Abstract
Three mechanistically different sample extraction methodologies, namely, silica spin columns, phenol-chloroform, and an automated magnetic capture of polymer-complexed DNA (via an Automate Express instrument), were compared for their abilities to purify nucleic acids from blood culture fluids for use in TaqMan assays for detection of Staphylococcus aureus. The extracts from silica columns required 100- to 1000-fold dilutions to sufficiently reduce the powerful PCR inhibitory effects of the anticoagulant sodium polyanetholsulfonate, a common additive in blood culture media. In contrast, samples extracted by either phenol-chloroform or the Automate Express instrument required little or no dilution, respectively, allowing for an approximate 100-fold improvement in assay sensitivity. Analysis of 60 blood culture bottles indicated that these latter two methodologies could be used to detect lower numbers of pathogens and that a growing S. aureus culture could be detected 2 hours earlier than when using silica columns. Of the three tested methodologies, the Automate Express instrument had the shortest time to result, requiring only approximately 80 minutes to process 12 samples. These findings highlight the importance of considering the mechanism when selecting a DNA extraction methodology, given that certain PCR inhibitors act in a similar fashion to DNA in certain chemical environments, resulting in copurification, whereas other methodologies use different chemistries that have advantages during the DNA purification of certain types of samples.
Molecular analysis of blood cultures is increasingly being used to identify blood-borne pathogens. Although it is considered the gold standard, culturing blood can generate false-negative results up to 31% of the time and false-positive results up to 10% of the time,1, 2, 3, 4 making confirmation by PCR desirable. Achieving accurate PCR results from blood culture samples is difficult, however, because of the presence of sodium polyanetholsulfonate (SPS), a powerful PCR inhibitor that is added routinely to blood culture media to i) lower the barrier for bacterial growth by inhibiting the complement pathway, ii) prevent blood coagulation, and iii) interfere with the activity of some antibiotics.5, 6, 7, 8, 9, 10, 11 In addition to SPS, several blood components (including heme, hematin, hemoglobin, lactoferrin, and IgG) can interfere with PCR, as do the anticoagulants used during blood collection, such as heparin and EDTA.12, 13, 14, 15, 16, 17, 18, 19, 20 These inhibitors must be sufficiently removed from a blood culture sample to minimize the potential for false-negative PCR results.
Other studies have examined the issue of detecting bacteria in blood cultures,9, 21, 22, 23 but there has been no previous formal study that compares three mechanistically different forms of nucleic acid extraction in their ability to remove PCR inhibitors found in clinical blood culture fluids and addresses whether the choice of methodology can reduce the time to result (TTR). To date, the most successful methodology to remove SPS from spiked blood cultures relies on an organic extraction procedure that is time consuming and labor intensive, requires the use of a fume hood, and is not amenable to automation.9
For the present study, the phenol-chloroform method24 was included as representing the gold standard, and silica-based spin columns25 were included as the most commonly used sample extraction methodology at present (as a fast, convenient, affordable, and high-performing approach). Both technologies have their drawbacks, however. Phenol-chloroform utilizes hazardous organic solutions that require the use of a fume hood and is labor intensive, and silica-based extraction methodologies, either in spin column or magnetic bead format, can result in the carryover of PCR inhibitors into the extracts.26, 27 To complete the comparison, an automated platform that uses hydrophilic polymer-embedded magnetic particles to purify DNA was included as a third methodology. This third methodology purifies DNA through a nondirect binding-based mechanism that is decidedly different from both phenol-chloroform and silica-based purification. In this nondirect mechanism, DNA is complexed with a high molecular weight (HMW) polysaccharide in solution, which is captured on the surface of ferric oxide particles that can be removed from the liquid phase through the use of a magnet.
HMW polysaccharide-based purification has proven to be particularly useful in forensics applications, where PCR inhibitors commonly encountered,28 but this newer technology has not yet been tested on blood cultures that contain SPS. After several analytical experiments designed to examine the utility of these extraction methodologies in removing SPS and other inhibitors found in culture media, the three methodologies were compared for their ability to purify DNA from 60 blood cultures for accurate TaqMan analysis and to determine whether any of the methodologies offers an advantage in reducing TTR. The target organisms for the present study were methicillin-resistant Staphylococcus aureus (MRSA) and methicillin-sensitive S. aureus (MSSA). These pathogens are responsible for approximately 756,000 hospitalizations and >18,650 deaths per year, at a cost of approximately $26.8 billion to the U.S. health care system.29, 30, 31 Improving the accuracy and speed of detecting these pathogens is extremely important, given that an improper or delayed diagnosis can result in increased morbidity and mortality for infected individuals.
Materials and Methods
Laboratory Culture
A frozen glycerol stock of methicillin-sensitive S. aureus from the American Type Culture Collection (12600; ATCC, Manassas, VA) was plated onto a brain heart infusion agar plate (B1010; Teknova, Hollister, CA), and a single colony was selected and inoculated into brain heart infusion broth (B9993; Teknova) for overnight aerobic incubation at 37°C.
The colony forming units (CFU) per milliliter of the S. aureus culture used in these experiments was determined by creating a 10-fold dilution series in brain heart infusion broth that was spread onto brain heart infusion plates. Colonies were counted after 48 hours at 37°C. The count from the plate yielding between 10 and 100 colonies was used to back-calculate the CFU/mL of the culture.
Laboratory Samples
Blood from one male donor was collected by a phlebotomist at the Serological Research Institute (Richmond, CA) into a plastic BD Vacutainer blood collection tube spray-coated with K2-EDTA (366643; Becton Dickinson, Franklin Lakes, NJ). Polyanetholsulfonic acid sodium salt, also referred to as sodium polyanetholsulfonate (SPS) (P2008; Sigma-Aldrich, St. Louis, MO), was dissolved in water to 20 μg/μL and eleven 1.3-fold serial dilutions were made, down to 1.1 μg/μL. Ten-microliter volumes of the dilution series were added to 40-μL blood samples, so that 11.2 to 153.8 μg SPS was contained in each sample.
BACTEC Plus Aerobic/F medium (442192; Becton Dickinson) containing blood at 3:1 v/v was spiked with 10-fold dilutions of S. aureus broth culture to generate samples ranging from 8 × 102 to 8 × 108 CFU/mL. The BACTEC Plus Aerobic/F medium contains 0.05% SPS, or 18.8 μg SPS per 50-μL blood culture sample.
BACTEC Plus Aerobic/F medium containing blood at 3:1 v/v was spiked with S. aureus to 13,000 CFU/mL. From this culture, samples were drawn in volumes of 1, 2, 4, 8, 16, 32, 64, 128, and 256 μL. To standardize sample volumes for extraction, samples with volumes of ≤32 μL were taken up to 50 μL with BACTEC Plus Aerobic/F medium (P5368; Sigma-Aldrich); samples with volumes of ≥64 μL were centrifuged at 11,000 × g for 1 minute, and the supernatant was removed by aspiration until just 50 μL remained.
