Abstract
On the basis of the recently recognized potential of bone marrow stem cells to give rise to hepatocytes, we here investigated the role of G-CSF priming PBMCs played in the liver of cirrhotic rats. The animal model of liver cirrhosis was induced by injecting CCl4 in SD rats, and G-CSF was administered in hematopoietic stem cell mobilization doses. After the liver cirrhosis model was established, the female cirrhotic rats were divided into two groups. Group I only received G-CSF mobilization, group II received G-CSF mobilized PBMCs transplanted from the male cirrhotic rats. PKH26 staining and sex-determining region for the Y-chromosome gene were used to trace the transplanted cells. Liver function related factors were assayed under the animal automatic biochemistry analyzer, and the liver pathological changes were evaluated by HE staining. The comparative liver functions of the two groups were investigated by analysis of two sample t-tests. A P value of <0.05 was considered as significant in all analyses. Our results showed that the transplanted PBMCs could locate in the livers of the female rats. In addition, compared with the group I, rats in group II displayed significant liver improvement in serum ALB, ALT, AST and TBIL (p<0.05). However, the semi-quantitative classification of the liver pathological changes in both groups did not indicate a significant difference (p>0.05). The results indicated that mobilized PBMC transplant could contribute to liver function in cirrhotic livers, which might be an alternative therapy for liver cirrhosis.
Keywords: peripheral blood monocytes, transplant, granulocyte colony stimulating factor, liver cirrhosis
Introduction
Liver cirrhosis is one of the most common liver diseases in China (Liu and Fan, 2007[16]) . Currently, liver transplantation is the only effective therapy. However, its widespread use is still limited by the following factors: lack of donor organs, risk of rejection, operative damage, and high costs. Therefore, it is very important to find an alternative therapy for liver cirrhosis.
Recent evidence shows that bone marrow (BM) derived stem cells could be converted into hepatocyte-like cells in vitro (Ruhnke et al., 2005[18]; Yan et al., 2007[25]; Khurana and Mukhopadhyay, 2008[11]). In vivo, several reports demonstrated that BM derived stem cells might accelerate the recovery and improvement of liver function in the animal model of liver injury (Lagasse et al., 2000[15]; Körbling et al., 2002[14]; Wang et al., 2003[23]) . Moreover, several recent clinical trials showed G-CSF mobilization or G-CSF mobilized bone marrow-derived hematopoietic stem cells could contribute to liver function in cirrhotic livers (Terai et al., 2006[21]; Yannaki et al., 2006[26]; am Esch et al., 2005[2]; Gordon et al., 2006[6]; Gaia et al., 2006[5]). Taken together, these findings suggest that G-CSF mobilization or G-CSF priming of peripheral blood monocytes transplantation could contribute to liver function in cirrhotic livers.
In previous studies we have demonstrated that PBMCs from patients with decompensated liver cirrhosis can differentiate into functional hepatocytes both in vitro and in vivo (Yan et al., 2007[25], 2008[24]). In addition, simple randomized clinical trials also demonstrated that autologous PBMC transplantation played one important role for decompensated liver cirrhosis (Han et al., 2008[7]). Although the preliminary results seemed to be favorable, liver biopsy was not undertaken in these patients and its potential mechanism was still unclear.
In this study, we established an animal model of liver cirrhosis, and then transplanted the mobilized PBMCs into the cirrhotic rats to observe both the kinetics of liver function related factors and the changes in liver histopathology. Our results showed that GSF priming PBMCs could improve the liver function of the cirrhotic rats significantly, but similar changes in liver histopathology were observed between the G-CSF mobilization therapy and G-CSF mobilized PBMC transplantation therapy. This study indicated that mobilized PBMC transplantation might be an alternative therapy for liver cirrhosis.
Materials and Methods
CCl4-induced liver cirrhosis model
Female and male SD rats were 6 weeks old, weighing between 135 and 150 g. Rats were bred and maintained in an air-conditioned facility with specific pathogen-free conditions, and were subjected to a 12:12-h day light/darkness and allowed unlimited access to food and water. Morphological and behavioral changes of the rats were monitored. The female or male liver cirrhosis model was induced by CCl4 injected intraperitoneally at a dose of 0.3 mL/100 g body weight of 60 mL/L CCl4 (Sigma, St. Louis, USA) dissolved in an equal volume of castor oil (Sigma, St. Louis, USA). The injection was given twice a week for 16 weeks to establish liver fibrosis, and were subjected to the corresponding therapy only when animals displayed stable liver cirrhosis four weeks after the discontinuation of carbon tetrachloride (Nussler et al., 2006[17]). The same volume of castor oil alone was used as a control. Liver cirrhosis was determined by histopathological examination.
