Abstract
Background
Several clusters of serogroup C meningococcal disease among men who have sex with men (MSM) have been reported in the United States in recent years. The epidemiology and risk of meningococcal disease among MSM is not well described.
Methods
All meningococcal disease cases among men aged 18–64 years reported to the National Notifiable Disease Surveillance System between January 2012 and June 2015 were reviewed. Characteristics of meningococcal disease cases among MSM and men not known to be MSM (non-MSM) were described. Annualized incidence rates among MSM and non-MSM were compared through calculation of the relative risk and 95% confidence intervals. Isolates from meningococcal disease cases among MSM were characterized using standard microbiological methods and whole-genome sequencing.
Results
Seventy-four cases of meningococcal disease were reported among MSM and 453 among non-MSM. Annualized incidence of meningococcal disease among MSM was 0.56 cases per 100 000 population, compared to 0.14 among non-MSM, for a relative risk of 4.0 (95% confidence interval [CI], 3.1–5.1). Among the 64 MSM with known status, 38 (59%) were infected with human immunodeficiency virus (HIV). HIV-infected MSM had 10.1 times (95% CI, 6.1–16.6) the risk of HIV-uninfected MSM. All isolates from cluster-associated cases were serogroup C sequence type 11.
Conclusions
MSM are at increased risk for meningococcal disease, although the incidence of disease remains low. HIV infection may be an important factor for this increased risk. Routine vaccination of HIV-infected persons with a quadrivalent meningococcal conjugate vaccine in accordance with Advisory Committee on Immunization Practices recommendations should be encouraged.
Keywords: Neisseria meningitidis, meningococcal infections, homosexuality, male
Meningococcal disease is a serious bacterial infection caused by Neisseria meningitidis and can lead to high morbidity and mortality rates [1, 2]. During 2010–2015, clusters of serogroup C meningococcal disease among men who have sex with men (MSM) were reported in the United States in New York City (NYC), Los Angeles County (LAC), and Chicago, disproportionately affecting MSM with human immunodeficiency virus (HIV) infection [3]. In addition, clusters among MSM have been reported in Berlin and Paris [4, 5]. However, little is known about the overall burden or risk of meningococcal disease among MSM, including HIV-infected MSM, in the United States.
MSM have not historically been considered at increased risk for meningococcal disease in the United States and are not routinely vaccinated against meningococcal disease outside of the adolescent quadrivalent meningococcal conjugate vaccination program unless certain underlying conditions are present [1]. However, HIV infection is increasingly recognized as a risk factor for meningococcal disease, and in June 2016, the Advisory Committee on Immunization Practices (ACIP) recommended routine quadrivalent meningococcal conjugate vaccine for all HIV-infected persons aged ≥2 months [6].
In this evaluation, we estimate the incidence of meningococcal disease among MSM in the United States, describe characteristics of MSM with meningococcal disease, estimate the risk of meningococcal disease by HIV status among MSM compared to men not known to be MSM, and describe the molecular characteristics of disease-causing strains to better understand the epidemiology of meningococcal disease in this population.
METHODS
Surveillance and Descriptive Epidemiology
In the United States, meningococcal disease cases are reported to the Centers for Disease Control and Prevention (CDC) through the National Notifiable Disease Surveillance System (NNDSS) and are classified by state and local public health personnel according to the Council of State and Territorial Epidemiologists case definition [7]. As sex of meningococcal disease patients’ sex partners is not collected through NNDSS, CDC issued requests for this information in 2013 and 2015 through the Epidemic Information Exchange (Epi-X), CDC’s system for rapid and secure exchange of public health information between CDC and state and local health departments, and received responses from health departments from all 50 states and the District of Columbia. State and local health departments were asked to identify MSM among meningococcal patients aged 18–64 years prospectively during case investigations. In addition, health departments conducted retrospective reviews of cases reported since 2012, identifying men as MSM based on interviews completed during the investigation of the case or disclosures of sexual contact with another man during contact tracing.
For patients identified as MSM, supplemental demographic, laboratory, and risk factor data, such as HIV status, smoking, and drug use were abstracted from case investigation records and linked to the patient record in NNDSS for analysis. Additional data on sexual behaviors, such as multiple or anonymous sex partners, were collected by state and local health departments during the investigation of cases that occurred as part of a cluster. HIV infection status and risk behavior data were not available for men not known to be MSM. Cases were classified as MSM if identified as such during case investigation or as non-MSM if not known to be MSM. MSM cases were further classified into jurisdiction-specific groups (NYC, LAC, and Chicago) if an outbreak or cluster of meningococcal disease among MSM was reported by a health department during the observation period, or as sporadic cases reported outside of the jurisdictions with reported outbreaks or clusters. For these jurisdictions, all cases with residence in that jurisdiction were included in jurisdiction-specific estimates, regardless of serogroup or molecular linkage to the cluster. Cases that were epidemiologically linked to an outbreak or cluster but with residence outside of the jurisdiction were not included.
Laboratory Methods
Available isolates and clinical specimens from MSM cases were sent to the CDC’s Bacterial Meningitis Laboratory where confirmatory testing and molecular typing for N. meningitidis were performed. Serogroup was determined by slide agglutination and/or serogroup-specific real-time polymerase chain reaction.
