Abstract
Objective
Postoperative ileus (POI), the most frequent complication after intestinal surgery, depends on dendritic cells (DCs) and macrophages. Here, we have investigated the mechanism that activates these cells and the contribution of the intestinal microbiota for POI induction.
Design
POI was induced by manipulating the intestine of mice, which selectively lack DCs, monocytes or macrophages. The disease severity in the small and large intestine was analysed by determining the distribution of orally applied fluorescein isothiocyanate-dextran and by measuring the excretion time of a retrogradely inserted glass ball. The impact of the microbiota on intestinal peristalsis was evaluated after oral antibiotic treatment.
Results
We found that Cd11c-Cre+ Irf4flox/flox mice lack CD103+CD11b+ DCs, a DC subset unique to the intestine whose function is poorly understood. Their absence in the intestinal muscularis reduced pathogenic inducible nitric oxide synthase (iNOS) production by monocytes and macrophages and ameliorated POI. Pathogenic iNOS was produced in the jejunum by resident Ly6C– macrophages and infiltrating chemokine receptor 2-dependent Ly6C+ monocytes, but in the colon only by the latter demonstrating differential tolerance mechanisms along the intestinal tract. Consistently, depletion of both cell subsets reduced small intestinal POI, whereas the depletion of Ly6C+ monocytes alone was sufficient to prevent large intestinal POI. The differential role of monocytes and macrophages in small and large intestinal POI suggested a potential role of the intestinal microbiota. Indeed, antibiotic treatment reduced iNOS levels and ameliorated POI.
Conclusions
Our findings reveal that CD103+CD11b+ DCs and the intestinal microbiome are a prerequisite for the activation of intestinal monocytes and macrophages and for dysregulating intestinal motility in POI.
Keywords: Postoperative Ileus; Immunity; Microbiota; Intestinal Monocytes and Macrophages, Dendritic cells
Video 1.
Significance of this study.
What is already known on this subject?
Postoperative ileus (POI) is the most frequent complication after intestinal surgery.
The accumulation of dendritic cells and macrophages has been observed in POI.
Intestinal macrophages produce inducible nitric oxide synthase (iNOS) that impairs intestinal peristalsis in POI.
What are the new findings?
Intestinal muscularis CD103+CD11b+ DCs depend on the transcription factor Irf4 and initiate POI by stimulating iNOS production in monocytes and macrophages.
Infiltrating Ly6C+ monocytes and resident Ly6C– macrophages produce iNOS and cause small intestinal POI, whereas only Ly6C+ monocytes induce large intestinal POI.
Antibiotic treatment reduces iNOS and ameliorates POI.
How might it impact on clinical practice in the foreseeable future?
Modulating the intestinal microbiota may be a prophylactic strategy against POI.
Introduction
Intestinal phagocytes, such as macrophages and dendritic cells (DCs), are crucial in maintaining gut homeostasis1–3 and in regulating intestinal motility.4–7 Under homeostasis, exposure to the luminal microbiota does not induce proinflammatory responses,5 because these cells possess a tolerogenic signature.8 However, such conditioning is impaired in acute inflammation, so that these cells acquire a proinflammatory signature and induce intestinal diseases.4 8–11 The most frequent adverse condition after intestinal surgery, postoperative ileus (POI), critically depends on the activation of intestinal phagocytes, such as macrophages and DCs.4 9 12 We have previously shown in a murine model of POI that surgical injury to the intestinal tract caused intestinal DCs to locally produce the proinflammatory mediator interleukin-12 (IL-12), which stimulated memory Th1 cells to produce interferon-γ (IFNγ), which in turn activated macrophages to express inducible nitric oxide synthase (iNOS). Its product NO paralyses intestinal muscle cells, resulting in POI.4 9 12 These findings established the molecular cascade linking intestinal DCs that sense local injury and intestinal macrophages that stop peristalsis. However, the identity of the relevant DCs and macrophages, their individual roles in regulating intestinal peristalsis in POI and the signals that regulate their local activation are unclear.
Intestinal macrophages and DCs express an overlapping pattern of surface molecules, which often hampers definitive conclusions concerning their specific functions. Intestinal DCs are defined by the expression of CD11c, CD103, major histocompatibility complex (MHC) class II and differential expression of CD11b.13 CD103+CD11b– DCs depend on the transcription factors Batf3 and Irf8.14 15 They cross-present antigen to CD8+ T cells,16 induce regulatory T cells and modulate T cell responses.13 DCs expressing CD11b and CD103 develop from the common Flt3L-dependent pre-cDC progenitor17 18 by mechanisms involving the transcription factors Irf4.19 They are unique to the intestinal tract and have been shown to contribute to intestinal immunity.18 20 21 However, their role in intestinal disease is much less understood than that of the other phagocyte subsets.