BACTEC Plus Aerobic/F medium containing blood at 3:1 v/v was also spiked with S. aureus to 2060 CFU/mL. Fifty-microliter samples were drawn from this culture before the spike and immediately after the spike. The culture was then incubated at 37°C under shaking aerobic conditions, and 50-μL samples were drawn every hour for 9 hours.
The samples relevant to each experiment were processed on different days, a period sometimes spanning several weeks. To preserve sample uniformity across extended time frames, all sample aliquots for a particular experiment were frozen at −80°C immediately after their creation and were thawed only just before processing. Freezing all of the samples before analysis eliminated the potential that one sample set for a particular methodology might have an altered DNA integrity if stored at 4°C for an extended period of time before processing, which would bias the findings of the study.
Clinical Samples
Sixty remnant clinical samples collected into resin-containing aerobic or lytic anaerobic BACTEC 9240 blood culture bottles were used for the present study (442260, 442265; Becton Dickinson). All samples were collected under Institutional Review Board-approved guidelines, and all patient identifiers were removed from these samples before testing. No linkage existed between these deidentified samples and patient personal health information.
Bacterial isolates were identified by standard laboratory protocol using a MicroScan WalkAway instrument (Siemens Healthcare Diagnostics, Deerfield, IL) with panels and/or Staphaurex (R30859902; Remel, Lenexa, KS). Bacteria were grown on tryptic soy agar plates with 5% sheep blood (R01198; Remel) to obtain purified colonies and then were Gram-stained (12525, 12542, 12557, and 12531; Becton Dickinson). Those appearing Gram-positive and resembling cocci in clusters under microscopic examination were tested for catalase activity using a 3% solution of H2O2 (H324-500; Fisher Scientific, Pittsburgh, PA). Catalase-positive colonies were further analyzed for their coagulase reaction using Staphaurex (R4661007; Remel) and for oxacillin resistance using a MicroScan Gram-positive panel (R4661007; Siemens). Coagulase-positive, catalase-positive, Gram-positive cocci in clusters were identified as S. aureus. Aliquots of fluids from these deidentified blood culture bottles were frozen at −80°C. A total of 30 S. aureus-positive cultures (12 MRSA and 18 MSSA) and 30 S. aureus-negative cultures were shipped frozen on dry ice, in a blinded fashion, to Life Technologies for nucleic acid extraction and TaqMan analysis. The 30 S. aureus-negative culture fluids consisted of culture-negative fluids (n = 20), coagulase-negative Staphylococcus species (n = 3), Streptococcus pneumoniae (n = 2), β-hemolytic Group G Streptococcus species (n = 1), Salmonella species (n = 2), Citrobacter species (n = 1), and Candida albicans (n = 1).
Sample Extraction Protocols
On separate days, three replicates from each of the sample aliquots were thawed and processed using either the DNeasy blood and tissue kit (69506; Qiagen, Hilden, Germany), phenol-chloroform (15593031; Invitrogen, Carlsbad, CA), or an ABI Automate Express forensic DNA extraction system (4441763; Applied Biosystems, Foster City, CA).
DNeasy Blood and Tissue Kit
The S. aureus spiked samples and the deidentified clinical blood culture samples were pretreated with lysozyme for 30 minutes at 37°C. Lysozyme pretreatment involved adding 180 μL of an enzymatic lysis buffer to each 50-μL sample, which consisted of lysozyme (20 mg/mL, ≥40,000 U/mg; L6876; Sigma-Aldrich) dissolved in 20 mmol/L Tris-HCl, pH 8.0 (AM9855G; Ambion-Applied Biosystems, Austin, TX), 2 mmol/L Na2 EDTA (AM9260G; Ambion), and 1.2% Triton X-100 (T8787; Sigma-Aldrich), which equates to 3.6 mg lysozyme/50-μL sample. After the 37°C incubation, 25 μL of kit-provided proteinase K solution in 200 μL AL buffer was added to the samples and incubated for ≥10 minutes at 56°C. The samples were passed through the silica spin columns, washed, and dried. To normalize the volume of the extracts across the three sample preparation methodologies, silica columns were eluted using 50 μL of AE buffer, which is also the manufacturer's recommended elution volume for samples expected to contain <1 μg of DNA. The lysozyme pretreatment step was excluded for the SPS spiking experiments, because these experiments targeted a human gene and not bacteria (lysozyme degrades the cell wall of Gram-positive bacteria).32
Phenol-Chloroform Extraction
Both the S. aureus-spiked samples and the clinical blood culture samples were digested using the same lysozyme solution described for the samples processed using the DNeasy blood and tissue kit. After lysozyme pretreatment, 200 μg proteinase K (20 mg/mL) (AM2546; Ambion) in 260 μL PrepSEQ proteinase K buffer (4400787; Applied Biosystems) was added to the samples before transferring the samples to a 56°C heat block for 30 minutes. The phenol-chloroform extraction procedure involved two 500-μL UltraPure phenol:chloroform:isoamyl alcohol (25:24:1, v/v; 15593031; Invitrogen) extraction steps, followed by two 500-μL chloroform (C7559; Sigma-Aldrich) extraction steps. For each extraction step, the solution was vigorously shaken and centrifuged at 11,000 × g for 1 minute to separate the solution into its aqueous and organic layers. The upper aqueous layer was transferred to a clean tube for continued processing. After the last chloroform extraction step, the nucleic acids in the aqueous layer were precipitated with a 1:10 v/v solution of 3 mol/L NaOAc (AM9740; Ambion) and 2 volumes of 100% ethanol (E7023; Sigma-Aldrich), with 0.15 μg/μL Glycoblue (AM9516; Ambion) added. The samples were stored at −80°C for 20 minutes and then were centrifuged at 11,000 × g for 10 minutes. The nucleic acid pellets were washed once with 70% ethanol, partially dried, and then dissolved in 50 μL of DNA suspension buffer (10 mmol/L Tris and 0.1 mmol/L EDTA; T0221; Teknova).
Automate Express System
Cartridges from PrepFiler Express forensic DNA extraction kits (4441352; Applied Biosystems) were run on an Automate Express forensic DNA extraction system (4441763; Applied Biosystems) equipped with a version 1.5 β protocol card. Samples were lysed using a kit-provided lysis buffer supplemented with 10 mmol/L dl-dithiothreitol (43815; Sigma-Aldrich) for 40 minutes at 70°C. The manufacturer's user manual does not recommend lysozyme or proteinase K treatment, so none was performed. The Automate Express system generated eluates that were approximately 50 μL in volume.
Nucleic Acid Concentration Measurements
Samples were analyzed using a NanoDrop 8000 spectrophotometer (Thermo Scientific, Rockford, IL). The concentration of nucleic acids present in each sample was evaluated using A260 measurements.