G-CSF mobilization and PBMC administration
After the decompensated liver cirrhosis model was established, the female cirrhotic rats were divided into two groups. Group I only received G-CSF mobilization, with recombinant human granulocyte colony stimulating factor (rhG-CSF, Qi Lu Pharmaceutical Co, Ltd., China) at 5 µg/kg/d administered subcutaneously daily for 4 days as a control (Gaia et al., 2006[5]); group II received sex-matched mobilized peripheral blood monocyte transplantation. PBMCs were injected intravenously into the rats' tails after suspension in phosphate buffer saline. All animal experiments received approval from the institutional animal care committee.
Detection of PKH26 labeled PBMCs
Liver tissue was analyzed two weeks after injection of PBMCs. Five-micrometer frozen sections were made and then the sections were placed directly under a fluorescence microscope. Green fluorescence was chosen to emit the section.
Primer in situ labeling for SRY gene
Liver tissues of female rats were collected for detecting the male sex-determining Region Y (SRY) gene at week two of the transplantation, and primed in situ labeling (PRINS) was performed according to the previous reports (Koch et al., 1989[13], 1995[12]), with Dig-11-dUTP used as the report molecules. Primers of SRY were designed according to nucleotide sequences of the human SRY. The oligonucleotide primers used were:
Forward: 5'-AGGCGCAAGTTGGCTCAAC-3'
Reverse: 5'-GGCCTTTTTTCGGCTTCTG-3' (101bp).
Examination of the level of liver function related markers
Venous blood of rats from different groups was collected from the retro-orbital vein at week twenty after CCL4-induction, also at week two, and two months after corresponding treatment. Liver function related factors including ALT, AST, TBIL, ALB, and CHO of the rats at different points were monitored by an animal automatic biochemistry analyzer.
Analysis of liver histopathology
All the rats were terminated with CO2 narcosis, and liver tissue was harvested for analysis. Liver samples were collected into PBS and fixed overnight in 40 g/L paraformaldehyde in PBS at 4 °C. Serial 5-μm sections of the right lobes of the livers were stained with hematoxylin and eosin (HE) and were examined histopathologically. In accordance with the grading and staging criteria of chronic liver pathology (Jonker et al., 1990[9], 1992[8]), we adopted the semi-quantitative method to evaluate the inflammatory, fatty degeneration, and necrosis of the hepatocytes for rats of different groups. Score criteria are the following: score 0 means no fatty degeneration, and necrosis, score 1 means mild fatty degeneration and necrosis, score 2 means moderate fatty degeneration and necrosis, and score 3 means severe fatty degeneration and necrosis. The criteria of the liver fibrosis are the following: level 0 means normal liver tissue, level 1 means increased quantity of collagen fibers but no compartment development, level 2 means no complete fiber compartment development, level 3 means thinner of complete compartment development, and level 4 means thickening of complete compartment development.
Statistical analysis
Data are expressed as mean ± SD. The comparative liver functions of the two groups were investigated by analysis of two sample t-tests. A p value of <0.05 was considered as significant in all analysis. Statistical analysis was done using the SPSS version 13.0 software.
Results
Characterization of cell origins
The mobilized PBMCs from the cirrhotic rats were identified by markers specific for monocytes (CD14), HSCs (CD34, and CD133). The results showed that the percentage of CD34, CD133 expressed in the cells were about 20.1 %, 68.9 % respectively, whereas the percentage of CD14 expressed in the PBMCs was about 89.4 %. Thus, we called the isolated cells in our experiment PBMCs.
The PBMCs staining
With PKH26-GL, the isolated PBMCs from the male cirrhotic rats were labeled with 10 µM PKH26-GL. The percentage of labeled cells was more than 90 %, and the labeled PBMCs were shown as red under a fluorescence microscope (Figure 1A(Fig. 1)). After the labeled PBMCs were transplanted into the liver of female cirrhotic rats one month later, sporadic cells with red fluorescence among the hepatocytes were observed (Figure 1B(Fig. 1)), suggesting that PKH26-GL-labeled PBMCs migrated into the liver of female rats.