In contrast to DCs, monocytes and macrophages in the intestine lack CD103 expression and are defined by expression of F4/80, CX3CR1 and CD64.13 22 These cells are further subdivided by Ly6C into Ly6C–CX3CR1high macrophages and Ly6C+CX3CR1low monocytes, which originate from recently recruited Ly6C+ blood monocytes. Some resident Ly6C– macrophages in the liver, the brain and the skin are long-living cells, which are derived from earlier yolk sac and fetal liver progenitors.23–25 They can maintain themselves during life with little contribution of circulating Ly6C+ monocytes.25 26 However, intestinal Ly6C– macrophages have an exceptionally short half-life of 3 weeks27 and are constantly replaced by blood-derived Ly6C+ monocytes.17 18 28 29 Hence, the majority of intestinal monocytes and macrophages represents a continuum of differentiation with an important role in intestinal homeostasis by regulating different functional capabilities in health and disease.7 30 In the present study, we have investigated the different phagocyte subsets in the small and large intestine and their role in POI.
Results
CD103+CD11b+ DCs in the intestinal muscularis depend on Irf4 and are crucial for POI
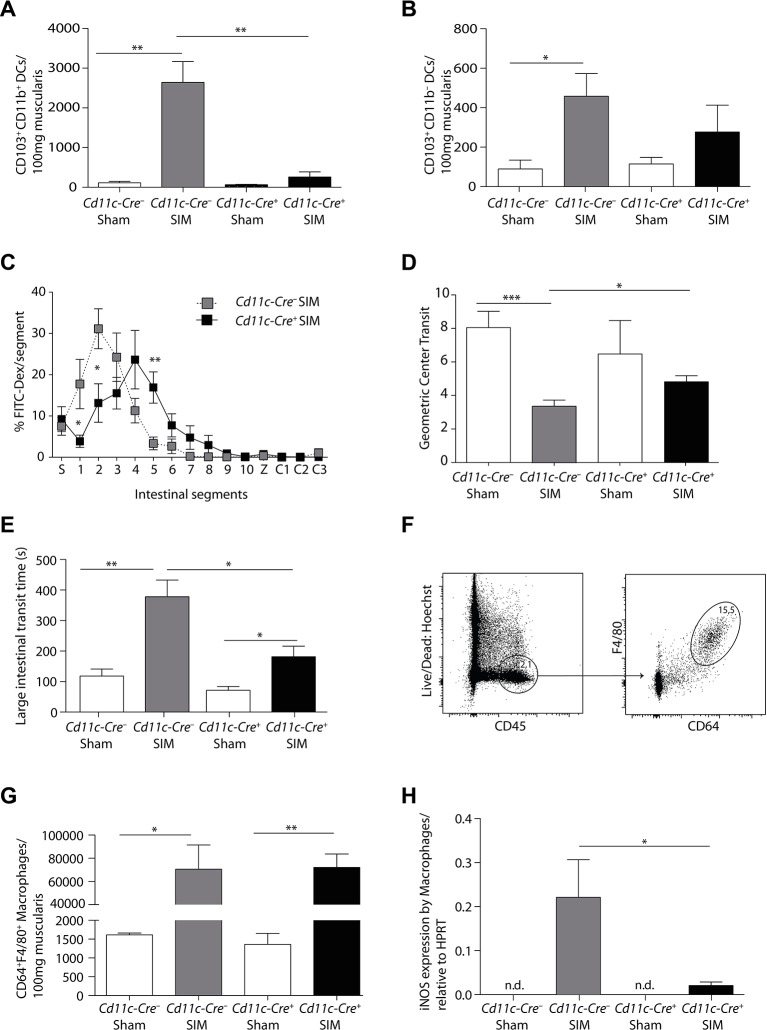
We have previously shown that intestinal CD103+CD11b+ DCs produced IL-12 in POI, which stimulated IFNγ production by pathogenic memory Th1 cells.4 To determine the necessity of this DC subset for POI, we used Cd11c-Cre+ Irf4flox/flox (Cd11c-Cre+) mice, which have been reported to lack these cells in the mesenteric lymph nodes and partially in the lamina propria.19 We first established that these DCs were also absent from the intestinal muscularis after small intestinal manipulation (SIM) (figure 1A, online supplementary figure 1), whereas the minor population of CD103+CD11b– DCs was unchanged (figure 1B). Importantly, both small (figure 1C,D) and large intestinal POI (figure 1E) were reduced in the absence of CD103+CD11b+ DCs, identifying these DCs as critical inducers of POI.
Figure 1.