The genomes of all isolates were sequenced using Illumina technology and assembled as described in Kretz et al [8]. Multilocus sequence typing (MLST) alleles were identified based on a BLAST search of the assembled genomes against the PubMLST allele lists [9]. Protein sequences were likewise typed according to PubMLST sequence collection. PorA, PorB, and FetA were classified according to their respective variable regions, NadA was categorized by the Novartis convention of variant and peptide ID [10], NhbA was identified by PubMLST peptide identifier, and FHbp was identified by the PubMLST peptide identifier and the Pfizer peptide identifier (subfamilies A and B).
For each comparison of genome-wide similarity, core single- nucleotide polymorphisms (SNPs) were identified using kSNP v3 software [11], with a kmer length of 25, and SNP allele differences between strains were calculated using pairwise comparison. SNPs were then compared across all genomes. A maximum likelihood phylogenetic tree was built based on core SNPs, using MEGA6 [12], the Tamura-Nei substitution model with 500 bootstrap iterations.
Statistical Analysis
A descriptive analysis of demographic characteristics, clinical features, and outcomes was conducted. Significant differences (P < .05) among MSM and non-MSM were assessed through χ2 or Fisher exact test. Annualized incidence was calculated as the number of meningococcal disease cases per 100 000 men aged 18–64 years per year using national and jurisdiction-specific population estimates for all men, all MSM, HIV-infected and -uninfected MSM, and non-MSM. The populations of men aged 18–64 years in the United States and by jurisdiction were derived from the 2012 American Community Survey [13]. The proportion of males who are MSM, defined as sex with another man in the past 5 years, was estimated to be 3.9% nationwide [14], and the proportions of MSM in LAC, NYC, Chicago, and all other United States jurisdictions were estimated to be 6.8%, 7.3%, 6.6%, and 3.3%, respectively [15]. The proportions of HIV-infected MSM nationwide and by jurisdiction were derived from national and jurisdiction-specific surveillance reports [16–19]. The overall and jurisdiction-specific numbers of MSM, HIV-infected MSM, HIV-uninfected MSM, and non-MSM were calculated by multiplying the male population aged 18–64 years by the estimated proportion of MSM and by HIV status. Relative risk (RR) and 95% confidence intervals (CIs) of meningococcal disease among MSM were calculated by comparing the annualized incidence of MSM and non-MSM cases. Case-fatality ratios (CFR) were calculated by dividing the number of cases with death reported as the outcome by the total number of cases with known outcome within each category. Data were analyzed using SAS software version 9.3 (SAS Institute, Cary, North Carolina).
Ethical Considerations
All data were de-identified prior to transmission and analysis at CDC. This evaluation was determined to be public health practice and exempted from full review by the CDC Institutional Review Board.
RESULTS
Demographics, Surveillance, and Clinical Characteristics
Between 1 January 2012, and 30 June 2015, 527 cases of meningococcal disease in men aged 18–64 years were reported through NNDSS. Among these, 74 (14.0%) were identified as occurring in MSM, with 46 (62.6%) cases among MSM reported in jurisdictions that experienced a cluster of meningococcal disease: 23 cases in NYC, 14 in LAC, and 9 in Chicago. Twenty-eight sporadic cases among MSM were reported from 19 states. The remaining 453 cases (86.0%) were reported among non-MSM from 46 states and the District of Columbia (Figure 1).
Figure 1.
Number and proportion of meningococcal disease cases by month among men who have sex with men (MSM) and men not known to be MSM (non-MSM) aged 18–64 years, January 2012–June 2015. Abbreviations: LAC, Los Angeles County; MSM, men who have sex with men; NYC, New York City.
The largest proportion of meningococcal disease cases reported as MSM occurred among men aged 26–35 years (43.2%) and for non-MSM cases among men aged 18–25 years (31.4%). Race and ethnicity among meningococcal disease patients varied by jurisdiction (Table 1). Among infections with a known serogroup, N. meningitidis serogroup C accounted for 62 of 73 (84.9%) infections in MSM compared to 98 of 381 (25.7%) infections in non-MSM patients. HIV infection was reported in 38 of 64 (59.4%) MSM with known status. No data on CD4 count or viral load were available. While MSM cases overall had higher CFRs than non-MSM cases (32.4% vs 23.5%), the difference was not statistically significant (P = .112) (Table 1). No association between HIV status and fatal outcome (P = .558) was observed among MSM.
Table 1.
