SP) cells in thymi of AireGW/+ mice compared with WT (Supplemental Figure 4). In contrast, no change in %CD4SP was seen between aCTLA-4 and iso control antibody treatment in WT thymi. These findings suggest that CTLA-4 blockade does not inhibit thymic negative selection of melanoma-reactive CD4+ T cells. Instead, aCTLA-4 antibody may be functioning in an extrathymic manner to enhance antimelanoma immunity.
Aire deficiency does not exacerbate colitis severity in aCTLA-4 antibody–treated mice.
Immune-related colitis is a frequent ipilimumab-mediated toxicity in metastatic melanoma patients and can be life-threatening (2, 28). Likewise in mice, intestinal mucosal inflammation in experimental autoimmune colitis is exacerbated by aCTLA-4 antibody administration (29). Whether Aire deficiency promotes the colitogenic effects of CTLA-4 blockade, however, is not known. This question is of direct clinical relevance because exacerbation of immune-mediated adverse events is a potential limiting factor in combining CTLA-4 blockade with additional immune modulating therapies.
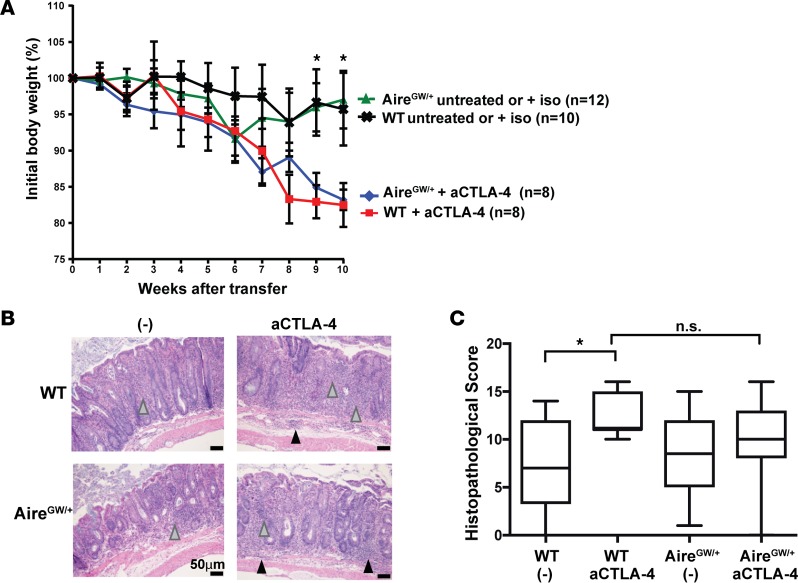
We utilized an autoimmune colitis model induced by adoptive transfer of naive T cells into RAG−/− recipients (30) to test the combinatorial effects of Aire deficiency and CTLA-4 blockade on colitis severity. Naive CD4+ (CD25−CD45RBhi) Teffs from WT or AireGW/+ mice were transferred into RAG–/– mice, and reconstituted recipients were treated with aCTLA-4 or iso control antibody. As expected based on prior studies (29), aCTLA-4 antibody treatment of recipients reconstituted with WT cells resulted in more severe weight loss compared with no treatment or iso control antibody (Figure 5A). Consistent with this finding, aCTLA-4 antibody administration was associated with more severe colonic inflammation by histological evaluation than control treatment in recipients of WT cells (Figure 5, B and C). Importantly, weight loss and colonic inflammation was not exacerbated by Aire deficiency in donors, even if recipients were treated with aCTLA-4 antibody. Thus, in this model of immune-mediated colitis, Aire deficiency does not exacerbate the colitogenic effects of aCTLA-4 antibody treatment. This finding is consistent with previous studies reporting that Aire does not regulate development of colitogenic T cells (31, 32).
Figure 5. Aire deficiency does not exacerbate the development of experimental autoimmune colitis associated with anti–CTLA-4 antibody treatment.
(A) Percent initial body weight of RAG–/– recipients after transfer of WT or AireGW/+CD4+CD25–CD45RBhi splenocytes. After transfer, recipients were treated with anti–CTLA-4 antibody (aCTLA-4), untreated, or treated with isotype control antibody (iso). Cumulative data from 2 independent experiments are shown. *P < 0.05 comparing aCTLA-4 antibody treatment versus untreated/iso treatment in recipients of WT cells. (B) Representative H&E-stained sections of descending colons and (C) average histopathological scores of colons from recipients of CD4+CD25–CD45RBhi splenocytes derived from WT and AireGW/+ mice and administered aCTLA-4 or untreated/iso (-). Gray arrowheads, immune cell infiltration in lamina propria; black arrowheads, immune cell infiltration in the submucosa. One-way ANOVA and two-tailed t tests with P values adjusted using Hommel’s correction for multiple comparisons. *P < 0.05.