Gene Analysis
TaqMan assays were performed according to the manufacturer's recommendations and were analyzed on an ABI 7500 real-time PCR system using the default thermal-cycling parameters. The ABI 7500 real-time system was equipped with SDS software v1.4.
Nucleic acid extracts were screened using TaqMan assays, including a Quantifiler Duo DNA quantification kit (4387746; Applied Biosystems), which measures the levels of ribonuclease P RNA component H1 (RNase P), a TaqMan S. aureus detection kit (4368606; Applied Biosystems), which targets the rnpB gene, and an assay described by Francois et al33 targeting the mecA gene, which confers resistance to methicillin and its derivatives. The 2X master mixes of both the Quantifiler Duo and S. aureus assays contained TaqMan exogenous internal positive control reagents (IPC), which were used to assess the presence of PCR inhibitors. The master mix for the mecA assay was also supplemented with this same internal positive control (4308323; Applied Biosystems). The IPC reagents in all master mixes included the necessary primers, probe, and IPC template for successful IPC amplification and detection.
Quantitative PCR
This study required accurate quantitation of the human RNase P gene, as well as S. aureus-specific rnpB and mecA genes. In order for a TaqMan assay to be considered quantitative, the following three conditions had to be met: i) the assay had to be run on a real-time instrument that had passed spectral and thermal calibration; ii) a dilution series of a standard (target sample of a known concentration) had to be included in the same reaction plate containing the unknown experimental sample or samples, with the CT values of these standards used to generate a best-fit standard curve line that was used, in turn, to determine the concentrations of the target gene within the experimental samples based on their CT values; and iii) the IPC CT value for the experimental sample or samples was ≤3.3 cycles from the mean IPC CT value of the no-template controls (NTCs). The NTCs included in the experiments consisted of 2 μL of nuclease-free water (AM9937; Ambion). By definition, the NTC should be free of PCR inhibitors. If the IPC CT value of an experimental sample was ≤3.3 cycles from the mean IPC CT value of the NTC samples, then the experimental sample was considered sufficiently free of PCR inhibition for relatively accurate DNA quantitation. If the difference was >3.3 cycles, the sample was considered to contain significant levels of inhibitors and could not be accurately quantitated. These sample extracts were diluted with nuclease-free water (AM9930; Ambion), in 10-fold increments, until the IPC CT value of the sample was ≤3.3 cycles from the mean IPC CT value of the NTC samples. Once a diluted experimental sample displayed an IPC CT value that was not inhibited, the amount of target-specific nucleic acids present in the sample could be accurately quantified based on the CT value for the target gene and the equation describing the standard curve for the target gene.
Standard Curves
For the present study, an S. aureus standard was created by purifying the nucleic acids from an overnight culture of MSSA (12600; ATCC) using the Automate Express instrument equipped with a PrepFiler Express forensic DNA extraction kit. The sample extract was quantified using a NanoDrop 8000 spectrophotometer and a threefold dilution series was created that ranged from 23 fg/μL to 50 pg/μL.
Results
Analytical Studies
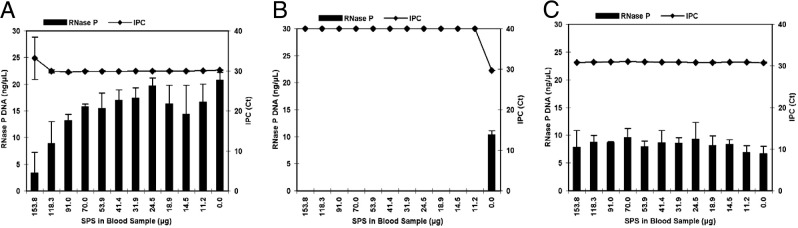
The effectiveness of traditional phenol-chloroform extraction, silica spin columns, and the Automate Express platform for removing SPS from blood samples was evaluated in a spiking experiment. SPS was spiked into blood samples in quantities ranging from 11.2 to 153.8 μg SPS per 50-μL blood sample. This range was intentionally designed to meet and exceed the 18.8 μg SPS that is found in a standard 50-μL blood culture sample, to account for situations in which higher volumes of blood culture are processed. Phenol-chloroform was found to effectively remove 11.2 to 70.0 μg SPS; these samples had stable IPC CT values (Figure 1A). Although the IPC CT values for the samples containing 91.0 to 118.3 μg SPS remained steady, the concentration of detectable RNase P DNA declined, compared with samples containing less SPS, suggesting that SPS was differently affecting the PCR efficiency of these assays. Phenol-chloroform did not adequately remove the highest concentration of SPS tested (153.8 μg/50-μL sample), based on IPC CT values for these samples that were higher than for the other samples tested. In contrast, silica columns failed to remove even the lowest concentration of SPS tested (11.2 μg/50-μL sample). The IPC CT values of these samples were undetermined (expressed as CT = 40), indicating the PCR was completely inhibited for these samples (Figure 1B). The Automate Express system successfully removed the highest concentration of SPS tested (153.8 μg/50-μL sample); no change in the IPC CT values was observed (Figure 1C).
Figure 1.
Detecting RNase P-specific target by PCR. Comparison of phenol-chloroform (A), spin column (B), and Automate Express (C) methods for purifying nucleic acids from blood samples spiked with different quantities of SPS and the use of this material in TaqMan assays for the detection of human RNase P. Samples were processed in triplicate. Data are expressed as means ± SD. IPC, internal positive control; SPS, sodium polyanetholsulfonate.