Postoperative ileus depends on Irf4-dependent CD103+CD11b+ DCs. (A,B) The number of CD103+CD11b+ (A) and CD103+CD11b– DCs (B), pregated on Hoechst–CD45+CD11c+MHCII+ cells, were determined by flow cytometric analysis 24 hours after small intestinal manipulation (SIM) or sham manipulation in Cd11c-Cre– Irf4flox/flox (Cd11c-Cre–) and Cd11c-Cre+ Irf4flox/flox (Cd11c-Cre+) mice (representative data of SIM n=8, sham n=5). (C–E) Analysis of small intestinal (C–D) and large intestinal motility (E) in Cd11c-Cre+ or Cd11c-Cre– mice 24 hours after SIM (FITC-Dex=Fluorescein isothiocyanate-dextran, S=stomach, 1–10=small intestinal segments, Z=caecum, C1-3=large intestinal segments) (SIM n=8, sham n=4). (F) Gating scheme for macrophages in Cd11c-Cre– mice 24 hours after intestinal manipulation. (G) The number of muscularis macrophages (Hoechst–CD45+F4/80+CD64+) in Cd11c-Cre+ or Cd11c-Cre− mice was determined 24 hours after SIM or sham manipulation by flow cytometry (representative data of SIM n=8, sham n=5). (H) CD64+F4/80+ macrophages were isolated by cell sorting 24 hours after SIM or sham. Nos2 (inducible nitric oxide synthase) expression was determined by quantitative PCR and the expression was normalised to Hprt (HPRT) (nd=non-detectable) (n=8 SIM, n=4 sham). Results are given as mean +/-SEM. *p<0.05; **p<0.01;***p<0.001.
gutjnl-2017-313856supp001.jpg (896.7KB, jpg)
CD103+CD11b+ DCs regulate iNOS expression by small intestinal monocytes and macrophages
The expression of iNOS by intestinal macrophages mediates POI by paralysing intestinal myocytes.4 31 To investigate whether CD103+CD11b+ DCs regulate iNOS production, we first enumerated CD64+F4/80+ cells in the muscularis. We found a strong increase of these cells after SIM, suggesting that some of these cells might originate from recruited monocytes (figure 1F,G). Such increase was not reduced in Cd11c-Cre+ mice, which lack CD103+CD11b+ DCs (figure 1G). Therefore, we hypothesised that CD64+F4/80+ cells were unable to produce pathogenic iNOS. Indeed, iNOS expression by CD64+F4/80+ cells was significantly increased after SIM only in Cd11c-Cre– littermates, but not in Cd11c-Cre+ mice (figure 1H). These data indicate that Irf4-dependent CD103+CD11b+ DCs locally activate muscularis monocytes and macrophages to produce iNOS.
Increase of Ly6C+ monocytes in the manipulated small intestine depends on chemokine receptor 2 (CCR2)
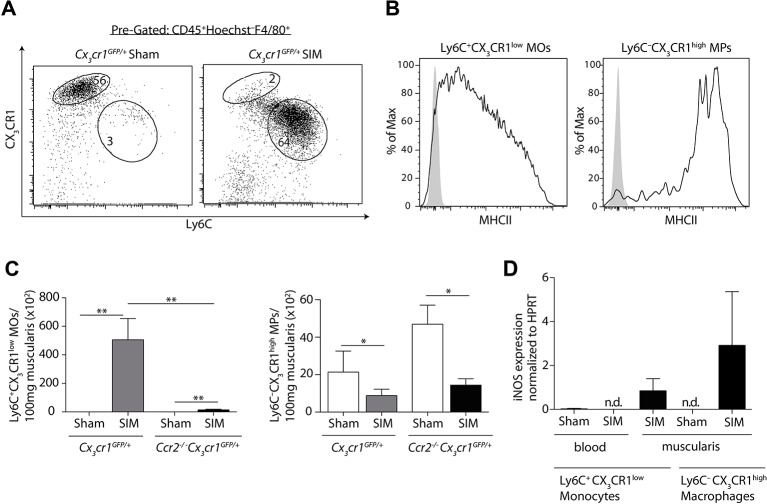
Distinct monocytes and macrophages differing in Ly6C and CX3CR1 expression have been described in the circulation and in the intestine.8 32 33 We asked whether iNOS-producing monocytes or macrophages mediated POI in the intestinal muscularis. The small intestinal muscularis of sham-manipulated mice mainly harboured Ly6C–CX3CR1high macrophages (figure 2A). SIM induced a significant increase of cells expressing high levels of Ly6C, but low levels of CX3CR1 (Ly6C+CX3CR1low) (figure 2A). These cells expressed low levels of MHC class II on their surface (figure 2B), suggesting that these cells originated from recruited Ly6C+blood monocytes. Notably, all CX3CR1low cells expressed CD64 (online supplementary figure 2, middle histogram), indicating that the recently described lamina propria CD64–CD103–CX3CR1low DCs20 21 are not present in the manipulated muscularis. Given that CCR2 is crucial for the abundance of Ly6C+ blood monocytes, the direct progenitor of Ly6C+ monocytes in the muscularis,32 we hypothesised that CCR2 mediates the increase of these cells in POI. To facilitate detection of Ly6C+CX3CR1low monocytes also in mice lacking CCR2, we generated Ccr2–/– Cx3cr1GFP mice. We found that the abundance of Ly6C+ monocytes but not of Ly6C– macrophages was severely decreased after SIM of Ccr2–/– Cx3cr1GFP mice (figure 2C). These data demonstrate that the increased abundance of Ly6C+ monocytes within the manipulated muscularis depends on the recruitment of CCR2-dependent Ly6C+blood monocytes.
