Abstract
Background
Ginsenosides are the main ingredients of ginseng, which, in traditional Eastern medicine, has been claimed to have therapeutic values for many diseases. In order to verify the effects of ginseng that have been empirically observed, we utilized the reverse docking method to screen for target proteins that are linked to specific diseases.
Methods
We constructed a target protein database including 1,078 proteins associated with various kinds of diseases, based on the Potential Drug Target Database, with an added list of kinase proteins. We screened 26 kinds of ginsenosides of this target protein database using docking.
Results
We found four potential target proteins for ginsenosides, based on docking scores. Implications of these “hit” targets are discussed. From this screening, we also found four targets linked to possible side effects and toxicities, based on docking scores.
Conclusion
Our method and results can be helpful for finding new targets and developing new drugs from natural products.
Keywords: drug target, ginsenoside, ligand screening, protein docking, reverse docking
1. Introduction
Korean ginseng is well known as a medicinal herb that has widely been used in the traditional medicine field. Ginseng has pharmacological actions because of the saponin ginsenoside, about 40 types of which have been found. Depending on the method of production, ginseng can be classified into white ginseng (Ginseng Radix Alba) and red ginseng (Ginseng Radix Rubra). White ginseng is produced by air drying fresh ginseng, whereas red ginseng is produced by first steaming fresh ginseng and then air drying it [1]. Owing to this difference in production methods, red ginseng contains more Rg1 and Rb1 ginsenosides than white ginseng.
Virtual screening, which has become a de facto standard in modern-day drug discovery, is a computational method for identifying potent molecules binding to a specific target protein from a large and chemically diverse molecule library [2]. It is a one-target many-ligand concept [3]. Virtual screening-based drug designs have successfully resulted in some approved drugs in recent history. Central to virtual screening is a computational method called docking [4]. A docking program predicts the binding mode of a small molecule/target protein complex. In order to find the most plausible binding modes, a docking program ranks possible conformations using a scoring function.
Reverse docking is a recent method that does the opposite of virtual screening through the use of docking. One screens a database of target proteins against an active compound—the one-ligand many-target concept, and just as in virtual screening, uses docking to find correct binding modes for ligand–target protein complexes [3]. However, in reverse docking, target proteins for a given ligand rather than ligands for a given target protein are ranked. Utilizing the list of ranked target proteins, the relevance of a given ligand for particular diseases or its side effects can be estimated. Therefore, the reverse docking method is useful for drug repositioning [5], in which one looks for new targets of drugs already approved or of natural products the exact effects of which are not yet known [6].
Ginsenosides are known to have many therapeutic values: they have antiallergic, antioxidant, and immune-stimulatory properties, and can modulate blood pressure, metabolism, and immune functions. Ginsenosides are named according to their retention factor value in thin-layer chromatography. Rb1, Rb2, Rb3, Rc, Rd, Rg3, Rg5, Rh2, Rh3, Rs3, and compound K are 20(S)-protopanaxadiols, and Re, Rf, Rg1, Rg2, Rg4, Rh4, and Rh5 are 20(S)-protopanaxatriols [7]. Furthermore, several ginsenosides, such as the ocotillol saponins F2, F3, F5, and F11 [8] and the pentacyclic oleanane saponin Ro [9] have also been identified. Recent studies have linked these ginsenosides to multiple bioactivities including neuroprotection, antioxidation, angiogenesis modulation, and cytotoxicity [10]. In this work, we utilized the reverse docking technique to elucidate and/or confirm therapeutic values and side effects of ginsenosides by screening a target protein database against them. Chen et al [11] have used a reverse docking method, called INVDOCK, to extract possible drug targets by predicting interactions between compounds including ginsenoside Rg1, which are found in medicinal plants, and human and mammalian proteins. However, in order to fully assess the therapeutic values of ginseng, one needs to analyze the full array of ginsenosides. We also utilized more comprehensive target protein databases developed recently. In terms of the reverse docking method, INVDOCK is proprietary and little is known about its performance as a docking method. In our work, the well-known commercial docking program Glide (version 6.7; Schrödinger, Inc., New York, NY, USA) was used as part of our protocol, making it easy to replicate. In addition to potential drug targets, we also examined interactions of ginsenosides with toxicity- and side effect-related target proteins. Details of the methodology are described in the Materials and methods section. Our analysis of the targets, which were found to interact with ginsenosides, is summarized in the Results section. Our results not only validate the previously identified therapeutic values of ginsenosides, but also give insights into the overlooked ones.