Figure 1. PKH26 stained PMBCs in vitro and in vivo (×100).
A: PKH26 stained PMBCs in vitro;
B: PKH26 stained PMBCs scattered in the cirrhotic liver of SD rats with red fluorescence
The result of PRINS on SRY gene
The PRINS has been conducted successfully to identify the rat SRY gene in the liver tissues of female cirrhotic rats. As is shown in Figure 2A(Fig. 2), the rat SRY gene could be detected in the livers of the female rats that accepted the transplantation of PBMCs from the male, but could not be detected in the livers of the female rats that only accepted G-CSF mobilization (Figure 2B(Fig. 2)). This result indicated that the transplanted PBMCs could be located in the liver of the female cirrhotic rats.
Figure 2. sry gene was identified in the cirrhotic liver of the female rats (×100).
A: sry gene was detected in the cirrhotic liver of the female rats that underwent the transplantation of PBMCs from the male cirrhosis;
B: sry gene was not detected in the cirrhotic liver of the female rats that didn't undergo the transplantation of PBMCs from female rats.
Kinetics of the liver function in cirrhotic rats
The liver functions of the rats treated with CCL4 showed significant damage on the 16th week. After the corresponding treatment, rats in both group I and group II displayed significant liver function improvement. Moreover, compared with those in group I, rats in group II displayed significant liver improvement as shown in Table 1(Tab. 1).
Table 1. Kinectics of liver function of the cirrhotic in different groups (n=6).
Liver histopathological changes of the cirrhotic rats
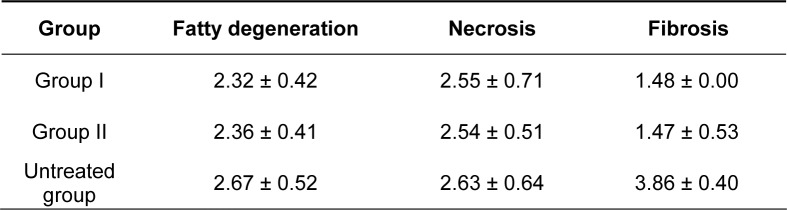
After two months of PBMC transplantation, the rats were terminated and the pathological results indicated that the changes of fibrosis in rats from group I and group II were similar (Figures 3A(Fig. 3), 3B(Fig. 3)), significantly improved over the untreated cirrhotic rats (Figure 3C(Fig. 3)). According to the grading and staging criteria of chronic liver pathology, we adopted the semi-quantitative method to evaluate the inflammatory, fatty degeneration, and necrosis of the hepatocytes. As is shown in Table 2(Tab. 2), no significant difference was observed between the two groups (p>0.05).
Figure 3. Histopathological changes of the cirrhotic liver of the rats after different treatment (×200).
A: Histopathological changes of liver of the cirrhotic rats that only accepted G-CSF mobilization therapy
B: Histopathological changes of liver of the cirrhotic rats that accepted G-CSF mobilization priming PBMCs transplant therapy
C: Histopathological changes of liver of the cirrhotic rats that didn't accept any therapy
Table 2. Pathological score of the cirrhotic rats in different groups after corresponding therapy (n=6, x̅ ± s).
Discussion
A variety of chronic injuries of the liver caused by viral hepatitis, alcohol abuse, drugs, autoimmune attack and metabolic diseases can lead to liver cirrhosis. In human and animal models bone marrow-derived hepatocytes have been identified in liver biopsies after sex-mismatched bone marrow transplantations (Theise et al., 2000[22]; Alison et al., 2000[1]). Yet, our study showed the following: the cells infused into the cirrhotic rats were mobilized PBMCs, and the kinetics of liver functions of the cirrhotic rats were analyzed both in G-CSF mobilization group and G-CSF primed PBMC transplantation group. In addition, liver biopsy was made to compare the liver histopathological changes between the G-CSF mobilization and the G-CSF primed PBMC transplantation group.