Figure 2.
The abundance of inducible nitric oxide synthase (iNOS)-producing Ly6C+ monocytes depends on CCR2. (A) Small intestinal muscularis monocytes (MOs) and macrophages (MPs), pregated on Hoechst–CD45+F4/80+ cells, were analysed for their expression of CX3CR1 and Ly6C 24 hours after small intestinal manipulation (SIM) by flow cytometry. (B) Flow cytometric analysis of major histocompatibility complex (MHC) class II expression on Ly6C+CX3CR1low monocytes (left) and Ly6C–CX3CR1high macrophages (right), pregated on live/dead–CD45+F4/80+, in the muscularis 24 hours after SIM. The solid line represents the MHC class II expression, whereas the grey area indicates control staining (Fluorescence Minus One) (representative data of n=6). (C) The number of Ly6C+CX3CR1low monocytes (left bar graph) and Ly6C–CX3CR1high macrophages (right bar graph) were determined in the muscularis 24 hours after SIM or sham in Cx3cr1GFP/+ and Cx3cr1GFP/+ Ccr2−/− mice (representative data of n=8). (D) Ly6C+CX3CR1low monocytes and Ly6C–CX3CR1high macrophages from the small intestinal muscularis and from the blood were isolated by cell sorting 24 hours after SIM or sham. Nos2 (iNOS) expression was determined by quantitative PCR and the expression was normalised to Hprt (HPRT) (representative data of blood n=8, muscularis n=8) (nd = not detectable). Results are given as mean +/− SEM. *p<0.05; **p<0.01.
gutjnl-2017-313856supp002.jpg (1.2MB, jpg)
Ly6C– macrophages and Ly6C+ monocytes are both sufficient to induce small intestinal POI
We next asked whether Ly6C+ monocytes and/or Ly6C– macrophages produced pathogenic iNOS in POI. We first examined the progenitors of Ly6C+ muscularis monocytes, namely Ly6C+ blood monocytes. However, we were unable to detect iNOS expression in blood monocytes of manipulated or sham-operated mice (figure 2D). In contrast, Ly6C+ monocytes as well as Ly6C– macrophages within the small intestinal muscularis upregulated iNOS after SIM, indicating their local activation (figure 2D).
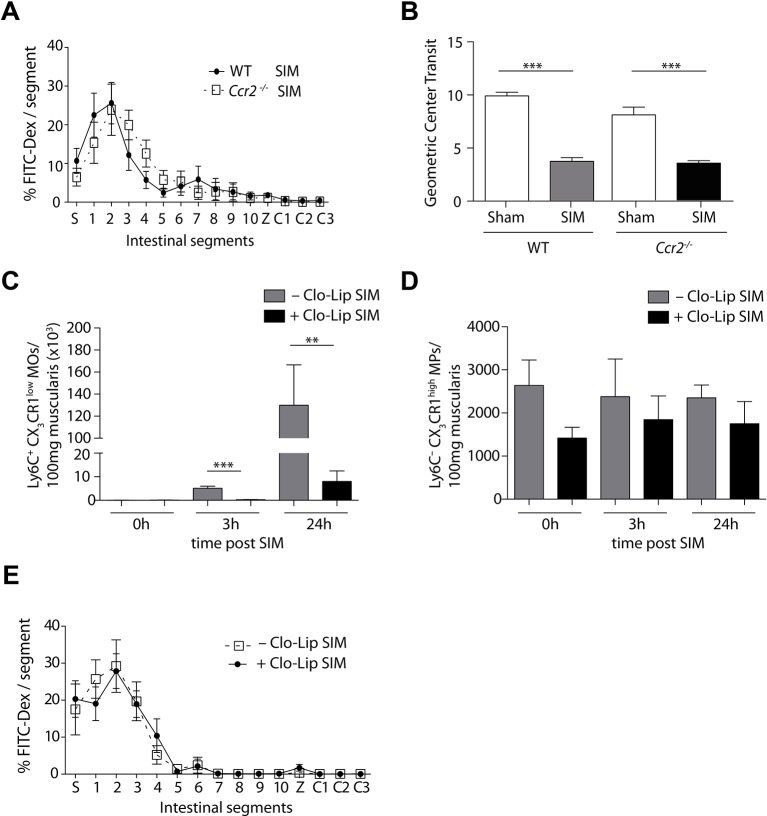
Next, we analysed the requirement of these iNOS-producing Ly6C– macrophages and Ly6C+ monocytes for POI by selective targeting. To target Ly6C+ monocytes, we again employed Ccr2–/– mice and found that these mice were not protected from small intestinal POI (figure 3A,B). To confirm these data, we conditionally depleted Ly6C+ monocytes by clodronate liposomes. We found a strong reduction of Ly6C+ monocytes, whereas the number of Ly6C– macrophages and CD103+CD11b+ DCs was not reduced at various time points after depletion (figure 3C,D, online supplementary figure 3). However, depletion of Ly6C+ monocytes did not reduce POI (figure 3E), confirming the findings in CCR2-deficient animals (figure 3A,B).