Demographic and Clinical Characteristics of Men Who Have Sex With Men (MSM) and Men Not Known to Be MSM Aged 18–64 Years With Meningococcal Disease, January 2012–June 2015
| Characteristic | MSM | Non-MSM (n = 453) | P Valuea | ||||
|---|---|---|---|---|---|---|---|
|
| |||||||
| Sporadic (n = 28) | New York City (n = 23) | LAC (n = 14) | Chicago (n = 9) | All MSM (n = 74) | |||
| Age group, y | |||||||
| 18–25 | 5 (17.9) | 7 (30.4) | 2 (14.3) | 1 (11.1) | 15 (20.3) | 142 (31.4) | <.001 |
| 26–35 | 11 (39.3) | 10 (43.5) | 8 (57.1) | 3 (33.3) | 32 (43.2) | 99 (21.8) | |
| 36–45 | 5 (17.8) | 3 (13.0) | 2 (14.3) | 4 (44.4) | 14 (18.9) | 61 (13.5) | |
| 46–55 | 7 (25.0) | 2 (8.7) | 2 (14.3) | 1 (11.1) | 12 (16.2) | 87 (19.2) | |
| 56–64 | 0 (0.0) | 1 (4.4) | 0 (0.0) | 0 (0.0) | 1 (1.4) | 64 (14.1) | |
|
| |||||||
| Ethnicity | |||||||
| Hispanic | 9 (32.1) | 3 (13.0) | 7 (50.0) | 1 (11.1) | 20 (27.0) | 82 (18.1) | .003 |
| Non-Hispanic | 19 (67.9) | 16 (69.6) | 7 (50.0) | 8 (88.9) | 50 (67.6) | 276 (60.9) | |
| Unknown | 0 (0.0) | 4 (17.4) | 0 (0.0) | 0 (0.0) | 4 (5.4) | 95 (21.0) | |
|
| |||||||
| Race | |||||||
| White | 19 (67.9) | 6 (26.1) | 9 (64.3) | 3 (33.3) | 37 (50.0) | 268 (59.2) | <.001 |
| Black | 5 (17.8) | 15 (65.2) | 3 (21.4) | 6 (66.7) | 29 (39.2) | 92 (20.3) | |
| Asian | 1 (3.6) | 0 (0.0) | 1 (7.1) | 0 (0.0) | 2 (2.7) | 5 (1.1) | |
| Other | 1 (3.6) | 2 (8.7) | 0 (0.0) | 0 (0.0) | 3 (4.1) | 19 (4.2) | |
| Unknown | 2 (7.1) | 0 (0.0) | 1 (7.1) | 0 (0.0) | 3 (4.1) | 69 (15.2) | |
|
| |||||||
| Type of infectionb | |||||||
| Bacteremia | 16 (57.1) | 12 (52.1) | 2 (14.3) | 3 (33.3) | 33 (44.6) | 93 (20.5) | NA |
| Meningitis | 13 (46.4) | 14 (60.9) | 5 (35.7) | 5 (55.6) | 37 (50.0) | 86 (19.0) | |
| Other | 6 (21.4) | 4 (17.4) | 3 (21.4) | 1 (11.1) | 13 (17.6) | 57 (12.6) | |
| Unknown | 2 (7.1) | 0 (0.0) | 5 (35.7) | 0 (0.0) | 3 (4.1) | 260 (57.4) | |
|
| |||||||
| Deaths | 11 (39.3) | 6 (26.1) | 5 (35.7) | 2 (22.2) | 24 (32.4) | 90 (23.5) | .112 |
|
| |||||||
| Serogroup | |||||||
| B | 1 (3.6) | 1 (4.3) | 3 (21.4) | 0 (0.0) | 5 (6.8) | 125 (27.6) | <.001 |
| C | 21 (75.0) | 22 (95.7) | 10 (71.4) | 9 (100.0) | 62 (83.8) | 98 (21.6) | |
| W | 2 (7.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 2 (2.7) | 51 (11.3) | |
| Y | 2 (7.1) | 0 (0.0) | 1 (7.1) | 0 (0.0) | 3 (4.1) | 75 (16.6) | |
| Other | 1 (3.6) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (1.4) | 32 (7.1) | |
| Unknown | 1 (3.6) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (1.4) | 72 (15.9) | |
|
| |||||||
| HIV status | |||||||
| Infected | 13 (46.4) | 15 (65.2) | 5 (35.7) | 5 (55.6) | 38 (51.4) | Data not available | NA |
| Uninfected | 7 (25.0) | 8 (34.8) | 7 (50.0) | 4 (44.4) | 26 (35.1) | ||
| Unknown | 8 (28.6) | 0 (0.0) | 2 (14.3) | 0 (0.0) | 10 (13.5) | ||
Data are presented as No. (%).
Abbreviations: HIV, human immunodeficiency virus; LAC, Los Angeles County; MSM, men who have sex with men; NA, not available; non-MSM, not known to be men who have sex with men.
P value of All MSM vs non-MSM categories.
Categories are not mutually exclusive; sum of percentages may exceed 100%.
Among all MSM with available information, 20 of 63 (31.7%) patients reported smoking and 26 of 53 (49.1%) reported use of recreational drugs including marijuana. Of the 46 cluster-associated MSM cases from NYC, LAC, and Chicago, 31 patients had sexual partnership data available, of whom 14 (45.2%) reported multiple sex partners or engagement in anonymous sex. Of the 32 cluster-associated cases from NYC and Chicago, 23 had data available related to the use of online dating applications or websites to meet partners; 14 of 23 (60.9%) patients reported using these methods.