2. Materials and methods
2.1. Construction of drug target database
In this work, we used the Potential Drug Target Database (PDTD) as our drug target database [12], [13]. The PDTD contains 1,207 entries covering 841 known and potential drug targets with known three-dimensional structures presented in the Research Collaboratory for Structural Bioinformatics (RCSB) Protein Data Bank. The target proteins collected in the PDTD were selected from the literature and online databases, such as DrugBank and the Therapeutic Target Database [13]. We selected 529 entries, which are classified as therapeutic in the PDTD, and then constructed a drug target database for reverse docking. Furthermore, 549 kinase targets were collected from an online database and added to the drug target database [14]. Diseases related to the targets in our database are bacterial/fungal/viral/parasitic infections, blood and neuronal disorders, inflammation, renal disorders, cardiovascular disorders, gastrointestinal disorders, cancer, and kinase-related disorders (Table 1). Each record of a target was annotated by hyperlinks to other databases, such as DrugBank, Therapeutic Target Database, the Expert Protein Analysis System (ExPASy) proteomics server, and Kyoto Encyclopedia of Genes and Genomes (KEGG).
Table 1.
Diseases related to target proteins in our database1)
| (1) Bacterial infections (44) |
| (2) Diseases of the blood & blood-forming organs (51) |
| (3) Fungal infections (9) |
| (4) Disorders of gastrointestinal functions (15) |
| (5) Hormone- & hormone antagonist-related diseases (82) |
| (6) Immunomodulation (45) |
| (7) Inflammation (24) |
| (8) Neoplastic diseases (85) |
| (9) Parasitic infectious diseases (27) |
| (10) Renal & cardiovascular disorders (30) |
| (11) Diseases of the synaptic & neuroeffector junctional sites, central nervous system (25) |
| (12) Vitamin deficiency diseases (25) |
| (13) Viral infections (67) |
| (14) Kinase-related disorders (549) |
Numbers in parentheses indicate how many of the targets are in our database
2.2. Selection of toxicity- and side-effect-related protein targets
In addition to the disease-related targets from PDTD, we included proteins that are linked to side effects and toxicities based on available data from the medical biochemistry literature. It is well known that the inhibition of some proteins important in normal cellular function may result in toxicity or side effects [15]. These proteins are involved in important cellular metabolism processes such as amino acid and nucleotide metabolism, the glycolytic pathway, and the urea cycle. There are 73 Protein Data Bank (PDB) entries for 20 types of proteins connected to toxicities and side effects.
2.3. Preparation of ginsenoside ligand and target structure
We prepared 26 kinds of ginsenoside structures [7] with known structures presented in the PubChem [16]. The structures were generated by the two-dimensional Sketcher in Maestro, after which LigPrep (version 3.4; Schrödinger Inc., New York, NY, USA) was used to convert them into three-dimensional structures. During the preparation, the force field was set to OPLS-2005, and all the combinations of stereoisomers were generated. They were assigned protonation states at pH 7.0. Our target database contained 1,078 protein structures, and the cocrystal complexes were downloaded from the RCSB Protein Data Bank. Missing residues and atoms of each protein structure were repaired using the Schrödinger Protein Preparation Wizard. The Protein Preparation Wizard automatically fixes incomplete residues and determines the tautomeric states of histidine, glutamine, and asparagine residues. Water molecules and cofactors were removed except for ones influencing the binding site. Hydrogen atoms were added, and the positions were optimized to 0.3 Å root-mean square deviation (RMSD) with heavy atoms fixed.
2.4. Reverse docking using Glide
All the reverse dockings in this research were performed with Schrödinger's Glide (version 6.7; Schrödinger Inc.). We used the standard precision mode of Glide, which is efficient and accurate for most of the targets [17], [18]. Glide generates the possible binding modes of ligand–protein complexes and scores them with GlideScore, a mixture of interaction energy and parameter-based penalty functions that roughly represents binding energy. The reverse docking procedure was performed as follows: (1) using a Python script, we generated an input file for each of the 26 kinds of ginsenosides for docking with all the target proteins in our database; (2) using Glide, each ginsenoside was docked to all 1,078 target proteins; and (3) docking results were sorted by docking scores and arranged into a matrix form. The resulting docking profiles were clustered for further analysis.