The quantity and purity of the nucleic acids (Figure 1) were assessed using 260 nm absorbance (A260) and 260/280 and 260/230 ratios. The 260 nm readings of the phenol-chloroform and silica column-processed samples were unreasonably high, considering the amount of blood processed. For the phenol-chloroform processed samples, the most likely explanation for this observation is the presence of Glycoblue in the extracts. This additive was included in the extraction process, at a concentration recommended by the manufacturer and known not to inhibit PCR, to aid in pellet visualization and to minimize the chance of aspirating the pellet during washing. For the silica column-processed samples, the level of 260 nm absorbance strongly correlated with the amount of SPS included in the samples (R2 = 0.97), and spectrophotometric analysis of pure SPS dissolved in water displayed a similar absorbance profile (data not shown). The presence of Glycoblue in the phenol-chloroform extracts and SPS in the silica extracts made it impossible to quantify the amount of nucleic acids in these samples. The confounding effect of Glycoblue on the 260 nm measurements for the phenol-chloroform extracts could have been avoided either by using glycogen that is not covalently linked to a blue dye or by using no glycogen at all,34 whereas the confounding nature of SPS on the 260 nm measurements for the silica-based extracts is unavoidable.34 In contrast, extracts generated from the Automate Express instrument were found to be pure, according to 260/280 and 260/230 ratios, and contained an average of 16.6 ± 1.4 ng/μL DNA, which is expected for the volume of blood processed.35
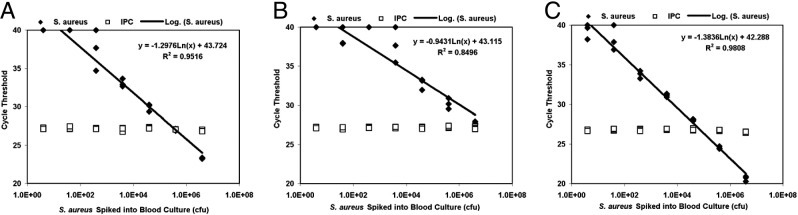
After demonstrating that SPS is a potent PCR inhibitor that is removed to different extents by the three chosen sample extraction methodologies, an experiment was performed to determine the detection limit for a blood-borne bacterial pathogen for each of the three extraction methodologies. For this experiment, a 10-fold dilution series of S. aureus was spiked into BACTEC culture medium containing blood at 3:1 v/v so that 50-μL samples contained 4, 40, 400, 4000, 40,000, 4 × 105, or 4 × 106 CFU of S. aureus. Spiked samples were processed using the three described sample extraction methodologies and the extracts were screened using a TaqMan assay specific to S. aureus. The nucleic acid extracts from samples processed using phenol-chloroform or silica columns required dilution, because using the undiluted extracts resulted in failed TaqMan analysis (Figure 2, A and B). To overcome the presence of inhibitors in these samples, it was necessary to dilute these DNA extracts by 10-fold and 1000-fold, respectively, before the concentration of inhibitors were reduced such that the IPC CT values of these samples approached the IPC CT value of the NTC samples. In contrast, extracts from the Automate Express instrument were not inhibited, as judged by the IPC CT values of these samples, so these extracts were entered into TaqMan assays undiluted. To maintain consistency across the spiked samples, all of the extracts by a particular methodology were diluted to the same extent.
Figure 2.
Detecting S. aureus-specific target by PCR. Comparison of phenol-chloroform (A), spin column (B), and Automate Express (C) methods for purifying nucleic acids from blood culture samples spiked with 10-fold dilution series of S. aureus and the use of this material in TaqMan assays for the detection of S. aureus. Each concentration of S. aureus was tested in triplicate.
Data from noninhibited TaqMan assays showed that phenol-chloroform was unable to purify sufficient nucleic acids from the samples spiked with 4, 40, and 400 CFU of S. aureus for reliable detection, because at least one sample at each concentration was not detected (CT = 40), whereas each of the three samples spiked with ≥4000 CFU had measurable CT values (Figure 2A). The considerable variation in CT values observed across the three samples spiked to 400 CFU/sample was likely due to i) the stochastic nature of TaqMan analysis for samples containing low levels of target; ii) the inherent variability introduced when operator skill is involved in retaining the sample through the multiple steps required for phenol-chloroform extraction, DNA precipitation, DNA pellet solubilization, and extract dilution; and iii) limitations in pipette accuracy for creating triplicate samples that are of identical concentration. Similar variability in CT values was observed for the samples spiked to 4000 CFU that were processed using silica columns. Silica column-processed samples were reliably detected only once the samples contained ≥40,000 CFU (Figure 2B). In contrast, the extracts from the Automate Express platform, which did not require any dilution, allowed for S. aureus to be detected at ≥400 CFU/sample (Figure 2C). Linear regression analysis of these data indicated that phenol-chloroform processing and the Automate Express system are more reliable than silica columns at extracting a wide concentration range of S. aureus nucleic acids from samples for accurate quantitative detection by TaqMan analysis; R2 values were 0.95, 0.98, and 0.84 for phenol-chloroform, Automate Express, and silica column methodologies, respectively.
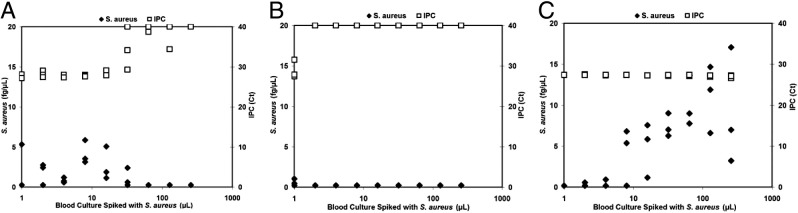
Next, different sample volumes were tested to determine whether each methodology has an optimal sample volume that results in the most reliable detection of low-titer pathogens in blood culture. For these experiments, 13,000 CFU/mL of S. aureus was added to BACTEC Plus Aerobic/F medium containing blood at 3:1 v/v. Sample volumes from 1 to 256 μL were drawn from this mixture and processed by the three sample extraction methodologies after volume normalization to 50 µL, as described in Materials and Methods. The extracts were screened using TaqMan assays specific for S. aureus. Phenol-chloroform-based extraction resulted in unreliable detection for sample volumes of 1 to 2 μL, whereas for sample volumes of 4, 8, and 16 μL reliably yielded CT values of <40 (Figure 3A). With sample volumes ≥32 μL, the phenol-chloroform methodology did not reliably detect S. aureus; most IPC CT values were at 40, suggesting SPS and other PCR inhibitors present in the samples were not effectively removed during sample extraction. In agreement with previously generated data, silica columns were unable to adequately purify nucleic acids from any of the sample volumes tested. The 1-μL samples did not contain enough SPS to prevent IPC amplification, but these samples also did not contain enough S. aureus for reliable detection. The samples containing ≥2 μL of blood culture fluid were not amplified, as judged by the failure to detect the IPC. This finding suggests that sufficient amounts of SPS remained in the extracts to prevent PCR amplification (Figure 3B). The Automate Express platform successfully processed sample volumes of ≤256 μL without any observed PCR inhibition. However, only samples containing ≥16 μL of culture fluid contained sufficient levels of S. aureus for reliable detection using this methodology (Figure 3C).
Figure 3.
Detecting S. aureus-specific target by PCR. Comparison of phenol-chloroform (A), spin column (B), and Automate Express (C) methods for purifying nucleic acids from different volumes of blood culture fluid samples spiked with low-titer S. aureus and the use of this material in TaqMan assays for the detection of S. aureus. Each volume was tested in triplicate. Note that the S. aureus genome weighs approximately 3 fg.