Genomic Characterization and Diversity of the Isolates
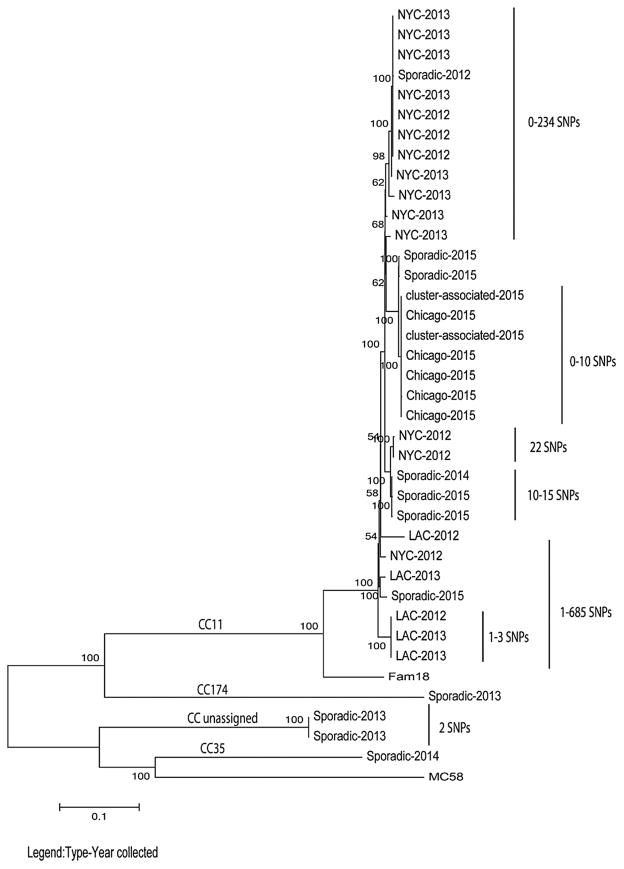
Whole-genome sequencing analysis was performed to assess the genetic relatedness of 37 isolates from cluster and sporadic cases. All 24 isolates from cluster-associated cases in Chicago (n = 5), NYC (n = 14), and LAC (n = 5) belonged to sequence type (ST) 11 and clonal complex (CC) 11. Of the 13 sporadic isolates, there were 9 ST11/CC11, 2 ST4221/CC unassigned, 1 ST1679/CC35, and 1 ST1466/CC174 (Table 2). The Chicago isolates formed a distinct clade in the phylogenetic tree, with 0–10 core SNPs difference out of the 13 026 core SNPs identified among the 37 isolates. All but 3 NYC isolates clustered together in 1 phylogenetic group, with 0–234 SNPs difference between the isolates within the phylogenetic group and 0–337 SNPs difference between all NYC isolates. Three isolates from LAC are very closely related to each other, with 1–3 SNPs difference, while the other 2 LAC isolates are more distantly related, with 1–685 SNPs difference between all LAC isolates (Figure 2). Nine CC11 isolates from the sporadic cases were either in one of the clades associated with MSM clusters or closely related to these clades. All non-CC11 isolates formed distinct phylogenetic groups. The SNP difference among the sporadic isolates ranged from 2 to 7256 SNPs.
Table 2.
Molecular Profile of Selected Available Isolates From Clustered and Sporadic Meningococcal Cases Among Men Who Have Sex With Men Aged 18–64 Years, January 2012–June 2015
| Origin of Case | No. | Serogroup | ST | CC | PorA | PorB | FetA |
|---|---|---|---|---|---|---|---|
| Chicago | 5 | C | 11 | CC11 | P1.18-1,3 | 2–2 | 3–6 |
| New York City | 11 | C | 11 | CC11 | P1.5-1,10-8 | 2–2 | 3–6 |
| New York City | 1 | C | 11 | CC11 | P1.5-1,10-8 | 2–75 | 3–6 |
| New York City | 1 | C | 11 | CC11 | P1.5-1,10-8 | 2–223 | 3–6 |
| New York City | 1 | C | 11 | CC11 | P1.5-1,10-1 | 2–2 | NA |
| Los Angeles County | 5 | C | 11 | CC11 | P1.5-1,10-8 | 2–2 | 3–6 |
| Sporadic | 1 | C | 11 | CC11 | P1.5-1,10-8 | 2–78 | 3–6 |
| Sporadic | 2 | B | 4221 | Unassigned | P1.22,14 | 3–36 | 5–148 |
| Sporadic | 2 | C | 11 | CC11 | P1.5-1,10-8 | 2–210 | 3–6 |
| Sporadic | 1 | C | 11 | CC11 | P1.5-1,10-8 | 2–2 | 3–6 |
| Sporadic | 1 | C | 1679 | CC35 | P.22-1,14 | 3–39 | NA |
| Sporadic | 1 | C | 11 | CC11 | New-10–8 | 2–210 | 3–6 |
| Sporadic | 1 | Y | 1466 | CC174 | P1.21,16 | 3–35 | 3–7 |
| Sporadic | 4 | C | 11 | CC11 | P1.18-1,3 | 2–2 | 3–6 |
Abbreviations: CC, clonal complex; NA, not applicable; ST, sequence type.
Figure 2.
Molecular phylogenetic analysis based on whole-genome sequencing from sporadic and cluster-associated meningococcal isolates among men who have sex with men aged 18–64 years, January 2012–June 2015. The maximum likelihood tree was based on the Tamura-Nei model. The tree is drawn to scale with branch lengths measured in the number of substitution per site. Internal nodes are labeled with bootstrap values (500 iterations). The scale bar is based on the 13 026 positions in the core single-nucleotide polymorphism (SNP) matrix. Labels represent the location of the outbreak and the year collected, FAM18 and MC58 as outgroups. A distance scale bar is shown at the bottom left. *Cluster-associated cases refers to 2 cases epidemiologically linked to the Chicago cluster who were not residents of Chicago. Abbreviations: LAC, Los Angeles County; NYC, New York City.