3. Results
3.1. Reverse docking results
To identify the efficacy of the 26 kinds of ginsenosides, we performed reverse docking with a database of 1,078 target proteins. For a detailed analysis, we clustered the entire docking results by scores. Fifty-two percent of all target proteins were docked by one or more of the 26 ginsenosides. Rh4, in particular, interacted with 72% of the targets. The average GlideScore of the docked results was −5.285 kcal/mol. Rc had an average of −6.249 kcal/mol across all the docked targets, which was the highest. In addition, 336 target proteins interacted with more than 21 ginsenosides, which suggested that several ginsenosides can have activities toward a single target. For further analysis as to what kind of implications these data might have on the therapeutic value of ginsenosides, we calculated the binding energy of each target protein's native ligand to compare it with the binding energies of the ginsenosides.
3.2. Identification of therapeutic targets
In order to identify specific targets related to diseases for which ginsenosides can be therapeutic, we compared the binding energies of ginsenosides with those of native ligands. In Table 2, the top 10 targets for each ginsenoside are listed. We also checked if these targets were experimentally found to be associated with ginsenosides. There were 63 cases in which the binding energies of ginsenosides were below both those of native ligands and −8 kcal/mol. We selected four potential targets for ginsenosides based on their frequency of showing as targets for each ginsenoside and the number of ginsenosides that interacted with each target. These cases exhibited therapeutic targets for which ginsenosides could be effective.
Table 2.
Top 10 potential targets of ginsenosides according to docking scores
| PDB ID | Target name | Native ligand |
Ginsenoside |
Implicated by experiment2) | Reference | |
|---|---|---|---|---|---|---|
| Docking score1) | Type | Docking score | ||||
| 3OS3 | Dual specificity mitogen-activated protein kinase kinase 1 | −7.278 | Rf | −12.681 | Yes | [19] |
| 3O96 | RAC-alpha serine/threonine-protein kinase | −10.836 | Rs3 | −11.394 | No | |
| 1MCH | Immunoglobulin lambda light chain dimer (Mcg) | −7.988 | Rf | −10.217 | No | |
| 2ITX | Epidermal growth factor receptor precursor | −5.256 | R2 | −10.090 | Yes | [20] |
| 1TUI | Elongation factor 1-alpha | −8.649 | Ro | −9.718 | No | |
| 1ETT | Thrombin | −8.384 | Rg1 | −9.566 | Yes | [21] |
| 3FGU | Glucokinase | −3.853 | Ro | −9.346 | Yes | [22] |
| 1DHT | Estrogenic 17-beta hydroxysteroid dehydrogenase | −5.785 | Rh5 | −9.306 | No | |
| 2PVR | Casein kinase II subunit alpha | −5.499 | R2 | −9.240 | No | |
| 1OL6 | Aurora kinase A | −8.342 | Re | −9.211 | No | |
Docking scores from redocking of receptor proteins and native ligands of PDB entries
Whether or not there is an experimental result of the particular ginsenosides and the target in the literature
The target with which ginsenosides interacted most was mitogen-activated protein kinase 1 (MEK1). Ten different MEK1 proteins out of 16 in the target database had significant interactions with at least one ginsenoside, and 11 ginsenosides were involved in these interactions for a total of 28 interactions. MEK1 is known to be linked to cell proliferation in most colon cancer cells and thus has received attention as a new target for anticancer drugs treating colorectal cancer [23]. Experimentally, ginsenoside Rg3 was found to be effective for MEK1 [19]. Our docking study found other ginsenosides that are effective as well. In particular, ginsenoside Rh4 was shown to be highly interactive with 10 MEK1 proteins, with an average docking score of −10.077 kcal/mol (Table 3).
Table 3.