In recent years, clinical studies investigating the efficacy of potential therapies of bone marrow-derived stem cells were initiated, including cardiopathy (Schächinger et al., 2006[19]), diabetic disease (Kawamura et al., 2006[10]), system lupus erythema (Statkute et al., 2005[20]), inflammatory bowel diseases (Ditschkowski et al., 2003[4]), liver and pancreatic disease (Bengala et al., 2005[3]). In our department, we have carried out PBMC transplantation to treat decompensated liver cirrhosis, and obtained favorable results (Yannaki et al., 2005[27]) . To explain the lasting clinical amelioration of the cirrhosis, we established the animal model of liver cirrhosis, and then infused the mobilized PBMCs from the male cirrhotic rats into the female to observe the fate of the transplanted cells and their function. Results of cell tracing techniques including PKH26 staining, and PRINS (primed in situ labeling analysis) of the rat Y chromosome showed that the transplanted PBMCs could migrate into the cirrhotic liver and then locate in the periportal vein through blood circulation. Moreover, this study showed the mobilized PBMC transplantation could improve liver function in cirrhotic rats significantly more than those only receiving G-CSF mobilization therapy. However, similar histopathological changes were observed in the two groups after the corresponding treatment, which is consistent with the previous report. That the mobilized PBMCs could reverse liver cirrhosis to a certain extent might be explained by the following: G-CSF mobilized PBMCs secreted some chemokines, which might promote the transplanted PBMCs located in the injured liver; the mobilized PMBCs also secreted some cytokines, which promoted the proliferation of the transplanted cells or hepatic stem cells, and enhanced apoptosis of stellate cell of liver or the degeneration of the hepatic fibrous tissues.
In conclusion, this study indicated that G-CSF mobilized PBMC transplantation could improve liver function in cirrhotic rats significantly. However, in reversing liver histopathology, the G-CSF mobilization therapy showed a similar ability to reverse the liver histopathology with the G-CSF mobilized PBMC transplantation. This finding suggests that G-CSF mobilized PBMCs transplant might be considered as another future protocol for liver cirrhosis treatment.
Notes
Li Yan and Shiping Xu contributed equally as first authors.
Acknowledgements
This study was supported by grants from Chinese National Foundation of National Sciences (30900669)
References
- 1.Alison MR, Poulsom R, Jeffery R, Dhillon AP, Quaglia A, Jacob J, Novelli M, Prentice G, Williamson J, Wright NA. Hepatocytes from non-hepatic adult stem cells. Nature. 2000;406:257. doi: 10.1038/35018642. [DOI] [PubMed] [Google Scholar]
- 2.am Esch JS, 2nd, Knoefel WT, Klein M, Ghodsizad A, Fuerst G, Poll LW, Piechaczek C, Burchardt ER, Feifel N, Stoldt V, Stockschläder M, Stoecklein N, Tustas RY, Eisenberger CF, Peiper M, Häussinger D, Hosch SB. Portal application of autologous CD133+ bone marrow cells to the liver: a novel concept to support hepatic regeneration. Stem Cells. 2005;23:463–470. doi: 10.1634/stemcells.2004-0283. [DOI] [PubMed] [Google Scholar]
- 3.Bengala C, Guarneri V, Giovannetti E, Lencioni M, Fontana E, Mey V, Fontana A, Boggi U, Del Chiaro M, Danesi R, Ricci S, Mosca F, Del Tacca M, Conte PF. Prolonged fixed dose rate infusion of gemcitabine with autologous haemopoietic support in advanced pancreatic adenocarcinoma. Br J Cancer. 2005;93:35–40. doi: 10.1038/sj.bjc.6602673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ditschkowski M, Einsele H, Schwerdtfeger R, Bunjes D, Trenschel R, Beelen DW, Elmaagacli AH. Improvement of inflammatory bowel disease after allogeneic stem-cell transplantation. Transplantation. 2003;75:1745–1747. doi: 10.1097/01.TP.0000062540.29757.E9. [DOI] [PubMed] [Google Scholar]