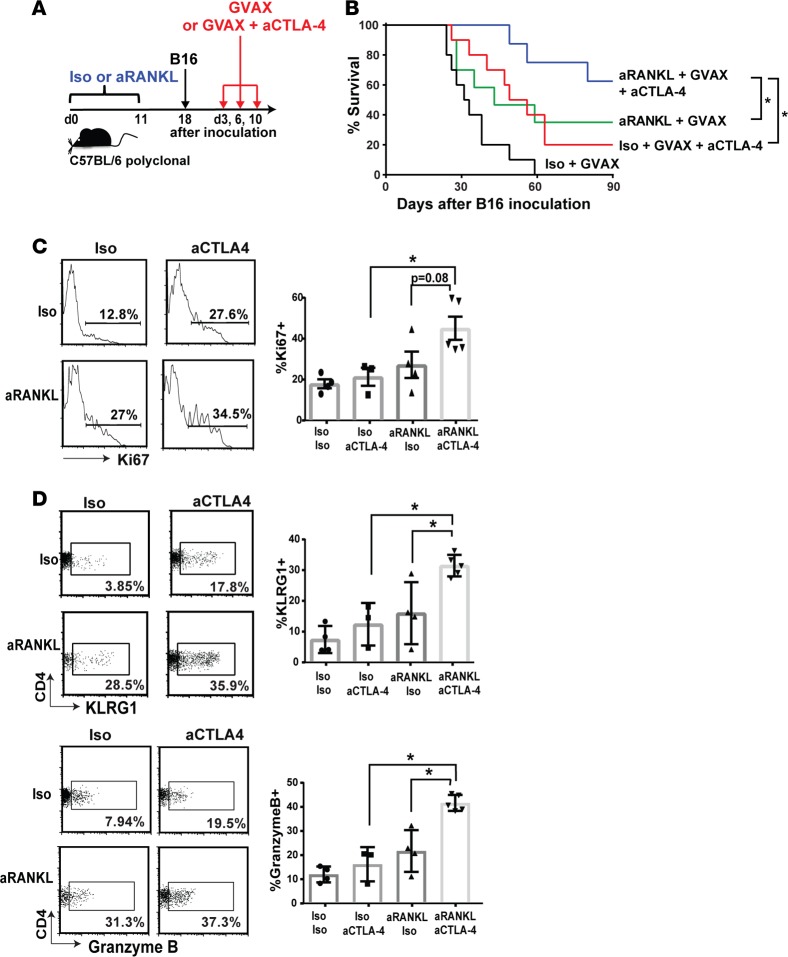
Anti-RANKL antibody enhances the immunotherapeutic effects of checkpoint inhibition in melanoma-bearing mice.
mTEC survival and Aire expression are dependent on RANKL signaling so that RANKL blockade with anti-RANKL (aRANKL) antibody depletes Aire-expressing mTECs in adult mice (15). This loss of Aire+ mTECS is associated with the rescue of TRP-1–specific CD4+ T cells from negative selection in the thymus (Supplemental Figure 5). aRANKL antibody thus represents a pharmacologic means to induce transient Aire deficiency through Aire-expressing mTEC depletion. Since anti-tumor effects of checkpoint inhibition could be enhanced by genetic Aire deficiency, we tested the effects of concurrent administration of aRANKL and aCTLA-4 antibodies on antimelanoma immunity. This treatment combination is clinically appealing, since aRANKL antibody is FDA approved for bone metastases, osteoporosis, and other indications, giving it a well-known safety profile (33).
Treatment with aRANKL and/or aCTLA-4 antibodies, however, did not have additive effects on improving survival in melanoma-bearing C57BL/6 polyclonal mice (data not shown). Previous studies have shown that vaccination with B16-GM-CSF (GVAX) potentiates an anti-B16 melanoma immune response through activation of innate immune cells and enhances presentation of tumor antigens to T cells (19, 34). Furthermore, addition of GM-CSF–secreting tumor vaccine to CTLA-4 blockade results in longer overall survival in patients with metastatic melanoma (16). We therefore utilized GVAX-vaccinated C57BL/6 mice to evaluate the effects of aRANKL and aCTLA-4 antibodies (Figure 6A). C57BL/6 polyclonal mice were administered aRANKL or iso control antibody and challenged with 2 × 104 B16 melanoma cells. At days 3, 6, and 10 following melanoma inoculation, mice were treated with aCTLA-4 antibody/GVAX or GVAX alone. As expected from prior reports (19), aCTLA-4 antibody/GVAX combination therapy was more effective than GVAX alone in prolonging host survival (Figure 6B). Of note, combination therapy with aRANKL/aCTLA-4 antibody/GVAX significantly improved host survival compared with iso control/aCTLA-4 antibody/GVAX therapy or aRANKL antibody/GVAX (Figure 6B). This suggests that, in the context of GVAX, aRANKL antibody and aCTLA-4 antibody have additive effects in improving host survival in response to melanoma challenge.
Figure 6. Combination aRANKL and aCTLA-4 antibody administration enhances immune rejection of melanoma.