Comparison of docking scores between ginsenoside Rh4 and native ligands docked to MEK1 proteins
| PDB ID | Native ligand1) |
Rh4 |
|---|---|---|
| Docking score | Docking score | |
| 2P55 | −6.706 | −9.0199 |
| 3DY7 | −6.887 | −9.797 |
| 3E8N | −7.692 | −11.158 |
| 3EQB | −5.45 | −9.955 |
| 3EQC | −6.163 | −9.168 |
| 3EQH | −9.175 | −9.258 |
| 3MBL | −8.253 | −11.28 |
| 3OS3 | −7.278 | −11.143 |
| 3PP1 | −7.692 | −9.457 |
PDB and cocrystal ligand redocking scores
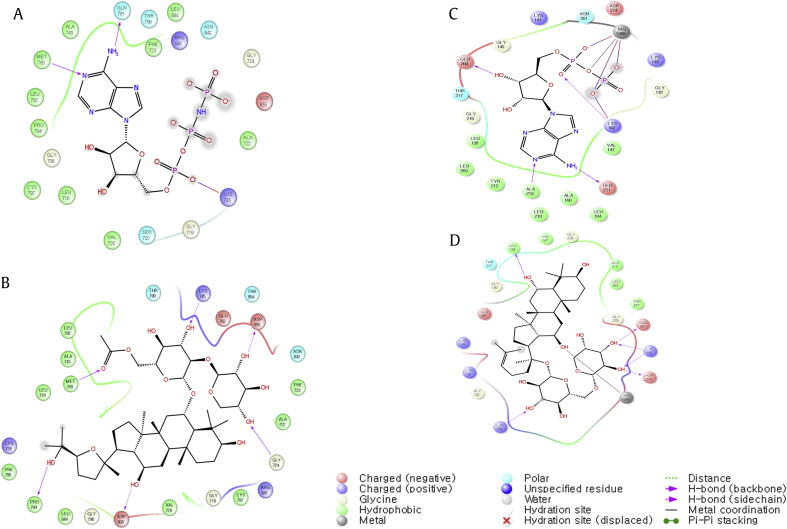
The next most active was epidermal growth factor receptor (EGFR). Nine different EGFR proteins interacted significantly with eight ginsenosides. EGFR has been identified as an oncogene, and its inhibitors have been developed for anticancer therapeutics, examples of which include gefitinib, erlotinib, afatinib, brigatinib, and icotinib for lung cancer, and cetuximab for colon cancer. However, these inhibitors turned out to be ineffective against mutant EGFRs, and for this reason, natural compounds have been studied as EGFR inhibitors [24]. Li et al [25] showed that ginsenoside Rh2 can substantially inhibit the growth of glioblastoma in vitro and in vivo in a mouse model. In glioblastoma, a more or less specific mutation of EGFR, EGFRvIII, is often observed. Furthermore, Sathishkumar et al [20] identified 14 compounds of Panax ginseng that can be developed as EGFR tyrosine kinase inhibitors using pharmacophore modeling and molecular docking. In our docking study, not only the wild type of EGFR, but also two types of mutants, L858R and G719S, showed interactions with ginsenosides (Table 4). The average docking score between EGFR and 14 ginsenosides was −8.546 kcal/mol. In Fig. 1A, binding pose interaction diagrams of complexes of EGFR wild-type protein (PDB ID: 2ITX) with the native ligand (ANP) at the top and with ginsenoside R2 at the bottom are shown. From these diagrams, one can see that the native ligand ANP interacts with LYS745 and MET793, and ginsenoside R2 makes the same contact as ANP, but also makes additional contacts with a few surrounding residues including PRO794. This comparison is consistent with the calculated binding affinity score, for which R2 doubled ANP (Table 4).
Table 4.
Interactions of EGFR and mutant EGFR target proteins with ginsenosides
| Type | PDB ID | Ginsenoside |
Interacting residues | |
|---|---|---|---|---|
| Type | Docking score | |||
| Wild | 2GS6 | Rb2 | −8.991 | MET769, GYS773, GLU734 |
| Rb3 | −8.232 | ASP776, MET769, LYS889, GLU738 | ||
| Ro | −8.105 | CYS773, ASP813, GLU738 | ||
| 2ITX | R2 | −10.090 | GLY724, LYS745, PRO794 | |
| 2ITY | Rb2 | −8.071 | ARG841, ASN842, GLU804 | |
| L858R | 2EB3 | R2 | −8.197 | MET793, GLU762, ASP800 |
| Rf | −8.295 | MET793, ASP800, GLU762, ASP855 | ||
| 2ITU | Rg1 | −8.987 | CYS797, GLN791, GLY724 | |
| Rg3 | −8.047 | GLN791, Pro794, | ||
| Ro | −8.447 | LYS728, GLN791, PRO794 | ||
| 2ITZ | Rb2 | −8.294 | PRO794, ASP855, LYS716 | |
| G719S | 2ITN | R2 | −8.037 | GLY724, LYS745, ASP837, MG |
| 2ITO | Ro | −8.224 | ARG841, ASP855, PHE795 | |
| 2ITP | Rc | −9.622 | PRO794, ASP800 | |
EGFR, epidermal growth factor receptor
Fig. 1.