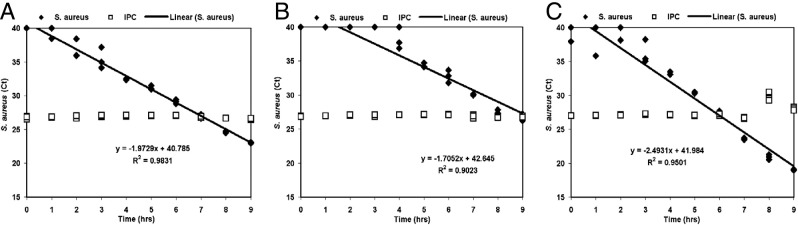
After the range of sample volumes that can effectively be processed by each methodology was established, experiments were performed to determine which methodology first detected a growing S. aureus culture. Although the Automate Express instrument was able to process larger volumes of sample than the other two methodologies (Figure 3C), equal sample volumes (50 μL) were compared for this experiment. S. aureus was spiked into a mixture of BACTEC medium and blood at 3:1 v/v to 2060 CFU/mL and incubated under shaking aerobic conditions at 37°C. Samples were drawn from this culture every hour and processed by the three sample extraction methodologies. It was necessary to dilute the extracts from phenol-chloroform and silica column-extracted samples by 100-fold and 1000-fold, respectively, to sufficiently dilute the inhibitors retained through the extraction process so that TaqMan analysis was not inhibited. As already noted, it was not necessary to dilute the Automate Express instrument extracts, because the use of these extracts in TaqMan assays resulted in IPC CT values similar to the IPC CT values of the NTC samples. Analysis of noninhibited samples indicated that phenol-chloroform-processed samples were reliably detected by TaqMan analysis just 2 hours after the initial spike (Figure 4A). The Automate Express platform generated extracts that were used to reliably detect S. aureus 3 hours after the initial spike (Figure 4C). Although the 8- and 9-hour samples processed by the Automate Express instrument had IPC CT values that were higher than the NTC IPC CT value (data not shown), these higher values likely reflect competition for PCR reagents, rather than indicating PCR inhibition, because the amount of target template was very high (CT ≤ 21). In contrast, the silica column-extracted samples generated eluates that could not be used to reliably detect S. aureus until 5 hours after the initial spike (Figure 4B). Linear regression analysis of these data indicated that phenol-chloroform processing and the Automate Express instrument are more reliable than silica columns at extracting nucleic acids across a wide concentration range of S. aureus in samples for quantitative detection by TaqMan analysis (R2 values of 0.98, 0.95, and 0.90 for phenol-chloroform, Automate Express, and silica column methodologies, respectively).
Figure 4.
Detecting S. aureus-specific target by PCR. Comparison of phenol-chloroform (A), spin column (B), and Automate Express (C) methods for purifying nucleic acids from samples taken every hour from a blood culture spiked to 2060 CFU of S. aureus per milliliter and the use of this material in TaqMan assays for the detection of S. aureus. Each time point was tested in triplicate.
Clinical Studies
Lastly, the performance of these sample extraction methodologies were compared using 60 clinical blood culture samples (50 μL), for which the extracts were screened using TaqMan assays specific for both S. aureus and the mecA gene to differentiate between MSSA and MRSA. Because of the presence of PCR inhibitors in some of the extracts, it was necessary to dilute these samples for accurate TaqMan analysis. The CT value of the S. aureus assay from the most concentrated sample that yielded an IPC CT value that was ≤3.3 cycles away from the CT of the NTC IPC was recorded. The CT values (mean ± SD) for the S. aureus-positive samples were 18.9 ± 2.6, 26.3 ± 2.7, and 17.6 ± 3.2 for phenol-chloroform, silica column, and Automate Express instrument processed samples, respectively.
The choice of sample extraction methodology did not affect the qualitative nature of TaqMan assay results. In all three methods used, 30 of the 60 samples were found to be positive for S. aureus (Table 1), with 12 of these positives being positive also for mecA (Table 2). The remaining 30 samples were found to be negative for S. aureus using the S. aureus TaqMan assay. Cross-referencing these data with the culture diagnosis revealed complete concordance with traditional phenotypic identification methods. These data indicate that the S. aureus TaqMan assay has a sensitivity and specificity of 100% [95% confidence interval (CI) = 88.7 to 100] and that the mecA TaqMan assay has a sensitivity of 100% (95% CI = 75.8 to 100) and a specificity of 100% (95% CI = 92.6 to 100).
Table 1.
Conventional Blood Culture Results Compared with TaqMan S. aureus Assay Outcome
| Outcome, S. aureus assay | Clinical results (no.) |
Total | |
|---|---|---|---|
| Positive | Negative | ||
| Positive | 30 | 0 | 30 |
| Negative | 0 | 30 | 30 |
| Total | 30 | 30 | 60 |
Clinical results represent the true diagnosis.
Table 2.
Conventional Blood Culture Results Compared with TaqMan mecA Assay Outcome
| Outcome, mecA assay | Clinical results (no.) |
Total | |
|---|---|---|---|
| Positive | Negative | ||
| Positive⁎ | 12 | 0 | 12 |
| Negative | 0 | 48 | 48 |
| Total | 12 | 48 | 60 |
Clinical results represent the true diagnosis.
All samples positive for mecA were also positive for S. aureus (Table 1).
Discussion
DNA extracts generated from the phenol-chloroform method and Automate Express instrument had essentially equal sensitivity and early detection limits for detecting S. aureus from aliquots of blood culture fluid. The small differences observed (Figure 2, Figure 4) were likely due to the small sampling size of these experiments, because just one of the triplicate samples in each experiment prevented statistically identical results. Although these two technologies qualitatively yielded the same data, the Automate Express platform was the only technology of the three tested that was capable of purifying nucleic acids from 50-μL blood culture samples without requiring dilution of the extracts before PCR (dilution is required if copurified PCR inhibitors are present). The Automate Express instrument effectively processed blood culture samples as large as 256 μL in volume, which may further enhance the probability of detecting low numbers of blood-borne bacterial pathogens (Figure 3). In contrast, phenol-chloroform and silica spin columns were unable to process large volume samples (32 μL and 2 μL, respectively) without requiring dilution of the extracts, a step that reduced assay sensitivity.
Our data (Figure 1, Figure 2, Figure 3, Figure 4) suggest that blood culture fluids contained additional PCR inhibitors aside from SPS that must be sufficiently removed for accurate PCR analysis. In one experiment (Figure 1), the samples were composed of blood and SPS only, with no culture medium or bacteria; the phenol-chloroform extraction method was used successfully to process the samples containing ≤91.0 μg SPS without the need to dilute the extracts for inhibition-free PCR. In the other experiments (Figure 2, Figure 3, Figure 4), the blood culture samples contained only 18.8 μg SPS from the BACTEC Plus Aerobic/F medium; however, these samples required 10-fold to 100-fold dilutions to achieve inhibition-free PCR. The requirement to dilute these blood culture extracts removed the apparent advantage of phenol-chloroform processing in recovering more nucleic acids than the Automate Express; the two methodologies recovered an average of 15.4 and 8.4 ng/μL RNase P DNA from the blood samples spiked with ≤91.0 μg SPS, respectively (Figure 1). Of note, manually performing the Automate Express chemistry regularly yielded approximately twofold more DNA than the automated platform (data not shown). Because phenol-chloroform-processed blood culture samples had to be diluted 10-fold to 100-fold to achieve inhibition-free PCR, the concentration of these extracts dropped to levels less than what was achieved using the Automate Express instrument. The lower DNA concentration for the phenol-chloroform-processed samples was indicated by the higher CT values generated, compared with those samples processed by the Automate Express (Figure 2, Figure 4). These data suggest that phenol-chloroform may not be as effective as the Automate Express forensic DNA extraction system at removing any additional PCR inhibitors (aside from SPS) present in the blood culture fluids.