(A) Schematic of treatment regimen, in which anti-RANKL (aRANKL) or isotype control (iso) antibody was injected every other day for 11 days, followed by s.c. B16 melanoma injection (2 × 104 cells) on day 18. GVAX with or without anti-CTLA-4 (aCTLA-4) antibody was given on days 3, 6, and 10 following melanoma injection. (B) Survival curves in GVAX-treated mice receiving indicated combinations of therapy. n = 10 for each group. Mann-Whitney U test. *P < 0.05. Tumor-infiltrating lymphocytes (TIL) were harvested on day 19 following 1 × 105 B16 melanoma cell inoculation. Representative flow cytometry plots and average cumulative frequencies of Ki67+ (C) and KLRG1+ and granzyme B+ (D) among CD4+ T cells was measured. n = 9–12 in each group. One-way ANOVA and two-tailed t tests with P values adjusted using Hommel’s correction for multiple comparisons. *P < 0.05.
To assess treatment effects on CD4+ T cells, we analyzed CD4+ TILs in GVAX-treated mice. Mice received either aRANKL or iso control antibody, were inoculated with 1 × 105 B16 melanoma cells, and then received either aCTLA-4/GVAX or iso control/GVAX. The frequency of Ki67+CD4+ T cells was significantly higher with aRANKL/aCTLA-4/GVAX, compared with iso/aCTLA-4/GVAX therapy (Figure 6C). Additionally, the frequency of KLRG1+ and granzyme B+ CD4+ T cells was increased in the aRANKL/aCTLA-4/GVAX-treated mice compared with the control groups (Figure 6D). These findings indicate that aRANKL and aCTLA-4 antibodies cooperate to increase the frequency of tumor-infiltrating CD4+ T cells expressing cytolytic markers.
Discussion
We report here that genetic Aire mutations in mice and protective Aire SNPs in humans are associated with increased efficacy of aCTLA-4 antibody. Additionally, we report that pharmacologic inhibition of Aire function with aRANKL antibody is sufficient to enhance the effects of CTLA-4 checkpoint inhibition. Findings in this paper suggest a model (Supplemental Figure 6) in which Aire normally purges self/melanoma antigen–reactive CD4+ T cells in the thymus, which limits the number of self/melanoma antigen–reactive CD4+ T cells available for peripheral activation by checkpoint inhibition. Genetic Aire deficiency or aRANKL antibody administration allows self/melanoma-reactive T cells to escape negative selection in the thymus, and the increased pool of self/melanoma-reactive T cells available for targeting by aCTLA-4 antibody enhances the efficacy of CTLA-4 blockade in immune rejection of melanoma.
In addition to aRANKL antibody, another therapeutic approach to increasing the pool of self/melanoma-reactive T cells in the periphery is through adoptive cellular transfer of self/melanoma-reactive T cells. Indeed, ex vivo expansion and reinfusion of T cells specific for self/melanoma antigens have beneficial effects for some patients who fail conventional therapy (23, 35). Of note, recent reports have demonstrated increased efficacy of adoptive cellular transfer when combined with aCTLA-4 antibody (36, 37). These findings provide additional supportive evidence that repopulating the periphery with self/melanoma-reactive T cells enhances the effects of CTLA-4 blockade. aRANKL antibody administration provides the advantage of being a relatively straightforward therapy when compared with adoptive cellular transfer, so it may be simpler to adapt to the clinical setting. Importantly, our data demonstrate the effects of aRANKL antibody prior to B16 inoculation. The effect of aRANKL antibody on already-established tumors will be the focus of future studies.
aCTLA-4 antibody treatment has been reported to have pleiotropic effects on T cells, which include intratumoral Treg depletion and promoting increased Teff/Treg ratios (18, 22, 38, 39). Interestingly, however, Aire deficiency and CTLA-4 blockade in combination did not result in intratumoral Treg depletion in our studies. Instead, decreased melanoma growth with Aire deficiency and CTLA-4 blockade was associated with increased intratumoral CD4+ T cell KLRG1 expression and cytolytic protein expression. These findings add to recent reports that CD4+ T cells have direct antitumor effects, especially if clonally expanded (22, 40), and that aCTLA-4 antibody treatment activates tumor-reactive cytotoxic CD4+ T cells (41).
CTLA-4 blockade is associated with colitis and other immune-related adverse events (2, 28). This finding is not surprising, since genetic deficiency of CTLA-4 in mice results in lymphoproliferation and multiple autoimmune manifestations (42, 43). Colitis can be life-threatening and is the most common ipilimumab-related toxicity in metastatic melanoma patients. Therefore, it is important to consider whether potential treatment modalities to be used in combination with CTLA-4 blockade will exacerbate colitis. We show in this study that lack of Aire function does not add to the severity of colitis in aCTLA-4 antibody–treated hosts. This result may seem unexpected, since Aire has a well-known role in preventing organ-specific autoimmune disease (44). Previous studies, however, have also reported that Aire does not regulate colitogenic T cell development (31, 32), possibly because colitogenic T cells may be directed against non–self-proteins such as colonic microbiota antigens. Thus, a combination of central tolerance blockade and checkpoint inhibition may improve a therapeutic index, compared with checkpoint inhibition alone.