Binding mode diagrams between target proteins and native ligands or ginsenosides. EGFR protein (PDB ID: 2ITX): (A) 2ITX-ANP, docking score −8.637 kcal/mol; (B) 2ITX-ginsenoside R2, docking score −10.090 kcal/mol, and Aurora kinase A protein (PDB ID: 1OL5); (C) 1OL5-ADP, docking score −7.979 kcal/mol; and (D) 1OL5-ginsenoside F3, docking score −8.480 (kcal/mol). EGFR, epidermal growth factor receptor.
Twelve kinds of ginsenosides interacted with the serine protease thrombin, which has received much attention due to its central role in the initiation of blood coagulation and hemostatic disorder [26]. Zhou et al [21] suggested that Rg1 could serve as a good resource for the screening and development of novel therapeutic agents for thrombotic disorders. In accordance with the experimental results, our docking study showed that the thrombin target protein interacts with Rg1, with a docking score of −9.566 kcal/mol. In addition, four kinds of serine proteinase alpha-thrombin target proteins and 10 kinds of ginsenosides showed higher interaction energies than native ligands, implying that they could be good targets for ginsenosides (Table 5).
Table 5.
Comparison of docking scores between ginsenosides and native ligands docked to thrombin and Aurora kinase A target proteins
| Target name | PDB ID | Native ligand1) docking score | Ginsenoside |
|
|---|---|---|---|---|
| Type | Docking score | |||
| Serine protease thrombin | 1ETR | −6.202 | Rg1 | −9.055 |
| 1ETT | −8.384 | Rg1 | −9.566 | |
| Serine proteinase alpha-thrombin | 1A4W | −7.177 | La | −8.802 |
| Rb2 | −8.321 | |||
| F5 | −8.153 | |||
| 1AE8 | −6.427 | Rg1 | −8.902 | |
| 1DWC | −6.613 | CK | −8.409 | |
| Rb1 | −8.329 | |||
| Rh3 | −8.329 | |||
| Re | −8.311 | |||
| R2 | −8.254 | |||
| 4THN | −7.264 | A1 | −8.131 | |
| Aurora kinase A | 1OL5 | −7.979 | A1 | −9.014 |
| F3 | −8.48 | |||
| Rg2 | −8.13 | |||
| 1OL6 | −8.342 | Re | −9.211 | |
| 2W1C | −7.005 | R2 | −8.559 | |
| 2W5B | −8.03 | Ro | −8.235 | |
| 2XRU | −7.778 | Rg5 | −8.168 | |
PDB and cocrystal ligand redocking scores
Ginsenosides also interacted with Aurora kinase A, which is a member of the serine/threonine kinase family. It plays an important role in completing mitotic events such as centrosome separation, biopolar spindle assembly, chromosome segregation, and cytokinesis [27]. Dysregulation of Aurora kinase A may lead to cancer because it is required for the completion of cytokinesis. Thus, Aurora kinase A has been identified as a potential anticancer drug target [28]. Our docking study showed that seven different ginsenosides interacted with four Aurora kinase A proteins, with an average docking score of −8.543 kcal/mol (Table 5). In case of Aurora kinase A target protein, no experimental results related to ginsenosides were found in the literature. However, an analysis of the binding poses of the native ligand and ginsenoside F3 in the binding site of Aurora kinase A target protein gave some clues as to whether or not ginsenosides can be an effective binder. In Fig. 1, ADP, which is the native ligand of Aurora kinase A protein (PDB ID: 1OL5), interacts strongly with LYS162 and magnesium metal ion. Ginsenoside F3 interacts with a few other residues as well as LYS162 and magnesium. The implication is that ginsenoside F3 binds to Aurora kinase A protein more strongly than the native ligand, as our prediction of binding scores also indicate.