Figure 3.
Selective depletion of Ly6C+ monocytes does not ameliorate small intestinal POI. (A,B) Measurement of small intestinal motility 24 hours after small intestinal manipulation (SIM) or sham manipulation in Ccr2−/− or C57Bl/6 (WT) mice (representative data of SIM, n=8, sham n=5) (FITC-Dex=Fluorescein isothiocyanate-dextran, S=stomach, 1–10=jejunal segments, Z=caecum, C1-3=colonic segments). (C,D) Flow cytometric analysis of Ly6C+CX3CR1low monocytes (MOs) (C) or Ly6C–CX3CR1high macrophages (MPs) (D), pregated on Hoechst–CD45+F4/80+ cells, in the small intestine of Cx3cr1GFP/+ mice. Mice were treated with clodronate liposome (CloLip) 12 hours before SIM and the number of cells was analysed 3 and 24 hours after SIM and in unmanipulated mice (0 hours) (representative data of n=3–6). (E) One day after SIM, FITC-dextran transit was analysed in CloLip-treated C57Bl/6 mice (n=9) (FITC-Dex=fluorescein isothiocyanate-dextran, S=stomach, 1–10=equal small intestinal segments, Z=caecum, C1-3=equal large intestinal segments). Results are given as mean +/− SEM. *p<0.05; **p<0.01; ***p<0.001.
gutjnl-2017-313856supp003.jpg (377.5KB, jpg)
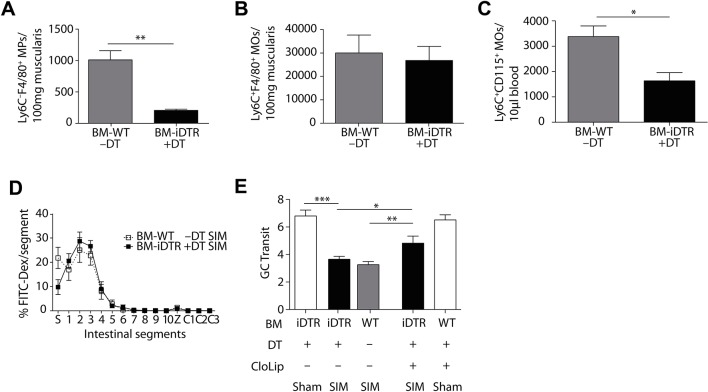
Next, we hypothesised that iNOS by Ly6C– macrophages are crucial for POI induction. As these cells are targeted neither by CCR2 nor clodronate (figures 2C and 3D), we employed Cx3cr1-Cre+ iDtrflox/+ mice based on their high CX3CR1 expression. In contrast to Ly6C–CX3CR1high macrophages in the intestine, this macrophage subset in other organs, such as microglia in the central nervous system (CNS), are radioresistant.24 To avoid depletion of these radioresistant microglia in the CNS, we generated bone marrow chimeric mice to avoid neurological side effects by the depletion of microglia in the CNS. Hence, C57BL/6 recipient mice were engrafted with bone marrow of Cx3cr1-Cre+ iDtrflox/+ mice. We found a reduction of Ly6C– macrophages within the muscularis after administration of diphtheria toxin (DT) into Cx3cr1-Cre+ iDtrflox/+ mice (BM-iDTR) (figure 4A), whereas the number of Ly6C+ monocytes in the muscularis was not changed (figure 4B). Notably, we found a slight reduction of Ly6C+ blood monocytes and of CD103+CD11b+ DCs (figure 4C and online supplementary figure 3). However, depleting Ly6C– macrophages in BM-iDTR mice did not ameliorate small intestinal POI (figure 4D,E). Hence, we speculated that the lack of Ly6C– macrophages might be compensated by Ly6C+ monocytes and vice versa. We therefore combined the depletion of Ly6C– macrophages in chimeric BM-iDTR mice with the conditional depletion of Ly6C+ monocytes by clodronate liposomes. Indeed, depletion of both Ly6C+ monocytes and Ly6C– macrophages markedly reduced POI (figure 4E), confirming that these iNOS-producing cells are sufficient to induce POI within the small intestine.