aRANKL antibody represents a pharmacologic means to block central tolerance and can enhance the effects of checkpoint inhibitors in mice. In addition to its role in mTEC development, RANKL has a well-recognized role in the development, function, and survival of osteoclasts (45). For these effects, aRANKL antibody (denosumab) is FDA-approved for treatment of osteoporosis in postmenopausal women (46) and of other bone-related diseases (33). There is therefore substantial clinical experience with aRANKL antibody in patients, which should facilitate clinical testing to repurpose aRANKL antibody as an immunotherapy for the treatment of metastatic melanoma. Because the thymus involutes with age, whether thymus output can be manipulated in adults for therapeutic purpose is an open question. The median age at diagnosis of melanoma is 59 years, when thymic function is often assumed to be minimal. Multiple studies in mice and humans suggest that thymic function remains active in adults. Thymic output has been reported to continue in aged mice (47), and thymic function is measurable in adult humans up to age 76 (48). Our data that aRANKL antibody increases thymic output of TRP-1–specific T cells in adult mice (Supplemental Figure 5 and ref. 15) support the possibility that thymus function can be altered for therapeutic purpose in adults. Of note, a case report of a patient with metastatic melanoma who was concomitantly treated with aRANKL and aCTLA-4 antibodies also supports further study of this combination therapy (49).
Finally, checkpoint inhibitors have been shown to have therapeutic effects in a number of different cancers, including prostate and renal cancer (50). Thus, central tolerance blockade by aRANKL as a strategy to enhance the efficacy of checkpoint inhibition may be relevant in multiple cancer settings.
Methods
Mice.
C57BL/6 AireGW/+ mice (17) and C57BL/6 AireGW/+ TRP-1 TCR Tg RAG−/− mice (11) were housed and bred in sterile, specific pathogen-free mouse facilities at the University of North Carolina at Chapel Hill and UCSF. C57BL/6 Aire WT littermate controls were used in all experiments. RAG1−/− mice were purchased from the Jackson Laboratory.
Antibodies and flow cytometry.
Anti-CD4 (clone RM4-5), anti-CD3 (clone 145-2c11), anti-Ki67 (clone SolA15), anti-KLRG1 (clone 2F1), anti- granzyme B (clone NGZB), anti-FOXP3 (clone FJK-16s), and anti-CD25 (clone PC61.5) antibodies were purchased from eBioscience. Anti-CD8a (clone 5H10) antibody was purchased from Invitrogen. Intracellular staining for cytokines and FOXP3 were performed as in ref. 11. All samples were run on a Dako CyAn flow cytometer (Beckman-Coulter) and analyzed using FlowJo (TreeStar Inc.).
Isolation of TILs.
TILs were isolated as previously described (11). Tumors were dissected and minced before incubation with collagenase type I/IV and DNase I. Lymphocytes were then enriched on Percoll density gradient prior to flow cytometric analysis.
B16 melanoma and antibody administration.
B16-F10 is a TRP-1–expressing C57BL/6-derived spontaneous melanoma cell line originally purchased from the American Tissue Culture Condition. B16 melanoma cells were cultured as described in ref. 51. Mice were injected s.c. with 1 × 105 B16 melanoma cells as in ref. 11. Tumor measurements and survival were determined as described in ref. 15.
For immunotherapy experiments, aCTLA-4 mAb (clone 9D9; 100 μg/mouse, BioXcell) or IgG iso control antibody (clone MPC-11; 100 μg/mouse, BioXcell) were injected i.p. on days 3, 6, and 9 following B16 melanoma cell injection as described in ref. 52. For combination antibody administration experiments, aRANKL antibody (clone IKK22/5; 100 μg/mouse, BioXcell) or iso control antibody (clone 2A3; 100 μg/mouse, BioXcell) was injected every other day for a total of 6 doses. Nineteen days following treatment initiation, mice were s.c. injected with 2 × 104 B16-F10 melanoma cells for survival analysis and 1 × 105 for immune-phenotyping. On days 3, 6, and 10 following melanoma inoculations, aCTLA-4 antibody/GVAX therapy was administered as described in ref. 19.
Adoptive transfer of magnetically isolated (using Miltenyi Biotec beads) CD4+ and CD8+ T cells from the spleen of WT and AireGW/+ mice was performed in a 1:1 donor/recipient ratio into C57BL/6 RAG−/− recipients using a retro-orbital i.v. route of injection. After 7 days, recipients were injected s.c. with 1 × 105 B16-F10 melanoma cells. Tumor growth, survival, and development of vitiligo were monitored daily.
In vitro B16 cytotoxicity assay.
Splenocytes from WT and AireGW/+ melanoma–bearing C57BL/6 mice were purified using CD4 beads (Miltenyi Biotec) and cultured in vitro with 5 μg/ml TRP-1 peptide at 37°C. Following overnight incubation, peptide-primed CD4+ and non-CD4+ T cells were harvested and used for in vitro cytotoxicity assay with B16-F10 melanoma cells. To determine in vitro killing of tumor targets, B16 cells were loaded with 0.5 mM CTV and coincubated for 12–14 hours with peptide-primed CD4+ and non-CD4+ cells. Proliferation of CTV B16 cells was analyzed by flow cytometry and quantified using CountBright Absolute counting beads (Thermo-Fisher Scientific). A serine protease inhibitor (3, 4 Dichloroisocoumarin, Enzo Life Sciences; 25 μM) was used for pan-granzyme inhibition.