3.3. Potential toxicity and side effects of ginsenosides
In order to identify potential side effects and toxicity of ginsenosides, we selected 73 targets from 20 kinds associated with toxicity and side effects, and examined their interaction with ginsenosides (Table 6). Acetylcholinesterase interacted with 25 ginsenosides (all except for Re), with an average docking score of −5.056 kcal/mol. The inhibition of acetylcholinesterase is related to cholinergic toxicity [29]. Ginsenosides Rh4, Rh5, and CK showed significant interaction with intestinal fatty acid-binding protein (I-FABP), with an average docking score of −8.502 kcal/mol. If intestinal fatty acid-binding protein is inhibited, triglyceride accumulation declines and as a result, normal adipogenesis is suppressed [30]. Carbonic anhydrase type II (CA II) was predicted to interact with 26 ginsenosides in our docking results, with an average docking score of −5.974 kcal/mol. The inhibition of CA II is known to cause nephrotoxicity since CA is a key enzyme that regulates the bicarbonate level in the blood [15]. Glutamate dehydrogenase was also shown to interact with ginsenoside Rh4, with a docking score of −6.614 kcal/mol. Glutamate dehydrogenase plays important roles in amino acid metabolism, and thus its inhibition could lead to nephrotoxicity [31].
Table 6.
Toxicity and side effect protein targets of ginsenosides identified by reverse docking
| Protein | Physiological function | Effect of deficiency/inhibition | Docking score |
|---|---|---|---|
| Acetylcholinesterase | Neurotransmission | Cholinergic toxicity | −5.056 |
| Intestinal fatty acid binding protein | Lipid uptake | Altered distribution of lipid profile & adipogenesis | −8.502 |
| Carbonic anhydrase, types II & IV | pH regulation in the blood & kidney | Nephrotoxicity | −5.974 |
| Glutamate dehydrogenase | Amino acid degradation | Nephrotoxicity | −5.406 |
4. Discussion
In this study, potential targets of ginsenosides were identified by reverse docking. Among them, MEK1, EGFR, and Aurora A, which are anticancer targets, were found to interact with ginsenosides with high binding affinity. In addition, thrombin, which is related to blood coagulation, was found to be highly interactive with ginsenosides. In summary, claims of anticancer properties of ginsenosides were in part verified [32] as well as therapeutic effects against heart diseases due to blood-thinning properties [33].
We also identified possible side effects/toxicities of ginsenosides by reverse docking. Acetylcholinesterase inhibition, which can lead to cholinergic toxicity, was found to be a possible effect of ginsenosides. CA II and glutamate dehydrogenase were also shown to bind ginsenosides, which could lead to nephrotoxicities. Intestinal fatty acid-binding protein was also found to be affected by ginsenosides; its inhibition could suppress adipogenesis. However, this effect could sometimes be regarded as therapeutic, especially when applied to obesity.
Further studies, including in vitro and in vivo experiments for the targets identified from our investigation, are warranted to confirm our findings. Such endeavors would not only affirm conventional beliefs about ginsenosides' therapeutic values, but also pave the way to developing new drugs from natural products.
Conflicts of interest
All authors declare no conflicts of interest.
Acknowledgments
This work was supported by National Research Foundation of Korea grants (No. 2013R1A2A2A01067638 and No. 2012M3C1A6035 362). We also thank Schrödinger, LLC, for providing software for our research.