Figure 4.
Small intestinal postoperative ileus is induced by Ly6C+ monocytes or Ly6C– macrophages. (A–C) Abundance of Ly6C–F4/80+ macrophages (MPs) (A), Ly6C+F4/80+ monocytes (B) and Ly6C+CD115+ monocytes (C) after sham (A) or small intestinal manipulation (SIM) in the small intestinal muscularis (B) or in the blood (C) of Cx3cr1-Cre+ iDtrflox/+ (BM-iDTR) or C57Bl/6 (BM-WT) bone marrow chimeras treated with diphtheria toxin (+DT) or left untreated (–DT) (representative data of n=7). (D,E) Measurement of small intestinal motility 24 hours after SIM or sham in Cx3cr1-Cre+ iDtrflox/+ (BM-iDTR) or C57Bl/6 (BM-WT) mice (SIM n=11, SIM DT and CloLip n=7, sham n=4) (FITC-Dex=Fluorescein isothiocyanate-dextran, S=stomach, 1–10=small intestinal segments, Z=caecum, C1-3=large intestinal segments). GC=geometric centre; CloLip=clodronate liposomes. Results are given as mean +/− SEM. *p<0.05; **p<0.01; ***p<0.001.
Only Ly6C+ monocytes induce large intestinal POI
We have shown previously and in figure 1 that small intestinal POI can spread to non-manipulated large intestinal segments.4 12 Hence, we next investigated the role of Ly6C– macrophages and Ly6C+ monocytes in inducing colonic POI. We found a profound influx of Ly6C+ monocytes into the large intestinal muscularis after SIM (figure 5A). Ly6C+ monocytes expressed low levels of MHC class II on their surface in comparison to Ly6C– macrophages (figure 5B). The increase of Ly6C+ monocytes in the colonic muscularis was dependent on CCR2 (figure 5C), suggesting that these cells originated from Ly6C+blood monocytes. In contrast, the number of Ly6C– macrophages was unaffected by the lack of this chemokine receptor (figure 5D). To investigate the role of Ly6C+ monocytes in inducing large intestinal POI, we employed CCR2-deficient animals. In contrast to the small intestine, the selective lack of Ly6C+ monocytes in CCR2-deficient animals strongly reduced large intestinal POI (figure 5E). Moreover, conditional depletion of Ly6C+ monocytes by clodronate liposomes also reduced POI (online supplementary figure 4A,B). Notably, the number of Ly6C– macrophages was not reduced (online supplementary figure 4C). These data demonstrate that recruited Ly6C+ monocytes are essential for colonic POI, implying that Ly6C– macrophages are dispensable.
Figure 5.
Large intestinal postoperative ileus depends on Ly6C+ monocytes. (A) Flow cytometric analysis of large intestinal muscularis monocytes (MOs) and macrophages (MPs), pregated on Hoechst–CD45+F4/80+ cells, 24 hours after small intestinal manipulation (SIM) in Cx3cr1GFP/+ mice. (B) Flow cytometric analysis of MHC class II expression on Ly6C+CX3CR1low monocytes (left) and Ly6C–CX3CR1high macrophages (right), pregated on live/dead–CD45+F4/80+ cells, in muscularis 24 hours after SIM. The solid line represents the major histocompatibility complex (MHC) class II expression, whereas the grey area indicates control staining (Fluorescence Minus One) (representative data of n=6). (C,D) Numerical analysis of Ly6C+ monocytes (C) and Ly6C– macrophages (D) in large intestinal muscularis 24 hours after SIM in Cx3cr1GFP/+ and Ccr2−/− Cx3cr1GFP/+ mice (representative data of n=8). (E) Large intestinal motility in Ccr2−/− and C57Bl/6 (WT) mice 24 hours after SIM (SIM n=26, sham n=5). (F) Large intestinal transit 24 hours after sham or large intestinal manipulation (LIM) of Ccr2−/− and C57Bl/6 (WT) mice (LIM n=8, WT LIM n=9, sham n=5). (G,H) Large intestinal Ly6C+CX3CR1low monocytes and Ly6C–CX3CR1high macrophages, pregated on Hoechst–CD45+F4/80+cells, were isolated from Cx3cr1GFP/+ mice by cell sorting 24 hours after either LIM (G) or SIM (H) and Nos2 (inducible nitric oxide synthase) expression was determined by quantitative-PCR. Expression was normalised to Hprt (HPRT) (nd=non-detectable) (representative data of n=8). (I) The number of CD103+CD11b+dendritic cells, pregated on Hoechst–CD45+CD11c+MHCII+ cells, were determined by flow cytometric analysis in small and large intestinal muscularis 24 hours after sham or SIM (representative data of SIM n=4, sham n=3). Results are given as mean +/− SEM. *p<0.05; **p<0.01.