The ability of the Automate Express forensic DNA extraction system to adequately purify nucleic acids from blood culture samples effectively reduced the cost of analysis in half, because it was not necessary to screen multiple dilutions. The benefit of analyzing undiluted sample extracts was clear, in that the Automate Express instrument generated extracts that allowed for S. aureus to be detected at an average CT of 17.6, which was 8.7 cycles earlier than observed for the samples processed using silica columns. This difference equates to a 415-fold difference in the original template concentration. Considering that S. aureus has a 24-minute doubling time,36 a culture that tests positive with the aid of Automate Express sample extraction would require approximately an additional 3.5 hours of exponential growth to reach the CFU/mL required for positive TaqMan-based detection using silica columns. This calculation is supported by our data (Figure 4) showing a 2-hour advantage in the point of first detection for samples processed by the Automate Express compared to samples processed using silica columns. Although this advantage is modest for the fast-growing S. aureus, it could prove to be more substantial for slower growing pathogens.
The time for a culture to reach the density required for accurate detection is one component that contributes to TTR. Another component is the time required to perform sample extraction. Both phenol-chloroform and silica columns required incubations for lysozyme and proteinase K treatment (60 minutes total), whereas the Automate Express system required only a single lysis step of 40 minutes. In addition to the increased time required for these incubations, the phenol-chloroform method also required the extracted material to be precipitated, which significantly lengthened the time required to generate nucleic acids suitable for PCR. There was also the issue of hands-on time versus automation. Taken together, the Automate Express system was the most rapid methodology evaluated in the present study for generating purified nucleic acid extracts, requiring just 80 minutes to process 12 samples, whereas silica column and phenol-chloroform-processed samples required approximately 100 and 180 minutes, respectively. The difference in the time required to generate extracts using these three different methodologies would increase as more samples are processed.
The data presented here indicate that silica-based purification methodologies copurified SPS, whereas the chemistries used by the phenol-chloroform method and the Automate Express did not. It may be that SPS and DNA (both of which are polyanions) interacted similarly with the silica under high-salt conditions, causing the observed copurification. The exact mechanism of DNA and SPS adsorption to silica under conditions of high ionic strength is not clear. One report indicates that DNA adsorption is due to weakened intermolecular electrostatic repulsion forces, dehydration of the DNA and silica surfaces, and intermolecular hydrogen bond formation in the DNA-silica contact layer,37 whereas other reports claim that cations such as sodium or guanidinium disrupt the hydration sphere around DNA and that these positively charged ions form a salt bridge between the negatively charged silica and the DNA backbone.38
In contrast to the silica fibers of the spin columns, the surface of the magnetic particles contained within the cartridges of the PrepFiler Express forensic DNA extraction kit were embedded with a hydrophilic polymer. The HMW polysaccharide presented in the lysis/binding buffer formed a complex with DNA on the addition of alcohol. This complex bound to the surface of the magnetic particles. It is unclear why the similarly charged SPS does not also become trapped in the HMW polysaccharide-DNA complex; possibly it was excluded because of its smaller size. The function of the soluble HMW polysaccharide in the PrepFiler kit was essential, and may be compared with that of glycogen, which is used to facilitate nucleic acid precipitation after phenol-chloroform extraction (as was done in the present study). The difference is that the DNA-polysaccharide complex preferentially bound to the magnetic particles, which nucleated the process, whereas the glycogen/DNA complexes required high-speed centrifugation to form a discrete pellet.
Aside from differences in handling of SPS and other inhibitors found in blood cultures, silica and HMW polysaccharide-based sample extraction methodologies differed in DNA binding capacity and DNA binding kinetics. The HMW polysaccharide-based method was capable of extracting more DNA from a sample with high DNA content than was possible using silica. In other experiments (data not shown), equal volumes of Dynabeads MyOne Silane beads (370-02D; Invitrogen, Carlsbad, CA), which are magnetic beads coupled to silanol groups that are functionally equivalent to silica, and the magnetic particles of a PrepFiler forensic DNA extraction kit (4392353l; Applied Biosystems) were compared in their DNA binding capacity during processing of nine different volumes of whole blood, ranging from 2 to 200 μL. The PrepFiler magnetic particles extracted 200 ng to 20 μg of DNA with a linear relationship between the amount of DNA extracted and the volume of blood processed (r2 = 0.99). In contrast, the amount of DNA extracted using the Dynabeads peaked at 890 ng of DNA for the 12.5-μL blood sample, and steadily dropped with higher sample volumes down to just 90 ng for the 200-μL sample. This drop in DNA extraction for the higher blood sample volumes may be due to competition with RNA and protein for the silanol binding sites. These data were consistent with another report, that silica saturates at 6.2 to 28.3 ng of nucleic acids per milligram of silica.39 These data suggest that, in HMW polysaccharide-based DNA purification, the magnetic particles can continue to bind HMW polysaccharide-DNA complexes, limited only by the point at which the weight of the polysaccharide-DNA complexes exceed the magnetic pull of the iron oxide core, preventing effective washing of the magnetic particles. In regards to DNA binding kinetics, HMW polysaccharide-based binding required 10 minutes to near completion, whereas binding of nucleic acids to silica occurred in <1 minute (data not shown).
In the present study, the findings for silica-based extraction of blood cultures are similar to data previously reported by Fredricks et al.9 Aside from the similarities in the silica-based extraction data, however, there are several striking differences between these two studies; the most important of which is the discovery of a sample extraction methodology that does not require the use of a fume hood and can be used to purify blood culture fluids without the requirement of diluting the extracts for inhibition-free PCR. Of the six different DNA purification methods tested by Fredricks et al,9 only a benzyl alcohol-guanidine hydrochloride organic extraction method generated extracts that were pure enough to be used undiluted in PCR and in pyrosequencing for pathogen identification.40 Although a direct comparison between the benzyl alcohol extraction method and the Automate Express chemistry was not performed for the present study, both are able to generate extracts that can be directly used in PCR assays. Of lesser importance, Fredricks et al9 reported that phenol-chloroform extracts require 1000-fold dilution to achieve inhibition-free PCR, whereas in the present study only 10-fold to 100-fold dilutions were required, depending on the experiment. This discrepancy may be explained by differences in methodology and chemicals used. In the present study, two rounds of phenol:chloroform:isoamyl alcohol extraction were followed by two rounds of chloroform extraction and glycogen-aided DNA precipitation. In contrast, Fredricks et al9 performed three rounds of phenol:chloroform extraction (with no isoamyl alcohol), followed by one round of chloroform extraction and DNA precipitation without the aid of glycogen. Finally, the present study included analysis of 60 clinical samples, to determine how each methodology handles real-world samples and so determined which methodology would be best for reducing TTR, whereas Fredricks et al9 dealt only with spiked-in samples and the detection of contaminating bacterial DNA in sterile culture medium.