References
- 1.Nocerino E., Amato M., Izzo A.A. The aphrodisiac and adaptogenic properties of ginseng. Fitoterapia. 2000;71:S1–S5. doi: 10.1016/s0367-326x(00)00170-2. [DOI] [PubMed] [Google Scholar]
- 2.Paul N., Kellenberger E., Bret G., Muller P., Rognan D. Recovering the true targets of specific ligands by virtual screening of the protein data bank. Proteins Struct Funct Bioinform. 2004;54:671–680. doi: 10.1002/prot.10625. [DOI] [PubMed] [Google Scholar]
- 3.Kharkar P.S., Warrier S., Gaud R.S. Reverse docking: a powerful tool for drug repositioning and drug rescue. Fut Med Chem. 2014;6:333–342. doi: 10.4155/fmc.13.207. [DOI] [PubMed] [Google Scholar]
- 4.Kitchen D.B., Decornez H., Furr J.R., Bajorath J. Docking and scoring in virtual screening for drug discovery: methods and applications. Nat Rev Drug Discov. 2004;3:935–949. doi: 10.1038/nrd1549. [DOI] [PubMed] [Google Scholar]
- 5.Chong C.R., Sullivan D.J. New uses for old drugs. Nature. 2007;448:645–646. doi: 10.1038/448645a. [DOI] [PubMed] [Google Scholar]
- 6.Harvey A.L. Natural products in drug discovery. Drug Discov Today. 2008;13:894–901. doi: 10.1016/j.drudis.2008.07.004. [DOI] [PubMed] [Google Scholar]
- 7.Shin B.K., Kwon S.W., Park J.H. Chemical diversity of ginseng saponins from Panax ginseng. J Ginseng Res. 2015;39:287–298. doi: 10.1016/j.jgr.2014.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Namba T., Matsushige K., Morita T., Tanaka O. Saponins of plants of Panax species collected in central Nepal and their chemotaxonomical significance. I. Chem Pharm Bull. 1986;34:730–738. doi: 10.1248/cpb.48.889. [DOI] [PubMed] [Google Scholar]
- 9.Sanada S., Kondo N., Shoji J., Tanaka O., Shibata S. Studies on the saponin of ginseng. I. Structures of ginsenoside-Ro, -Rb1, -Rc, and -Rd. Chem Pharm Bull. 1974;22:421–428. [Google Scholar]
- 10.Leung K.W., Wong A.S. Pharmacology of ginsenosides: a literature review. Chin Med. 2010;5:20. doi: 10.1186/1749-8546-5-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chen X., Ung C.Y., Chen Y. Can an in silico drug-target search method be used to probe potential mechanisms of medicinal plant ingredients? Nat Prod Rep. 2003;20:432–444. doi: 10.1039/b303745b. [DOI] [PubMed] [Google Scholar]
- 12.Cai J., Han C., Hu T., Zhang J., Wu D., Wang F., Liu Y., Ding J., Chen K., Yue J. Peptide deformylase is a potential target for anti-Helicobacter pylori drugs: reverse docking, enzymatic assay, and X-ray crystallography validation. Protein Sci. 2006;15:2071–2081. doi: 10.1110/ps.062238406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gao Z., Li H., Zhang H., Liu X., Kang L., Luo X., Zhu W., Chen K., Wang X., Jiang H. PDTD: a web-accessible protein database for drug target identification. BMC Bioinformatics. 2008;9:104. doi: 10.1186/1471-2105-9-104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zahler S., Tietze S., Totzke F., Kubbutat M., Meijer L., Vollmar A.M., Apostolakis J. Inverse in silico screening for identification of kinase inhibitor targets. Chem Biol. 2007;14:1207–1214. doi: 10.1016/j.chembiol.2007.10.010. [DOI] [PubMed] [Google Scholar]
- 15.Chen Y.Z., Ung C.Y. Prediction of potential toxicity and side effect protein targets of a small molecule by a ligand–protein inverse docking approach. J Mol Graph Mod. 2001;20:199–218. doi: 10.1016/s1093-3263(01)00109-7. [DOI] [PubMed] [Google Scholar]
- 16.Kim S., Thiessen P.A., Bolton E.E., Chen J., Fu G., Gindulyte A., Han L., He J., He S., Shoemaker B.A. PubChem Substance and Compound databases. Nucl Acids Res. 2016;44:D1202–D1213. doi: 10.1093/nar/gkv951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Friesner R.A., Banks J.L., Murphy R.B., Halgren T.A., Klicic J.J., Mainz D.T., Repasky M.P., Knoll E.H., Shelley M., Perry J.K. Glide: a new approach for rapid, accurate docking and scoring. 1. Method and assessment of docking accuracy. J Med Chem. 2004;47:1739–1749. doi: 10.1021/jm0306430. [DOI] [PubMed] [Google Scholar]
