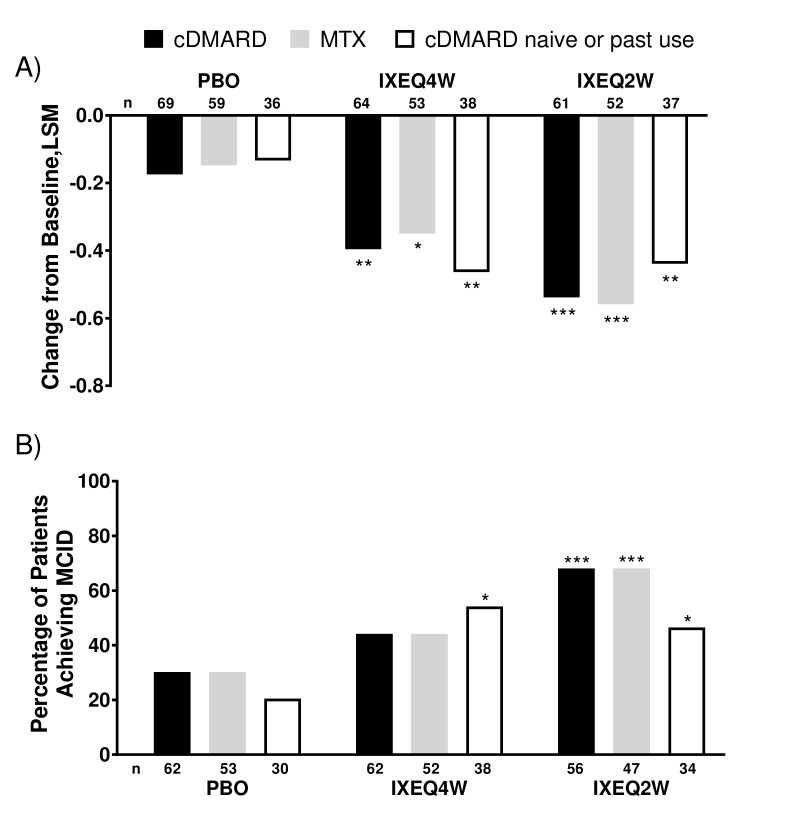
Figure 2.
HAQ-DI change from baseline and proportion of patients achieving MCID after 24 weeks in patients treated with PBO, IXEQ4W or IXEQ2W alone or in combination with cDMARDs or MTX. (A) LSM changes from baseline of HAQ-DI. (B) Proportion of patients achieving MCID. The study was not designed to test equivalence or non-inferiority of ixekizumab alone versus treatment with ixekizumab in combination with cDMARDs. cDMARD, conventional disease-modifying antirheumatic drugs; HAQ-DI, Health Assessment Questionnaire-Disability Index; IXEQ2W, 80 mg ixekizumab once every 2 weeks; IXEQ4W, 80 mg ixekizumab once every 4 weeks; LSM, least squares mean; MCID, minimal clinically important difference; MTX, methotrexate; n, number of patients; PBO, placebo. *P<0.05, **P<0.01, ***P<0.001, all versus PBO.