gutjnl-2017-313856supp004.jpg (800.2KB, jpg)
Ly6C– macrophages in the large intestine are unable to express iNOS
Next, we wished to clarify why CCR2 deficiency after SIM selectively protected against large intestinal, but not against small intestinal POI. We first hypothesised that this discrepancy occurred, because we had manipulated the small intestine, reasoning that this might have provided additional stimuli, such as the release of Damage Associated Molecular Patterns (DAMPs), which enabled local phagocyte activation in the manipulated small intestine, but not in the distant colon. To test this hypothesis, we changed the site of manipulation and manipulated the large intestine of CCR2-deficient animals. However, the large intestine of CCR2-deficient mice was still protected against POI (figure 5F). Consistently, iNOS was only produced by large intestinal Ly6C+ monocytes but not by Ly6C– macrophages (figure 5G,H). Thus, the site of intestinal manipulation did not decide whether Ly6C– macrophages produced iNOS and caused POI, implying that other mechanisms prevented colonic macrophages from iNOS production. Given that CD103+CD11b+ DCs initiate iNOS production by macrophages (figure 1), we analysed the presence of these cells after manipulation. We found that these cells are absent in the colon (figure 5I), suggesting that the presence of CD103+CD11b+ DCs might be required for iNOS production by Ly6C– macrophages.
POI depends on the intestinal microbiota
Another difference between the small and large intestine is the composition and quantity of the microbiota.34 35 To investigate the role of the microbiota for the activation of CD103+CD11b+ DCs, monocytes and macrophages, and the induction of POI, we depleted the microbiota by antibiotics (online supplementary figure 5). We noted a considerable reduction of IL-12 (figure 6A) and iNOS (figure 6B), suggesting that the activation of DCs, monocytes and macrophages depends on the microbiota. Notably, RNA and protein analyses revealed a strong reduction of IFNγ (figure 6C,D), suggesting that the previously described Th1 response was inhibited.4 We found that mice treated with antibiotics showed a moderate reduction in small intestinal POI (figure 6E,F). However, antibiotic treatment also reduced intestinal peristalsis in sham-operated animals (figure 6F). In the colon, POI was strongly reduced after antibiotic treatment (figure 6G), demonstrating the essential role of the microbiota for POI induction.
Figure 6.
Postoperative ileus depends on the intestinal microbiota. (A–D) The small intestinal muscularis of untreated (–Antibiotics) and antibiotic-treated (+Antibiotics) mice was isolated 24 hours after small intestinal manipulation (SIM). Il12a, Nos2 (iNOS) and Ifng (IFNγ) RNA (A–C) or IFNγ protein (D) expression was analysed by quantitative-PCR (A–C) or ELISA (D) (–Antibiotics SIM n=7 (A) n=4 (B–D), +Antibiotics SIM n=8). (E,F) Measurement of small intestinal motility 24 hours after SIM or sham manipulation (sham) in antibiotic-treated and untreated mice (–Antibiotics sham n=5, –Antibiotics SIM n=18, +Antibiotics sham n=7, +Antibiotics SIM n=22) (FITC-Dex=Fluorescein isothiocyanate-dextran, S=stomach, 1–10=jejunal segments, Z=caecum, C1-3=colonic segments). (G) The large intestinal motility of antibiotic-treated or untreated mice was measured 24 hours after SIM or sham manipulation (sham) (–Antibiotics sham n=5, –Antibiotics SIM n=11, +Antibiotics sham n=7, +Antibiotics SIM n=15). Results are given as mean +/− SEM. *p<0.05; **p<0.01; ***p<0.001.
gutjnl-2017-313856supp005.jpg (1.2MB, jpg)
Discussion
Macrophages and DCs play central roles in intestinal immunity,13 36 in the regulation of intestinal peristalsis and in POI.4 5 9 In the present study, we have identified the intestinal microbiota and Irf4-dependent CD103+CD11b+ DCs as crucial regulators of monocyte and macrophage responses in POI.