- 18.Halgren T.A., Murphy R.B., Friesner R.A., Beard H.S., Frye L.L., Pollard W.T., Banks J.L. Glide: a new approach for rapid, accurate docking and scoring. 2. Enrichment factors in database screening. J Med Chem. 2004;47:1750–1759. doi: 10.1021/jm030644s. [DOI] [PubMed] [Google Scholar]
- 19.Choi Y.J., Lee H.J., Kang D.W., Han I.H., Choi B.K., Cho W.H. Ginsenoside Rg3 induces apoptosis in the U87MG human glioblastoma cell line through the MEK signaling pathway and reactive oxygen species. Oncol Rep. 2013;30:1362–1370. doi: 10.3892/or.2013.2555. [DOI] [PubMed] [Google Scholar]
- 20.Sathishkumar N., Karpagam V., Sathiyamoorthy S., Woo M.J., Kim Y.J., Yang D.C. Computer-aided identification of EGFR tyrosine kinase inhibitors using ginsenosides from Panax ginseng. Comput Biol Med. 2013;43:786–797. doi: 10.1016/j.compbiomed.2013.02.020. [DOI] [PubMed] [Google Scholar]
- 21.Zhou Q., Jiang L., Xu C., Luo D., Zeng C., Liu P., Yue M., Liu Y., Hu X., Hu H. Ginsenoside Rg1 inhibits platelet activation and arterial thrombosis. Thromb Res. 2014;133:57–65. doi: 10.1016/j.thromres.2013.10.032. [DOI] [PubMed] [Google Scholar]
- 22.Joo C.N., Kim S.J. Effect of ginseng components (ginsenosides and fat soluble fraction) on rat liver glucokinase activity. Korean Soc Ginseng. 1994;18:1–9. [Google Scholar]
- 23.Davis R.J. The mitogen-activated protein kinase signal transduction pathway. J Biol Chem. 1993;268:14553–14556. [PubMed] [Google Scholar]
- 24.Singh P., Bast F. In silico molecular docking study of natural compounds on wild and mutated epidermal growth factor receptor. Med Chem Res. 2014;23:5074–5085. [Google Scholar]
- 25.Li S., Gao Y., Ma W., Guo W., Zhou G., Cheng T., Liu Y. EGFR signaling-dependent inhibition of glioblastoma growth by ginsenoside Rh2. Tumour Biol. 2014;35:5593–5598. doi: 10.1007/s13277-014-1739-x. [DOI] [PubMed] [Google Scholar]
- 26.Banner D.W., Hadváry P. Crystallographic analysis at 3.0-Å resolution of the binding to human thrombin of four active site-directed inhibitors. J Biol Chem. 1991;266:20085–20093. [PubMed] [Google Scholar]
- 27.Lo Iacono M., Monica V., Saviozzi S., Ceppi P., Bracco E., Papotti M., Scagliotti G.V. Aurora kinase A expression is associated with lung cancer histological-subtypes and with tumor de-differentiation. J Transl Med. 2011;9:100. doi: 10.1186/1479-5876-9-100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Umene K., Banno K., Kisu I., Yanokura M., Nogami Y., Tsuji K., Masuda K., Ueki A., Kobayashi Y., Yamagami W. Aurora kinase inhibitors: potential molecular targeted drugs for gynecologic malignant tumors (review) Biomed Rep. 2013;1:335–340. doi: 10.3892/br.2013.91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Dodds H.M., Rivory L.P. The mechanism for the inhibition of acetylcholinesterases by irinotecan (CPT-11) Mol Pharm. 1999;56:1346–1353. doi: 10.1124/mol.56.6.1346. [DOI] [PubMed] [Google Scholar]
- 30.Zhang B., MacNaul K., Szalkowski D., Li Z., Berger J., Moller D.E. Inhibition of adipocyte differentiation by HIV protease inhibitors. J Clin Endocrinol Metab. 1999;84:4274–4277. doi: 10.1210/jcem.84.11.6234. [DOI] [PubMed] [Google Scholar]
- 31.Bryła J., Lietz T., Jarzyna R., Michalik M., Pietkiewicz J. Differential in vivo and in vitro effect of gentamicin on glutamate synthesis and glutamate deamination in rabbit kidney-cortex tubules and mitochondria. Pharmacol Res. 1992;26:367–375. doi: 10.1016/1043-6618(92)90235-4. [DOI] [PubMed] [Google Scholar]
- 32.Nag S.A., Qin J.J., Wang W., Wang M.H., Wang H., Zhang R. Ginsenosides as anticancer agents: in vitro and in vivo activities, structure–activity relationships, and molecular mechanisms of action. Front Pharm. 2012;3:25. doi: 10.3389/fphar.2012.00025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kim J.H. Cardiovascular diseases and Panax ginseng: a review on molecular mechanisms and medical applications. J Ginseng Res. 2012;36:16–26. doi: 10.5142/jgr.2012.36.1.16. [DOI] [PMC free article] [PubMed] [Google Scholar]