- 5.Gaia S, Smedile A, Omedè P, Olivero A, Sanavio F, Balzola F, Ottobrelli A, Abate ML, Marzano A, Rizzetto M, Tarella C. Feasibility and safety of G-CSF administration to induce bone marrow-derived cells mobilization in patients with end stage liver disease. J Hepatol. 2006;45:13–19. doi: 10.1016/j.jhep.2006.02.018. [DOI] [PubMed] [Google Scholar]
- 6.Gordon MY, Levicar N, Pai M, Bachellier P, Dimarakis I, Al-Allaf F, M'Hamdi H, Thalji T, Welsh JP, Marley SB, Davies J, Dazzi F, Marelli-Berg F, Tait P, Playford R, Jiao L, Jensen S, Nicholls JP, Ayav A, Nohandani M, Farzaneh F, Gaken J, Dodge R, Alison M, Apperley JF, Lechler R, Habib NA. Characterization and clinical application of human CD34+ stem/progenitor cell populations mobilized into the blood by granulocyte colony-stimulating factor. Stem Cells. 2006;24:1822–1830. doi: 10.1634/stemcells.2005-0629. [DOI] [PubMed] [Google Scholar]
- 7.Han Y, Yan L, Han G, Zhou X, Hong L, Yin Z, Zhang X, Wang S, Wang J, Sun A, Liu Z, Xie H, Wu K, Ding J, Fan D. Controlled trials in hepatitis B virus-related decompensate liver cirrhosis: peripheral blood monocyte transplant versus granulocyte-colony-stimulating factor mobilization therapy. Cytotherapy. 2008;10:390–396. doi: 10.1080/14653240802129901. [DOI] [PubMed] [Google Scholar]
- 8.Jonker AM, Dijkhuis FW, Boes A, Hardonk MJ, Grond J. Immunohistochemical study of extracellular matrix in acute galactosamine hepatitis in rats. Hepatology. 1992;15:423–431. doi: 10.1002/hep.1840150312. [DOI] [PubMed] [Google Scholar]
- 9.Jonker AM, Dijkhuis FW, Kroese FG, Hardonk MJ, Grond J. Immunopathology of acute galactosamine hepatitis in rats. Hepatology. 1990;11:622–627. doi: 10.1002/hep.1840110415. [DOI] [PubMed] [Google Scholar]
- 10.Kawamura A, Horie T, Tsuda I, Abe Y, Yamada M, Egawa H, Iida J, Sakata H, Onodera K, Tamaki T, Furui H, Kukita K, Meguro J, Yonekawa M, Tanaka S. Clinical study of therapeutic angiogenesis by autologous peripheral blood stem cell (PBSC) transplantation in 92 patients with critically ischemic limbs. J Artif Organs. 2006;9:226–233. doi: 10.1007/s10047-006-0351-2. [DOI] [PubMed] [Google Scholar]
- 11.Khurana S, Mukhopadhyay A. In vitro transdifferentiation of adult hematopoietic stem cells: an alternative source of engraftable hepatocytes. J Hepatol. 2008;49:998–1007. doi: 10.1016/j.jhep.2008.05.019. [DOI] [PubMed] [Google Scholar]
- 12.Koch J, Hindkjaer J, Kølvraa S, Bolund L. Construction of a panel of chromosome-specific oligonucleotide probes (PRINS-primers) useful for the identification of individual human chromosomes in situ. Cytogenet Cell Genet. 1995;71:142–147. doi: 10.1159/000134094. [DOI] [PubMed] [Google Scholar]
- 13.Koch JE, Kølvraa S, Petersen KB, Gregersen N, Bolund L. Oligonucleotide-priming methods for the chromosome-specific labelling of alpha satellite DNA in situ. Chromosoma. 1989;98:259–265. doi: 10.1007/BF00327311. [DOI] [PubMed] [Google Scholar]
- 14.Körbling M, Katz RL, Khanna A, Ruifrok AC, Rondon G, Albitar M, Champlin RE, Estrov Z. Hepatocytes and epithelial cells of donor origin in recipients of peripheral-blood stem cells. N Engl J Med. 2002;346:738–746. doi: 10.1056/NEJMoa3461002. [DOI] [PubMed] [Google Scholar]
- 15.Lagasse E, Connors H, Al-Dhalimy M, Reitsma M, Dohse M, Osborne L, Wang X, Finegold M, Weissman IL, Grompe M. Purified hematopoietic stem cells can differentiate into hepatocytes in vivo. Nat Med. 2000;6:1229–1234. doi: 10.1038/81326. [DOI] [PubMed] [Google Scholar]