In vitro IL-2 ELISPOT assay.
ELISPOT assays for IL-2 release (BD Biosciences) were performed on splenic CD4+CD25– Teffs from B16-F10 tumor–bearing C57BL/6 WT or AireGW/+ mice. CD4+CD25− Teff cells were enriched by magnetic beads (Miltenyi Biotec). Cells (5 × 105) were plated for 20–22 hours on 96-well plates, which had been precoated with anti-mouse–purified IL-2 antibody overnight. Cells were left unstimulated in media (negative control) or stimulated with PMA-ionomycin (positive control). Cells were incubated in separate wells with 5 μg/ml of TRP1 (NCGTCRPGWRGAACNQKILTVR) purchased from Genemed Synthesis Inc. and OVA (ISQAVHAAHAEINEAGR) peptides purchased from InvivoGen. Culturing of the plates, washing, and counterstaining were performed according to manufacturers’ instructions. Visualization and analysis of the spots were performed on the ImmunoSpot counter (Cellular Technology Ltd.). IL-2 response of stimulated cultures was calculated by subtracting the number of spot-forming colonies in the negative unstimulated control from the number of spot-forming colonies in the wells stimulated by peptides/5 × 105 cells.
SNP genotyping.
Peripheral blood mononuclear cells (PBMC) collected as part of the ECOG-ACRIN Cancer Research Group study E1608 (16) were provided by the ECOG-ACRIN Central Biorepository and Pathology Facility. Following DNA extraction from PBMC, SNP genotyping assays (Applied Biosystems, TaqMan) were performed by the UNC-CH Mammalian Genotyping Core for the following SNPs of the Aire gene: rs1800522/rs1133779, rs2075876, rs56393821, rs1800520, and rs1055311. Genotyping for rs1055311 was confirmed by Sanger sequencing using two primers flanking exon 6 of the Aire gene (forward sequence: 5′-GAATGCAGGCTGTGGGAACT-3′; reverse sequence: 5′-AAGAGGGGCGTCAGCAATG-3′; product size 441 bp).
Experimental colitis.
CD4+ cells were enriched from WT and AireGW/+ female donor spleens using the CD4+ T cell Isolation Kit II (Miltenyi Biotec) according to manufacturer instructions. Cells were labeled and sorted for CD4+CD25–CD45RBhi cells by fluorescence-activated cell sorting, as previously described (53). Cells (5 × 105) were injected i.p. into 4- to 7-week-old RAG–/– recipient mice. Three days after transfer, recipient mice were injected i.p. with 100 μg of iso (clone MPC-11, BioXCell) or anti-CTLA4 (clone 9D9, BioXCell) antibody at 3-day intervals. Weights were measured weekly from initial cell transfer. Lack of survival was defined as death or weight loss > 20%.
Colon histopathology.
Colons were harvested at 10 weeks after adoptive transfer or earlier if weight loss of 20% was noted. Specimens were fixed in 10% formalin, sectioned (5 μm), and stained with H&E. Scoring for inflammation was performed as described (54, 55).
Statistics.
R, PRISM 5.0 (GraphPad Software Inc.), and Microsoft Office Excel software (2013) were used to analyze data. Two-tailed t tests were used to compare differences between 2 groups. One-way ANOVA and two-tailed t tests with P values adjusted using Hommel’s correction for multiple comparisons (56) were used to compare differences between more than 2 groups. Survival curves were compared by Mann-Whitney U test. P < 0.05 was considered significant.
Study approval.
Study approval for human samples was obtained from the ECOG Laboratory Science & Pathology and Executive Review Committees. All animal studies were approved by the Animal Care and Use Committees at UNC-CH and UCSF.
Author contributions
PB, MLZ, HHW, LKH, IK, MM, YH, and DS performed the experiments and performed statistical analyses. PB, MLZ, and MAS wrote the paper and prepared figures. JS, SL, and FZ performed statistical analyses. ASG, JMK, SM, LF, and MSA helped design experiments and reviewed the final manuscript. MZ, MSA, and MAS formulated the concept.
Supplementary Material
Acknowledgments
This work was supported by Department of Defense Career Development Award (MAS and PB); University Cancer Research Fund (MAS and SM); ECOG-ACRIN CA180820, CA21115, CA180794, CA23318, and CA66636; the University of Pittsburgh grants CA180844 and CA39229; and NIH R01 AI097457 (MSA).
Version 1. 09/21/2017
Electronic publication
Footnotes
Conflict of interest: The authors have declared that no conflict of interest exists.
Reference information: JCI Insight. 2017;2(18):e93265. https://doi.org/10.1172/jci.insight.93265.
Contributor Information
Pearl Bakhru, Email: pearl.bakhru@gmail.com.
Meng-Lei Zhu, Email: rabbitonline@gmail.com.
Hsing-Hui Wang, Email: hhwang@med.unc.edu.
Imran Khan, Email: Imran.Khan@ucsf.edu.
Maria Mouchess, Email: mmouchess@gmail.com.
Joshua Starmer, Email: starmer@unc.edu.