Relative Risk for Meningococcal Disease Among Men Who Have Sex With Men
The annualized incidence of meningococcal disease among MSM overall was 0.56 cases per 100 000 persons compared to 0.14 cases per 100 000 persons among all non-MSM, for a relative risk (RR) of 4.0 (95% CI, 3.1–5.1; P < .001) (Table 3). In areas outside of where meningococcal disease clusters occurred, MSM had 1.9 (95% CI, 1.3–2.8; P < .001) times the risk of meningococcal disease compared to non-MSM, while the risk of meningococcal disease among MSM was 14.6–31.8 times that of non-MSM in jurisdictions where clusters occurred (Table 3). HIV-infected MSM had 10.1 times (95% CI, 6.1–16.6; P < .001) the risk of meningococcal disease compared to HIV-uninfected MSM, with the highest RR occurring in areas where no disease cluster was identified (RR, 12.7; 95% CI, 5.1–31.9; P < .001) (Table 4). Though HIV status of non-MSM is unknown, when HIV-uninfected MSM were compared to non-MSM, HIV-uninfected MSM had 1.6 times (95% CI, 1.1–2.4; P = .016) the risk of meningococcal disease. However, when stratified by jurisdictional area, this increase was observed primarily in jurisdictions where clusters were identified (RR, 6.7–18.3), compared with RR of 0.6 (95% CI, .3–1.2) in jurisdictions where no clusters were identified (Table 3).
Table 3.
Annualized Meningococcal Disease Incidence Rate and Relative Risk Among Men Who Have Sex With Men (MSM) and Men Not Known to Be MSM Aged 18–64 Years, January 2012–June 2015
| Category | Cases, No. | Estimated Population, No. | Annualized Incidence Rate per 100 000 | Relative Risk (95% CI) | P Value |
|---|---|---|---|---|---|
| Overall estimates | |||||
| Non-MSM | 453 | 92 849 904 | 0.14 | Reference | |
| MSM | 74 | 3 768 102 | 0.56 | 4.0 (3.1–5.1) | <.001 |
| HIV-uninfected MSM | 26 | 3 290 927 | 0.23 | 1.6 (1.1–2.4) | .016 |
| HIV-infected MSM | 38 | 477 175 | 2.28 | 16.3 (11.7–22.7) | <.001 |
|
| |||||
| Sporadic | |||||
| Non-MSM | 417 | 86 940 086 | 0.14 | Reference | |
| MSM | 28 | 3 040 442 | 0.26 | 1.9 (1.3–2.8) | <.001 |
| HIV-uninfected MSMa | 7 | 2 652 873 | 0.08 | 0.6 (.3–1.2) | .112 |
| HIV-infected MSMa | 13 | 387 569 | 0.96 | 7.0 (4.0–12.1) | <.001 |
|
| |||||
| New York City | |||||
| Non-MSM | 19 | 2 400 680 | 0.23 | Reference | |
| MSM | 23 | 191 738 | 3.43 | 15.2 (8.3–27.8) | <.001 |
| HIV-uninfected MSM | 8 | 150 097 | 1.52 | 6.7 (2.9–15.4) | <.001 |
| HIV-infected MSM | 15 | 41 641 | 10.29 | 45.5 (23.1–89.6) | <.001 |
|
| |||||
| Los Angeles County | |||||
| Non-MSM | 13 | 2 947 624 | 0.13 | Reference | |
| MSM | 14 | 217 120 | 1.84 | 14.6 (6.8–31.1) | <.001 |
| HIV-uninfected MSMb | 7 | 182 339 | 1.10 | 8.7 (3.5–21.8) | <.001 |
| HIV-infected MSMb | 5 | 34 781 | 4.11 | 32.6 (11.6–91.4) | <.001 |
|
| |||||
| Chicago | |||||
| Non-MSM | 4 | 822 145 | 0.14 | Reference | |
| MSM | 9 | 58 171 | 4.42 | 31.8 (9.8–103.3) | <.001 |
| HIV-uninfected MSM | 4 | 44 987 | 2.54 | 18.3 (4.6–73.1) | <.001 |
| HIV-infected MSM | 5 | 13 184 | 10.84 | 77.9 (20.9–290.3) | <.001 |
Abbreviations: CI, confidence interval; HIV, human immunodeficiency virus; MSM, men who have sex with men; non-MSM, not known to be men who have sex with men.
HIV status available in 20 of 28 cases.
HIV status available in 12 of 14 cases.
Table 4.