The present findings support a recent report that different extraction technologies have advantages in certain applications and that no one methodology is ideally suited for all applications.41 Of the three technologies evaluated here, the HMW polysaccharide-based magnetic DNA purification chemistry performed by the Automate Express instrument was the only chemistry capable of generating extracts from culture medium that could be directly used in PCR without any observed PCR inhibition, and the extracts have exceptional 260/280 and 260/230 ratios that allow for accurate DNA quantitation. This technology differed from the other two tested in that it did not rely on lysozyme pretreatment. The inclusion of a lysozyme pretreatment step before the recommended protocol was found to not significantly benefit or harm the performance of the Automate Express platform in extracting S. aureus DNA for TaqMan-based PCR detection (data not shown). Also, the HMW polysaccharide-based magnetic extraction procedure did not require the use of a fume hood and was automated, making it a particularly attractive alternative to phenol-chloroform extraction. The Automate Express platform not only reduced hands-on time, but the DNA extraction workflow reduced the TTR to a greater extent than was possible when either silica columns or phenol-chloroform was used to extract the samples. Here, the PrepFiler Express forensic DNA extraction kits and the associated Automate Express platform have demonstrated utility outside of forensic applications, in that this technology proved to be very effective at removing the PCR inhibitors found in blood cultures.
Footnotes
Supported in large part by Life Technologies, which also supplied reagents.
Disclosures: J.F.R., M.R.F., and M.G.B. are employed by and hold stock options in Life Technologies.
References
- 1.Mallon P.W., Millar B.C., Moore J.E., Murphy P.G., McClurg R.B., Chew E.W., Crowe M.J. Molecular identification of Acinetobacter sp. in a patient with culture-negative endocarditis. Clin Microbiol Infect. 2000;6:277–278. doi: 10.1046/j.1469-0691.2000.00058-4.x. [DOI] [PubMed] [Google Scholar]
- 2.Cockerill F.R., 3rd, Reed G.S., Hughes J.G., Torgerson C.A., Vetter E.A., Harmsen W.S., Dale J.C., Roberts G.D., Ilstrup D.M., Henry N.K. Clinical comparison of BACTEC 9240 plus aerobic/F resin bottles and the isolator aerobic culture system for detection of bloodstream infections. J Clin Microbiol. 1997;35:1469–1472. doi: 10.1128/jcm.35.6.1469-1472.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Qian Q., Tang Y.W., Kolbert C.P., Torgerson C.A., Hughes J.G., Vetter E.A., Harmsen W.S., Montgomery S.O., Cockerill F.R., 3rd, Persing D.H. Direct identification of bacteria from positive blood cultures by amplification and sequencing of the 16S rRNA gene: evaluation of BACTEC 9240 instrument true-positive and false-positive results. J Clin Microbiol. 2001;39:3578–3582. doi: 10.1128/JCM.39.10.3578-3582.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Karahan Z.C., Mumcuoglu I., Guriz H., Tamer D., Balaban N., Aysev D., Akar N. PCR evaluation of false-positive signals from two automated blood-culture systems. J Med Microbiol. 2006;55:53–57. doi: 10.1099/jmm.0.46196-0. [DOI] [PubMed] [Google Scholar]
- 5.Wadsworth A., Maltaner F., Maltaner E. The inhibition of complementary activity by anticoagulants. J Immunol. 1937;33:297–303. [Google Scholar]
- 6.Traub W.H. Antagonism of polymyxin B and kanamycin sulfate by Liquoid (sodium polyanetholsulfonate) in vitro. Experientia. 1969;25:206–207. doi: 10.1007/BF01899127. [DOI] [PubMed] [Google Scholar]
- 7.Traub W.H., Lowrance B.L. Media-dependent antagonism of gentamicin sulfate by Liquoid (sodium polyanetholsulfonate) Experientia. 1969;25:1184–1185. doi: 10.1007/BF01900265. [DOI] [PubMed] [Google Scholar]
- 8.Traub W.H., Lowrance B.L. Anticomplementary, anticoagulatory, and serum-protein precipitating activity of sodium polyanetholsulfonate. Appl Microbiol. 1970;20:465–468. doi: 10.1128/am.20.3.465-468.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fredricks D.N., Relman D.A. Improved amplification of microbial DNA from blood cultures by removal of the PCR inhibitor sodium polyanetholesulfonate. J Clin Microbiol. 1998;36:2810–2816. doi: 10.1128/jcm.36.10.2810-2816.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Palarasah Y., Skjoedt M.O., Vitved L., Andersen T.E., Skjoedt K., Koch C. Sodium polyanethole sulfonate as an inhibitor of activation of complement function in blood culture systems. J Clin Microbiol. 2010;48:908–914. doi: 10.1128/JCM.01985-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Krogstad D.J., Murray P.R., Granich G.G., Niles A.C., Ladenson J.H., Davis J.E. Sodium polyanethol sulfonate inactivation of aminoglycosides. Antimicrob Agents Chemother. 1981;20:272–274. doi: 10.1128/aac.20.2.272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Akane A., Matsubara K., Nakamura H., Takahashi S., Kimura K. Identification of the heme compound copurified with deoxyribonucleic acid (DNA) from bloodstains, a major inhibitor of polymerase chain reaction (PCR) amplification. J Forensic Sci. 1994;39:362–372. [PubMed] [Google Scholar]
- 13.Higuchi R. Simple and rapid preparation of samples for PCR. In: Erlich H.A., editor. PCR Technology: Principles and Applications for DNA Amplification. Stockton Press; New York: 1989. pp. 31–38. [Google Scholar]
- 14.Beutler E., Gelbart T., Kuhl W. Interference of heparin with the polymerase chain reaction. Biotechniques. 1990;9:166. [PubMed] [Google Scholar]
- 15.Kreader C.A. Relief of amplification inhibition in PCR with bovine serum albumin or T4 gene 32 protein. Appl Environ Microbiol. 1996;62:1102–1106. doi: 10.1128/aem.62.3.1102-1106.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jung R., Lübcke C., Wagener C., Neumaier M. Reversal of RT-PCR inhibition observed in heparinized clinical specimens. Biotechniques. 1997;23:24. doi: 10.2144/97231bm03. 26, 28. [DOI] [PubMed] [Google Scholar]
- 17.Al-Soud W.A., Jönsson L.J., Rådström P. Identification and characterization of immunoglobulin G in blood as a major inhibitor of diagnostic PCR. J Clin Microbiol. 2000;38:345–350. doi: 10.1128/jcm.38.1.345-350.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Al-Soud W.A., Rådström P. Purification and characterization of PCR-inhibitory components in blood cells. J Clin Microbiol. 2001;39:485–493. doi: 10.1128/JCM.39.2.485-493.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lam N.Y., Rainer T.H., Chiu R.W., Lo Y.M. 2004. EDTA is a better anticoagulant than heparin or citrate for delayed blood processing for plasma DNA analysis; pp. 256–257. 50. [DOI] [PubMed] [Google Scholar]