Yafei Hou, Email: yafeihou2015@yahoo.com.
David Sailer, Email: dsailer@email.unc.edu.
Sandra Lee, Email: sjlee@jimmy.harvard.edu.
Fengmin Zhao, Email: fezhao@jimmy.harvard.edu.
John M. Kirkwood, Email: kirkwoodjm@upmc.edu.
Stergios Moschos, Email: stergios_moschos@med.unc.edu.
Lawrence Fong, Email: lfong@medicine.ucsf.edu.
Maureen A. Su, Email: masu@email.unc.edu.
References
- 1.Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12(4):252–264. doi: 10.1038/nrc3239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hodi FS, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363(8):711–723. doi: 10.1056/NEJMoa1003466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Schadendorf D, et al. Pooled Analysis of Long-Term Survival Data From Phase II and Phase III Trials of Ipilimumab in Unresectable or Metastatic Melanoma. J Clin Oncol. 2015;33(17):1889–1894. doi: 10.1200/JCO.2014.56.2736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wolchok JD, et al. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2013;369(2):122–133. doi: 10.1056/NEJMoa1302369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ribas A, et al. Pembrolizumab versus investigator-choice chemotherapy for ipilimumab-refractory melanoma (KEYNOTE-002): a randomised, controlled, phase 2 trial. Lancet Oncol. 2015;16(8):908–918. doi: 10.1016/S1470-2045(15)00083-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.McArthur GA, et al. Safety and efficacy of vemurafenib in BRAF(V600E) and BRAF(V600K) mutation-positive melanoma (BRIM-3): extended follow-up of a phase 3, randomised, open-label study. Lancet Oncol. 2014;15(3):323–332. doi: 10.1016/S1470-2045(14)70012-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Weber JS, et al. Nivolumab versus chemotherapy in patients with advanced melanoma who progressed after anti-CTLA-4 treatment (CheckMate 037): a randomised, controlled, open-label, phase 3 trial. Lancet Oncol. 2015;16(4):375–384. doi: 10.1016/S1470-2045(15)70076-8. [DOI] [PubMed] [Google Scholar]
- 8.Mahoney KM, Rennert PD, Freeman GJ. Combination cancer immunotherapy and new immunomodulatory targets. Nat Rev Drug Discov. 2015;14(8):561–584. doi: 10.1038/nrd4591. [DOI] [PubMed] [Google Scholar]
- 9.Gotter J, Brors B, Hergenhahn M, Kyewski B. Medullary epithelial cells of the human thymus express a highly diverse selection of tissue-specific genes colocalized in chromosomal clusters. J Exp Med. 2004;199(2):155–166. doi: 10.1084/jem.20031677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Träger U, et al. The immune response to melanoma is limited by thymic selection of self-antigens. PLoS ONE. 2012;7(4):e35005. doi: 10.1371/journal.pone.0035005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhu ML, Nagavalli A, Su MA. Aire deficiency promotes TRP-1-specific immune rejection of melanoma. Cancer Res. 2013;73(7):2104–2116. doi: 10.1158/0008-5472.CAN-12-3781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Conteduca G, et al. The role of AIRE polymorphisms in melanoma. Clin Immunol. 2010;136(1):96–104. doi: 10.1016/j.clim.2010.03.002. [DOI] [PubMed] [Google Scholar]
- 13.Chambers CA, Cado D, Truong T, Allison JP. Thymocyte development is normal in CTLA-4-deficient mice. Proc Natl Acad Sci USA. 1997;94(17):9296–9301. doi: 10.1073/pnas.94.17.9296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Page DB, Postow MA, Callahan MK, Allison JP, Wolchok JD. Immune modulation in cancer with antibodies. Annu Rev Med. 2014;65:185–202. doi: 10.1146/annurev-med-092012-112807. [DOI] [PubMed] [Google Scholar]
- 15.Khan IS, et al. Enhancement of an anti-tumor immune response by transient blockade of central T cell tolerance. J Exp Med. 2014;211(5):761–768. doi: 10.1084/jem.20131889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hodi FS, et al. Ipilimumab plus sargramostim vs ipilimumab alone for treatment of metastatic melanoma: a randomized clinical trial. JAMA. 2014;312(17):1744–1753. doi: 10.1001/jama.2014.13943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Su MA, et al. Mechanisms of an autoimmunity syndrome in mice caused by a dominant mutation in Aire. J Clin Invest. 