Annualized Meningococcal Disease Incidence Rate and Relative Risk Among Human Immunodeficiency Virus (HIV)–Infected Men Who Have Sex With Men (MSM) and HIV-Uninfected MSM Aged 18–64 Years, January 2012–June 2015
| Category | Cases, No. | Estimated Population, No. | Annualized Incidence Rate per 100 000 | Relative Risk (95% CI) | P Value |
|---|---|---|---|---|---|
| Overall estimatesa | |||||
| HIV-uninfected MSM | 26 | 3 290 927 | 0.23 | Reference | |
| HIV-infected MSM | 38 | 477 175 | 2.28 | 10.1 (6.1–16.6) | <.001 |
|
| |||||
| Sporadicb | |||||
| HIV-uninfected MSM | 7 | 2 652 873 | 0.08 | Reference | |
| HIV-infected MSM | 13 | 387 569 | 0.96 | 12.7 (5.1–31.9) | <.001 |
|
| |||||
| New York City | |||||
| HIV-uninfected MSM | 8 | 150 097 | 1.52 | Reference | |
| HIV-infected MSM | 15 | 41 641 | 10.29 | 6.8 (2.9–15.9) | <.001 |
|
| |||||
| Los Angeles Countyc | |||||
| HIV-uninfected MSM | 7 | 182 339 | 1.10 | Reference | |
| HIV-infected MSM | 5 | 34 781 | 4.11 | 3.7 (1.2–11.8) | .031 |
|
| |||||
| Chicago | |||||
| HIV-uninfected MSM | 4 | 44 987 | 2.54 | Reference | |
| HIV-infected MSM | 5 | 13 184 | 10.84 | 4.3 (1.1–15.9) | .033 |
Abbreviations: CI, confidence interval; HIV, human immunodeficiency virus; MSM, men who have sex with men.
Among 64 cases with known HIV status.
Among 28 cases with known HIV status.
Among 12 cases with known HIV status.
DISCUSSION
Since the first reported outbreak of meningococcal disease among MSM in 2001 in Toronto, several outbreaks or clusters have been reported among MSM in Europe and the United States [3, 20, 21]. Determining the need for routine meningococcal vaccination of MSM in nonoutbreak settings has been challenging given the lack of data on the burden and risk of disease in this population in the United States. In this evaluation, we demonstrate that although the incidence is low, the relative risk of meningococcal disease is higher among MSM than the non-MSM male population in the United States, with HIV infection identified as a likely factor for this increased risk observed among MSM in sporadic settings. Additionally, the majority of cases among MSM are due to serogroup C and occur among age groups not currently recommended to receive quadrivalent meningococcal conjugate vaccine through the routine adolescent program.
Evaluations in the United States, England, and South Africa demonstrate relative risks of 4.5–24.0 for meningococcal disease among HIV-infected compared to HIV-uninfected persons, with the highest risk in persons with high viral load or low CD4 count [22–24]. Disentangling the independent effects of HIV infection and being MSM on the risk of meningococcal disease is challenging, given the lack of data on HIV status of non-MSM. In addition, 59% of MSM in our evaluation were HIV-infected, compared to a national HIV prevalence of 21.6% among MSM [25]. However, given that the risk of meningococcal disease in the sporadic setting is elevated only among HIV-infected MSM, HIV may be an important driver for increased risk of disease among MSM in settings where clusters of disease have not been reported. On the other hand, in meningococcal disease clusters, risk was elevated for both HIV-infected and HIV-uninfected MSM, suggesting that additional factors may be involved in the occurrence of meningococcal disease clusters among MSM.
The sex of a meningococcal disease patient’s sex partners is likely a proxy for other unmeasured risk factors contributing to the development of meningococcal disease clusters among MSM. In the NYC and Chicago clusters, for example, the majority of cases among MSM were black and geographically clustered in certain sections of the cities, suggesting that increased transmission occurred among common social networks. Additionally, in this evaluation, a high prevalence of behaviors were reported that may favor transmission of, or increased susceptibility to, N. meningitidis in this population. Among MSM patients with known information, 31.7% smoke tobacco, compared to 17.8% of adults in the United States [26]. Nearly half (48.1%) of MSM patients reported use of recreational drugs, including marijuana, compared to the US average of 10.2% [27]. Among cluster-associated cases, a high proportion of patients reported sexual activity with multiple and/or anonymous partners or use of online dating applications or websites to find sex partners. However, the lack of a comparison group limits the ability to determine risk factors for meningococcal disease among MSM and identify MSM subgroups at greatest risk. No known clusters of meningococcal disease have occurred among other sexual minority groups, such as women who have sex with women, in the United States. Additional evaluations to identify risk factors will be necessary to inform preventative strategies among MSM.
A high CFR (32% overall) was observed among MSM, although it was not statistically different from that of non-MSM. While reports from the literature in the general population are mixed, with lower meningococcal disease CFR among HIV-infected persons compared with HIV-uninfected persons in the United States and United Kingdom [22, 24], and higher CFR among HIV-infected compared with HIV-uninfected persons in South Africa [23], we did not observe an association between HIV infection and case outcome among MSM. The high CFR among MSM in our evaluation may be related to the large proportion of cases due to serogroup C ST11, which is known to be a more virulent strain [28, 29].
All reported meningococcal clusters among MSM globally to date have been due to serogroup C ST11/CC11 [3, 20, 21]. Molecular characterization suggests that the strains responsible for clusters of meningococcal disease among MSM reported in this analysis may have come from a common ancestor and further diverged over time under different environmental conditions. Additional evaluations of carriage and disease-causing strains among MSM populations using whole-genome sequencing–based methods are important to understand disease transmission and the molecular epidemiology of meningococcal disease in this population.