- 16.Liu J, Fan D. Hepatitis B in China. Lancet. 2007;369:1582–1583. doi: 10.1016/S0140-6736(07)60723-5. [DOI] [PubMed] [Google Scholar]
- 17.Nussler A, Konig S, Ott M, Sokal E, Christ B, Thasler W, Brulport M, Gabelein G, Schormann W, Schulze M, Ellis E, Kraemer M, Nocken F, Fleig W, Manns M, Strom SC, Hengstler JG. Present status and perspectives of cell-based therapies for liver diseases. J Hepatol. 2006;45:144–159. doi: 10.1016/j.jhep.2006.04.002. [DOI] [PubMed] [Google Scholar]
- 18.Ruhnke M, Ungefroren H, Nussler A, Martin F, Brulport M, Schormann W, Hengstler JG, Klapper W, Ulrichs K, Hutchinson JA, Soria B, Parwaresch RM, Heeckt P, Kremer B, Fändrich F. Differentiation of in vitro-modified human peripheral blood monocytes into hepatocyte-like and pancreatic islet-like cells. Gastroenterology. 2005;128:1774–1786. doi: 10.1053/j.gastro.2005.03.029. [DOI] [PubMed] [Google Scholar]
- 19.Schächinger V, Erbs S, Elsässer A. Intracoronary bone marrow-derived progenitor cells in acute myocardial infarction. N Engl J Med. 2006;355:1210–1211. doi: 10.1056/NEJMoa060186. [DOI] [PubMed] [Google Scholar]
- 20.Statkute L, Traynor A, Oyama Y, Yaung K, Verda L, Krosnjar N, Burt RK. Antiphospholipid syndrome in patients with systemic lupus erythematosus treated by autologous hematopoietic stem cell transplantation. Blood. 2005;106:2700–9. doi: 10.1182/blood-2005-01-0330. [DOI] [PubMed] [Google Scholar]
- 21.Terai S, Ishikawa T, Omori K, Aoyama K, Marumoto Y, Urata Y, Yokoyama Y, Uchida K, Yamasaki T, Fujii Y, Okita K, Sakaida I. Improved liver function in patients with liver cirrhosis after autologous bone marrow cell infusion therapy. Stem Cells. 2006;24:2292–2298. doi: 10.1634/stemcells.2005-0542. [DOI] [PubMed] [Google Scholar]
- 22.Theise ND, Nimmakayalu M, Gardner R, Illei PB, Morgan G, Teperman L, Henegariu O, Krause DS. Liver from bone marrow in humans. Hepatology. 2000;32:11–6. doi: 10.1053/jhep.2000.9124. [DOI] [PubMed] [Google Scholar]
- 23.Wang X, Ge S, McNamara G, Hao QL, Crooks GM, Nolta JA. Albumin-expressing hepatocyte-like cells develop in the livers of immune-deficient mice that received transplants of highly purified human hematopoietic stem cells. Blood. 2003;101:4201–4208. doi: 10.1182/blood-2002-05-1338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yan L, Han Y, Wang J, Liu J, He Y, Wang H, Fan D. Peripheral blood monocytes from the decompensated liver cirrhosis could migrate into nude mouse liver with human hepatocyte-markers expression. Biochem Biophys Res Commun. 2008;371:635–638. doi: 10.1016/j.bbrc.2008.04.058. [DOI] [PubMed] [Google Scholar]
- 25.Yan L, Han Y, Wang J, Liu J, Hong L, Fan D. Peripheral blood monocytes from patients with HBV related decompensated liver cirrhosis can differentiate into functional hepatocytes. Am J Hematol. 2007;82:949–954. doi: 10.1002/ajh.21030. [DOI] [PubMed] [Google Scholar]
- 26.Yannaki E, Anagnostopoulos A, Kapetanos D, Xagorari A, Iordanidis F, Batsis I, Kaloyannidis P, Athanasiou E, Dourvas G, Kitis G, Fassas A. Lasting amelioration in the clinical course of decompensated alcoholic cirrhosis with boost infusions of mobilized peripheral blood stem cells. Exp Hematol. 2006;34:1583–1587. doi: 10.1016/j.exphem.2006.06.012. [DOI] [PubMed] [Google Scholar]
- 27.Yannaki E, Athanasiou E, Xagorari A, Constantinou V, Batsis I, Kaloyannidis P, Proya E, Anagnostopoulos A, Fassas A. G-CSF-primed hematopoietic stem cells or G-CSF per se accelerate recovery and improve survival after liver injury, predominantly by promoting endogenous repair programs. Exp Hematol. 2005;33:108–119. doi: 10.1016/j.exphem.2004.09.005. [DOI] [PubMed] [Google Scholar]