2008;118(5):1712–1726. doi: 10.1172/JCI34523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Simpson TR, et al. Fc-dependent depletion of tumor-infiltrating regulatory T cells co-defines the efficacy of anti-CTLA-4 therapy against melanoma. J Exp Med. 2013;210(9):1695–1710. doi: 10.1084/jem.20130579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.van Elsas A, Hurwitz AA, Allison JP. Combination immunotherapy of B16 melanoma using anti-cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) and granulocyte/macrophage colony-stimulating factor (GM-CSF)-producing vaccines induces rejection of subcutaneous and metastatic tumors accompanied by autoimmune depigmentation. J Exp Med. 1999;190(3):355–366. doi: 10.1084/jem.190.3.355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.O’Day SJ, et al. Efficacy and safety of ipilimumab monotherapy in patients with pretreated advanced melanoma: a multicenter single-arm phase II study. Ann Oncol. 2010;21(8):1712–1717. doi: 10.1093/annonc/mdq013. [DOI] [PubMed] [Google Scholar]
- 21.Wolchok JD, et al. Ipilimumab monotherapy in patients with pretreated advanced melanoma: a randomised, double-blind, multicentre, phase 2, dose-ranging study. Lancet Oncol. 2010;11(2):155–164. doi: 10.1016/S1470-2045(09)70334-1. [DOI] [PubMed] [Google Scholar]
- 22.Quezada SA, et al. Tumor-reactive CD4(+) T cells develop cytotoxic activity and eradicate large established melanoma after transfer into lymphopenic hosts. J Exp Med. 2010;207(3):637–650. doi: 10.1084/jem.20091918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hunder NN, et al. Treatment of metastatic melanoma with autologous CD4+ T cells against NY-ESO-1. N Engl J Med. 2008;358(25):2698–2703. doi: 10.1056/NEJMoa0800251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Curran MA, Kim M, Montalvo W, Al-Shamkhani A, Allison JP. Combination CTLA-4 blockade and 4-1BB activation enhances tumor rejection by increasing T-cell infiltration, proliferation, and cytokine production. PLoS One. 2011;6(4):e19499. doi: 10.1371/journal.pone.0019499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Muranski P, et al. Tumor-specific Th17-polarized cells eradicate large established melanoma. Blood. 2008;112(2):362–373. doi: 10.1182/blood-2007-11-120998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gogas H, et al. Prognostic significance of autoimmunity during treatment of melanoma with interferon. N Engl J Med. 2006;354(7):709–718. doi: 10.1056/NEJMoa053007. [DOI] [PubMed] [Google Scholar]
- 27.Harris JE, Harris TH, Weninger W, Wherry EJ, Hunter CA, Turka LA. A mouse model of vitiligo with focused epidermal depigmentation requires IFN-γ for autoreactive CD8+ T-cell accumulation in the skin. J Invest Dermatol. 2012;132(7):1869–1876. doi: 10.1038/jid.2011.463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Beck KE, et al. Enterocolitis in patients with cancer after antibody blockade of cytotoxic T-lymphocyte-associated antigen 4. J Clin Oncol. 2006;24(15):2283–2289. doi: 10.1200/JCO.2005.04.5716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Liu Z, et al. B7 interactions with CD28 and CTLA-4 control tolerance or induction of mucosal inflammation in chronic experimental colitis. J Immunol. 2001;167(3):1830–1838. doi: 10.4049/jimmunol.167.3.1830. [DOI] [PubMed] [Google Scholar]
- 30.Powrie F, Leach MW, Mauze S, Caddle LB, Coffman RL. Phenotypically distinct subsets of CD4+ T cells induce or protect from chronic intestinal inflammation in C. B-17 scid mice. Int Immunol. 1993;5(11):1461–1471. doi: 10.1093/intimm/5.11.1461. [DOI] [PubMed] [Google Scholar]
- 31.Anderson MS, Venanzi ES, Chen Z, Berzins SP, Benoist C, Mathis D. The cellular mechanism of Aire control of T cell tolerance. Immunity. 2005;23(2):227–239. doi: 10.1016/j.immuni.2005.07.005. [DOI] [PubMed] [Google Scholar]
- 32.Török HP, Tonenchi L, Glas J, Schiemann U, Folwaczny C. No significant association between mutations in exons 6 and 8 of the autoimmune regulator (AIRE) gene and inflammatory bowel disease. Eur J Immunogenet. 2004;31(2):83–86. doi: 10.1111/j.1365-2370.2004.00449.x. [DOI] [PubMed] [Google Scholar]
- 33. Chustecka Z. Denosumab Approved for Cancer Patients with Bone Metastases. Medscape. http://www.medscape.com/viewarticle/732891 Published November 19, 2019. Accessed August 22, 2017.