Several considerations regarding the estimated risk and risk factors for meningococcal disease among MSM should be noted. First, ascertainment of the sex of sex partners of meningococcal disease patients during case investigations is not systematic across state health departments and is dependent on disclosure by the patient. Thus, the risk of meningococcal disease among MSM may be underestimated in this evaluation due to misclassification. Likewise, HIV status and behavioral risk factors are not uniformly collected during case investigations, limiting the completeness of this information and thus the ability to identify subgroups at greatest risk. Additionally, meningococcal disease incidence estimates among MSM and by HIV status rely on the accuracy of the denominators available. While jurisdiction-specific estimates were used where meningococcal disease clusters occurred, the denominator for sporadic cases outside of these jurisdictions was based on a standard proportion of MSM, whereas true proportions of MSM among the male population likely vary considerably at the local level.
The increased risk of meningococcal disease among HIV-infected MSM in this evaluation provides further support for the recent ACIP recommendation for routine vaccination of all HIV-infected persons aged ≥2 months with a quadrivalent meningococcal conjugate vaccine. While HIV-uninfected MSM have a 1.6-fold increased risk compared to non-MSM, this increased risk is lower than that observed among other at-risk groups in whom routine quadrivalent meningococcal conjugate vaccine is recommended, such as persons with asplenia or complement component deficiencies, who have up to a 10 000-fold increased risk for meningococcal disease [1]. As routine vaccination of MSM with a quadrivalent meningococcal vaccine is not currently recommended by ACIP, high uptake of meningococcal disease among HIV-infected persons offers an opportunity to also potentially impact the risk of meningococcal disease among the MSM population as a whole.
Although rare, cases and clusters of meningococcal disease among MSM continue to occur in the United States. While this evaluation demonstrates the increased risk of disease among MSM, further study is needed to better understand transmission and risk factors in this population to inform public health prevention and response strategies. Ascertainment of HIV status of all meningococcal disease patients and the sex of sex partners of patients aged ≥16 years will be important to understand the true burden of disease in this population as well as to rapidly detect meningococcal disease clusters among MSM at the local level. In the meantime, clinicians and public health authorities should continue to increase awareness of meningococcal disease among MSM, identify vaccination strategies and other public health response measures to protect MSM during outbreaks of meningococcal disease in this population, and encourage routine vaccination of all HIV-infected MSM with a quadrivalent meningococcal conjugate vaccine.
Acknowledgments
Special thanks to the staff from state and local health departments who provided supplementary data and isolates from meningococcal cases for this evaluation, in particular Paula Del Rosso and Don Weiss from the New York City Department of Health and Mental Hygiene, and Jennifer Kyle, Rosie Glenn-Finer, and Shrimati Datta from the California Department of Public Health.
Financial support. This work was supported by the CDC.
Footnotes
Disclaimer. The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention (CDC).
Potential conflicts of interest. All authors: No potential conflicts of interest. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1.Cohn AC, MacNeil JR, Clark TA, et al. Centers for Disease Control and Prevention (CDC) Prevention and control of meningococcal disease: recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 2013;62:1–28. [PubMed] [Google Scholar]
- 2.Thigpen MC, Whitney CG, Messonnier NE, et al. Bacterial meningitis in the United States, 1998–2007. N Engl J Med. 2011;364:2016–25. doi: 10.1056/NEJMoa1005384. [DOI] [PubMed] [Google Scholar]
- 3.Kamiya H, MacNeil J, Blain A, et al. Meningococcal disease among men who have sex with men—United States, January 2012–June 2015. MMWR Morb Mortal Wkly Rep. 2015;64:1256–7. doi: 10.15585/mmwr.mm6444a6. [DOI] [PubMed] [Google Scholar]
- 4.Aubert L, Taha M, Boo N, et al. Serogroup C invasive meningococcal disease among men who have sex with men and in gay-oriented social venues in the Paris region: July 2013 to December 2014. Euro Surveill. 2015:20. doi: 10.2807/1560-7917.es2015.20.3.21016. [DOI] [PubMed] [Google Scholar]
- 5.Marcus U, Vogel U, Schubert A, et al. A cluster of invasive meningococcal disease in young men who have sex with men in Berlin. Euro Surveill. 2013;18 doi: 10.2807/1560-7917.es2013.18.28.20523. pii:20523. [DOI] [PubMed] [Google Scholar]
- 6.MacNeil JR, Rubin LG, Patton M, Ortega-Sanchez IR, Martin SW. Recommendations for use of meningococcal conjugate vaccines in HIV-infected persons—Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep. 2016;65:1189–94. doi: 10.15585/mmwr.mm6543a3. [DOI] [PubMed] [Google Scholar]