- 20.Huggett J.F., Novak T., Garson J.A., Green C., Morris-Jones S.D., Miller R.F., Zumla A. Differential susceptibility of PCR reactions to inhibitors: an important and unrecognised phenomenon. BMC Res Notes. 2008;1:70. doi: 10.1186/1756-0500-1-70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Loonen A.J., Jansz A.R., Kreeftenberg H., Bruggeman C.A., Wolffs P.F., van den Brule A.J. Acceleration of the direct identification of Staphylococcus aureus versus coagulase-negative staphylococci from blood culture material: a comparison of six bacterial DNA extraction methods. Eur J Clin Microbiol Infect Dis. 2011;30:337–342. doi: 10.1007/s10096-010-1090-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tang Y.W., Kilic A., Yang Q., McAllister S.K., Li H., Miller R.S., McCormac M., Tracy K.D., Stratton C.W., Han J., Limbago B. StaphPlex system for rapid and simultaneous identification of antibiotic resistance determinants and Panton-Valentine leukocidin detection of staphylococci from positive blood cultures. J Clin Microbiol. 2007;45:1867–1873. doi: 10.1128/JCM.02100-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Millar B.C., Jiru X., Moore J.E., Earle J.A. A simple and sensitive method to extract bacterial, yeast and fungal DNA from blood culture material [Erratum appeared in J Microbiol Methods 2001, 47:255] J Microbiol Methods. 2000;42:139–147. doi: 10.1016/s0167-7012(00)00174-3. [DOI] [PubMed] [Google Scholar]
- 24.Chomczynski P., Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem. 1987;162:156–159. doi: 10.1006/abio.1987.9999. [DOI] [PubMed] [Google Scholar]
- 25.Boom R., Sol C.J., Salimans M.M., Jansen C.L., Wertheim-van Dillen P.M., van der Noordaa J. rapid and simple method for purification of nucleic acids. J Clin Microbiol. 1990;28:495–503. doi: 10.1128/jcm.28.3.495-503.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kramvis A., Bukofzer S., Kew M.C. Comparison of hepatitis B virus DNA extractions from serum by the QIAamp blood kit, GeneReleaser, and the phenol-chloroform method. J Clin Microbiol. 1996;34:2731–2733. doi: 10.1128/jcm.34.11.2731-2733.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Böddinghaus B., Wichelhaus T.A., Brade V., Bittner T. Removal of PCR inhibitors by silica membranes: evaluating the Amplicor Mycobacterium tuberculosis kit. J Clin Microbiol. 2001;39:3750–3752. doi: 10.1128/JCM.39.10.3750-3752.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Brevnov M.G., Pawar H.S., Mundt J., Calandro L.M., Furtado M.R., Shewale J.G. Developmental validation of the PrepFiler forensic DNA extraction kit for extraction of genomic DNA from biological samples. J Forensic Sci. 2009;54:599–607. doi: 10.1111/j.1556-4029.2009.01013.x. [DOI] [PubMed] [Google Scholar]
- 29.Klein E., Smith D.L., Laxminarayan R. Hospitalizations and deaths caused by methicillin-resistant Staphylococcus aureus, United States, 1999–2005. Emerg Infect Dis. 2007;13:1840–1846. doi: 10.3201/eid1312.070629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Noskin G.A., Rubin R.J., Schentag J.J., Kluytmans J., Hedblom E.C., Jacobson C., Smulders M., Gemmen E., Bharmal M. National trends in Staphylococcus aureus infection rates: impact on economic burden and mortality over a 6-year period (1998–2003) Clin Infect Dis. 2007;45:1132–1140. doi: 10.1086/522186. [DOI] [PubMed] [Google Scholar]
- 31.Klevens R.M., Gorwitz R.J., Collins A.S. Methicillin-resistant Staphylococcus aureus: a primer for dentists. J Am Dent Assoc. 2008;139:1328–1337. doi: 10.14219/jada.archive.2008.0044. [DOI] [PubMed] [Google Scholar]
- 32.Wecke J., Lahav M., Ginsburg I., Giesbrecht P. Cell wall degradation of Staphylococcus aureus by lysozyme. Arch Microbiol. 1982;131:116–123. doi: 10.1007/BF01053992. [DOI] [PubMed] [Google Scholar]
- 33.Francois P., Pittet D., Bento M., Pepey B., Vaudaux P., Lew D., Schrenzel J. Rapid detection of methicillin-resistant Staphylococcus aureus directly from sterile or nonsterile clinical samples by a new molecular assay. J Clin Microbiol. 2003;41:254–260. doi: 10.1128/JCM.41.1.254-260.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hengen P.N. Methods and reagents: carriers for precipitating nucleic acids. Trends Biochem Sci. 1996;21:224–225. doi: 10.1016/0968-0004(96)30023-6. [DOI] [PubMed] [Google Scholar]
- 35.Adeli K., Ogbonna G. Rapid purification of human DNA from whole blood for potential application in clinical chemistry laboratories. Clin Chem. 1990;36:261–264. [PubMed] [Google Scholar]
- 36.Domingue G., Costerton J.W., Brown M.R. Bacterial doubling time modulates the effects of opsonisation and available iron upon interactions between Staphylococcus aureus and human neutrophils. FEMS Immunol Med Microbiol. 1996;16:223–228. doi: 10.1111/j.1574-695X.1996.tb00139.x. [DOI] [PubMed] [Google Scholar]
- 37.Melzak K.A., Sherwood C.S., Turner R.F.B., Haynes C.A. Driving forces for DNA adsorption to silica in perchlorate solutions. J Colloid Interface Sci. 1996;181:635–644. [Google Scholar]
- 38.Vogelstein B., Gillespie D. Preparative and analytical purification of DNA from agarose. Proc Natl Acad Sci USA. 1979;76:615–619. doi: 10.1073/pnas.76.2.615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tian H., Hühmer A.F., Landers J.P. Evaluation of silica resins for direct and efficient extraction of DNA from complex biological matrices in a miniaturized format. Anal Biochem. 2000;283:175–191. doi: 10.1006/abio.2000.4577. [DOI] [PubMed] [Google Scholar]
- 40.Jordan J.A., Jones-Laughner J., Durso M.B. Utility of pyrosequencing in identifying bacteria directly from positive blood culture bottles. J Clin Microbiol. 2009;47:368–372. doi: 10.1128/JCM.01991-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Rose H.L., Dewey C.A., Ely M.S., Willoughby S.L., Parsons T.M., Cox V., Spencer P.M., Weller S.A. Comparison of eight methods for the extraction of Bacillus atrophaeusspore DNA from eleven common interferents and a common swab. PLoS One. 2011;6(7):e22668. doi: 10.1371/journal.pone.0022668. [DOI] [PMC free article] [PubMed] [Google Scholar]