- 34.Dranoff G, et al. Vaccination with irradiated tumor cells engineered to secrete murine granulocyte-macrophage colony-stimulating factor stimulates potent, specific, and long-lasting anti-tumor immunity. Proc Natl Acad Sci USA. 1993;90(8):3539–3543. doi: 10.1073/pnas.90.8.3539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Chapuis AG, et al. Transferred melanoma-specific CD8+ T cells persist, mediate tumor regression, and acquire central memory phenotype. Proc Natl Acad Sci USA. 2012;109(12):4592–4597. doi: 10.1073/pnas.1113748109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chapuis AG, et al. T-Cell Therapy Using Interleukin-21-Primed Cytotoxic T-Cell Lymphocytes Combined With Cytotoxic T-Cell Lymphocyte Antigen-4 Blockade Results in Long-Term Cell Persistence and Durable Tumor Regression. J Clin Oncol. 2016;34(31):3787–3795. doi: 10.1200/JCO.2015.65.5142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chapuis AG, et al. Combined IL-21-primed polyclonal CTL plus CTLA4 blockade controls refractory metastatic melanoma in a patient. J Exp Med. 2016;213(7):1133–1139. doi: 10.1084/jem.20152021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kavanagh B, et al. CTLA4 blockade expands FoxP3+ regulatory and activated effector CD4+ T cells in a dose-dependent fashion. Blood. 2008;112(4):1175–1183. doi: 10.1182/blood-2007-11-125435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Quezada SA, Peggs KS, Curran MA, Allison JP. CTLA4 blockade and GM-CSF combination immunotherapy alters the intratumor balance of effector and regulatory T cells. J Clin Invest. 2006;116(7):1935–1945. doi: 10.1172/JCI27745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Malandro N, et al. Clonal Abundance of Tumor-Specific CD4(+) T Cells Potentiates Efficacy and Alters Susceptibility to Exhaustion. Immunity. 2016;44(1):179–193. doi: 10.1016/j.immuni.2015.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kitano S, et al. Enhancement of tumor-reactive cytotoxic CD4+ T cell responses after ipilimumab treatment in four advanced melanoma patients. Cancer Immunol Res. 2013;1(4):235–244. doi: 10.1158/2326-6066.CIR-13-0068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Tivol EA, Borriello F, Schweitzer AN, Lynch WP, Bluestone JA, Sharpe AH. Loss of CTLA-4 leads to massive lymphoproliferation and fatal multiorgan tissue destruction, revealing a critical negative regulatory role of CTLA-4. Immunity. 1995;3(5):541–547. doi: 10.1016/1074-7613(95)90125-6. [DOI] [PubMed] [Google Scholar]
- 43.Waterhouse P, et al. Lymphoproliferative disorders with early lethality in mice deficient in Ctla-4. Science. 1995;270(5238):985–988. doi: 10.1126/science.270.5238.985. [DOI] [PubMed] [Google Scholar]
- 44.Nagamine K, et al. Positional cloning of the APECED gene. Nat Genet. 1997;17(4):393–398. doi: 10.1038/ng1297-393. [DOI] [PubMed] [Google Scholar]
- 45.Xing L, Schwarz EM, Boyce BF. Osteoclast precursors, RANKL/RANK, and immunology. Immunol Rev. 2005;208:19–29. doi: 10.1111/j.0105-2896.2005.00336.x. [DOI] [PubMed] [Google Scholar]
- 46.Cummings SR, et al. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med. 2009;361(8):756–765. doi: 10.1056/NEJMoa0809493. [DOI] [PubMed] [Google Scholar]
- 47.Hale JS, Boursalian TE, Turk GL, Fink PJ. Thymic output in aged mice. Proc Natl Acad Sci USA. 2006;103(22):8447–8452. doi: 10.1073/pnas.0601040103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Poulin JF, et al. Direct evidence for thymic function in adult humans. J Exp Med. 1999;190(4):479–486. doi: 10.1084/jem.190.4.479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Smyth MJ, Yagita H, McArthur GA. Combination Anti-CTLA-4 and Anti-RANKL in Metastatic Melanoma. J Clin Oncol. 2016;34(12):e104–e106. doi: 10.1200/JCO.2013.51.3572. [DOI] [PubMed] [Google Scholar]
- 50.Egen JG, Kuhns MS, Allison JP. CTLA-4: new insights into its biological function and use in tumor immunotherapy. Nat Immunol. 2002;3(7):611–618. doi: 10.1038/ni0702-611. [DOI] [PubMed] [Google Scholar]
- 51.Kruczynski A, Hill BT. Classic in vivo cancer models: three examples of mouse models used in experimental therapeutics. Curr Protoc Pharmacol. 2002;Chapter 5:Unit5.24. doi: 10.1002/0471141755.ph0524s15. [DOI] [PubMed] [Google Scholar]
- 52.Curran MA, Montalvo W, Yagita H, Allison JP. PD-1 and CTLA-4 combination blockade expands infiltrating T cells and reduces regulatory T and myeloid cells within B16 melanoma tumors. Proc Natl Acad Sci USA. 2010;107(9):4275–4280. doi: 10.1073/pnas.0915174107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ostanin DV, et al. T cell-induced inflammation of the small and large intestine in immunodeficient mice. Am J Physiol Gastrointest Liver Physiol. 2006;290(1):G109–G119. doi: 10.1152/ajpgi.00214.2005. [DOI] [PubMed] [Google Scholar]
- 54.Ostanin DV, et al. T cell transfer model of chronic colitis: concepts, considerations, and tricks of the trade. Am J Physiol Gastrointest Liver Physiol. 2009;296(2):G135–G146. doi: 10.1152/ajpgi.90462.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Read S, Powrie F. Induction of inflammatory bowel disease in immunodeficient mice by depletion of regulatory T cells. Curr Protoc Immunol. 2001;Chapter 15:Unit 15.13. doi: 10.1002/0471142735.im1513s30. [DOI] [PubMed] [Google Scholar]
- 56.Hommel G. A Stagewise Rejective Multiple Test Procedure Based on a Modified Bonferroni Test. Biometrika. 1988;75:383–386. doi: 10.1093/biomet/75.2.383. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.