- 7.Centers for Disease Control and Prevention. Case definitions for infectious conditions under public health surveillance. MMWR Recomm Rep. 1997:46. [PubMed] [Google Scholar]
- 8.Kretz CB, Retchless AC, Sidikou F, et al. Whole genome characterization of the emerging epidemic meningococcal serogroup C and resurgence of serogroup W in Niger, 2015. Emerg Infect Dis. 2016 doi: 10.3201/eid2210.160468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jolley KA, Maiden MC. BIGSdb: Scalable analysis of bacterial genome variation at the population level. BMC Bioinformatics. 2010;11:1–11. doi: 10.1186/1471-2105-11-595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bambini S, De Chiara M, Muzzi A, et al. Neisseria adhesin a variation and revised nomenclature scheme. Clin Vaccine Immunol. 2014;21:966–71. doi: 10.1128/CVI.00825-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gardner SN, Hall BG. When whole-genome alignments just won’t work: kSNP v2 software for alignment-free SNP discovery and phylogenetics of hundreds of microbial genomes. PLoS One. 2013;8:e81760. doi: 10.1371/journal.pone.0081760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: molecular evolutionary genetics analysis version 6. 0. Mol Biol Evol. 2013;30:2725–9. doi: 10.1093/molbev/mst197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.United States Census Bureau. American Community Survey (ACS) [Google Scholar]
- 14.Purcell DW, Johnson CH, Lansky A, et al. Estimating the population size of men who have sex with men in the United States to obtain HIV and syphilis rates. Open AIDS J. 2012;6:98–107. doi: 10.2174/1874613601206010098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Grey JA, Bernstein KT, Sullivan PS, et al. Estimating the population sizes of men who have sex with men in US States and counties using data from the American Community Survey. JMIR Public Health Surveill. 2016;2:e14. doi: 10.2196/publichealth.5365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.New York City Department of Health and Mental Hygiene. HIV surveillance annual report. New York: NYC DOHMH; 2012. [Google Scholar]
- 17.Chicago Department of Public Health. HIV/STI Surveillance Report. Chicago, IL: City of Chicago; 2014. [Google Scholar]
- 18.Division of HIV and STD Programs LAC, Department of Public Health. 2012 Annual HIV surveillance report. Los Angeles, CA: 2013. Available at: http://publichealth.lacounty.gov/wwwfiles/ph/hae/hiv/2012AnnualHIVSurveillanceReport.pdf. [Google Scholar]
- 19.Centers for Disease Control and Prevention. HIV surveillance report, 2013. Atlanta, GA: CDC; 2015. Available at: http://www.cdc.gov/hiv/pdf/library/reports/surveillance/cdc-hiv-surveillance-report-vol-25.pdf. [Google Scholar]
- 20.European Centre for Disease Prevention and Control. Rapid risk assessment. Stockholm: ECDC; 2013. Invasive meningococcal disease among men who have sex with men. [Google Scholar]
- 21.Nanduri S, Foo C, Ngo V, et al. Outbreak of serogroup C meningococcal disease primarily affecting men who have sex with men—Southern California, 2016. MMWR Morb Mortal Wkly Rep. 2016 doi: 10.15585/mmwr.mm6535e1. [DOI] [PubMed] [Google Scholar]
- 22.Miller L, Arakaki L, Ramautar A, et al. Elevated risk for invasive meningococcal disease among persons with HIV. Ann Intern Med. 2014;160:30–7. doi: 10.7326/0003-4819-160-1-201401070-00731. [DOI] [PubMed] [Google Scholar]
- 23.Cohen C, Singh E, Wu HM, et al. Group for Enteric, Respiratory and Meningeal Disease Surveillance in South Africa (GERMS-SA) Increased incidence of meningococcal disease in HIV-infected individuals associated with higher case-fatality ratios in South Africa. AIDS. 2010;24:1351–60. doi: 10.1097/QAD.0b013e32833a2520. [DOI] [PubMed] [Google Scholar]
- 24.Simmons R, Kirwan P, Beebeejaun K, et al. Risk of invasive meningococcal disease in children and adults with HIV in England: a population-based cohort study. BMC Med. 2015;13:297. doi: 10.1186/s12916-015-0538-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Centers for Disease Control and Prevention. HIV infection risk, prevention, and testing behaviors among men who have sex with men—National HIV behavioral surveillance, 20 U.S. cities, 2014. [Accessed 9 August 2016];HIV surveillance special report 15. Available at: https://www.cdc.gov/hiv/pdf/library/reports/surveillance/cdc-hiv-hssr-nhbsmsm-2014.pdf.
- 26.Jamal A, Agaku I, O’Connor E, King B, Kenemer J, Neff L. Current cigarette smoking among adults—United States, 2005–2013. Morbidity and Mortality Weekly Report (MMWR) 2014;63:1108–12. [PMC free article] [PubMed] [Google Scholar]
- 27.Center for Behavioral Health Statistics and Quality. Behavioral health trends in the United States: results from the 2014 National Survey on Drug Use and Health. 2015. [Google Scholar]
- 28.Deghmane AE, Parent du Chatelet I, Szatanik M, et al. Emergence of new virulent Neisseria meningitidis serogroup C sequence type 11 isolates in France. J Infect Dis. 2010;202:247–50. doi: 10.1086/653583. [DOI] [PubMed] [Google Scholar]
- 29.Pollard AJ, Ochnio J, Ho M, Callaghan M, Bigham M, Dobsong S. Disease susceptibility to ST11 complex meningococci bearing serogroup C or W135 polysaccharide capsules, North America. Emerg Infect Dis. 2004;10:1812–5. doi: 10.3201/eid1010.040335. [DOI] [PMC free article] [PubMed] [Google Scholar]