Two subsets of intestinal CD103+ DCs with differential expression of CD11b have been defined previously.37 CD103+CD11b– DCs depend on the transcription factors Batf3 and Irf8,14 15 whereas CD103+CD11b+ DCs depend on the transcription factor Irf4.19 38 Consistent with these previous findings, we found that CD103+CD11b+ DCs in the intestinal muscularis depend on Irf4. Lack of these DCs in Cd11c-Cre+ Irf4flox/flox mice ameliorated POI and reduced iNOS production by intestinal macrophages. As iNOS expression depended on IFNγ by Th1 memory cells,4 Irf4-dependent CD103+CD11b+ DCs may be directly involved in the Th1 response and subsequent macrophage activation resulting in POI. These findings identify a novel role of this enigmatic intestinal DC subset in a disease model.
Macrophages are considered crucial in the regulation of intestinal peristalsis5 and in POI.4 9 In particular, iNOS production by these cells has been shown to induce POI by directly inhibiting smooth muscle cells.4 9 31 We found a strong CCR2-dependent increase of Ly6C+ monocytes in the intestine consistent with previous studies in other inflammatory models.8 17 39 These cells resemble recruited Ly6C+ blood monocytes, which can locally be activated to produce proinflammatory mediators.8 10 40 Also in POI, they underwent a functional switch from iNOS-negative blood monocytes to iNOS-producing monocytes in the manipulated intestinal muscularis. These data suggest that local tolerance mechanisms are disabled in POI. Notably, the expression of MHC class II on Ly6C+ monocytes in the small intestine exceeded the expression in the colon, which might be due to signals derived from the local manipulation of the small intestine.
Targeting Ly6C+ monocytes has been suggested for disease management of several inflammatory disorders.10 41 We aimed at achieving a beneficial effect in the initial phase after POI induction by reducing the abundance of recruited iNOS-producing Ly6C+ monocytes. However, lack of these cells in CCR2-deficient mice or their conditional depletion through clodronate liposomes only ameliorated large intestinal, but not small intestinal POI. These findings do not exclude a role of these cells during the resolution phase after POI induction. Further studies are required to delineate the role of muscularis macrophages in tissue repair.
The finding that depletion of Ly6C+ monocytes did not ameliorate small intestinal POI suggested compensatory mechanisms. Indeed, we found that Ly6C– macrophages in the small intestine were also able to produce iNOS after intestinal manipulation, thereby compensating for the lack of Ly6C+ monocytes. In contrast, Ly6C– macrophages in the colon failed to produce iNOS, indicating a differential and tolerant functional state. This finding was corroborated by recent studies demonstrating that Ly6C– macrophages in the colonic intestinal mucosa retain their tolerant and non-inflammatory signature even under inflammatory conditions.8 10 A plausible mechanism that prevents colonic Ly6C– macrophages to produce iNOS is the compositional and quantitative differences between the microbiota in the small intestine and the colon.34 35 The constant exposure to the abundant colonic microbiota inhibits iNOS production by Ly6C– macrophages, in order to avoid inappropriate responses against commensals, also in the context of POI. Another explanation is the regional distribution of CD103+CD11b+ DCs along the intestine. Consistent with previous findings,42 we found that the colonic muscularis hardly harboured CD103+CD11b+ DCs, suggesting that their absence in the colonic muscularis might be causative for the lack of iNOS expression by Ly6C– macrophages.
To analyse the role of Ly6C– macrophages in small intestinal POI, we designed a conditional deletion approach in bone marrow chimeric mice, by reconstituting lethally irradiated mice with bone marrow from Cx3cr1-Cre+ iDtrflox/+ mice (BM-iDTR). Application of DT into these mice depleted Ly6C– macrophages. Notably, we also found a slight reduction of CD103+CD11b+ DCs, which was less pronounced than in the transgenic Cd11c-Cre+ Irf4flox/flox mice. The application of DT was insufficient to reduce POI, suggesting that both, iNOS produced by Ly6C+ monocytes and Ly6C– macrophages, might be sufficient to trigger POI. To test for this possibility, we combined the DT-dependent deletion of Ly6C– macrophages with the conditional deletion of Ly6C+ monocytes by clodronate liposomes. And indeed, the loss of both cell subsets attenuated POI, verifying compensatory roles of Ly6C– macrophages and Ly6C+ monocytes in small intestinal POI.
We next asked whether iNOS production by monocytes and macrophages is regulated by CD103+CD11b+ DCs and by the intestinal microbiota. We found that iNOS production by monocytes and macrophages was severely reduced in the absence of CD103+CD11b+ DCs. This correlative finding does not indicate a direct interaction between these cells, and as described previously other leucocytes are involved in this signalling cascade.4 The activation of CD103+CD11b+ DCs and the attenuation of POI in the absence of these cells raised the question which mechanism activated these DCs. Translocation of microbial products into the intestinal tissue is a well-documented feature in POI43 44 and MyD88-dependent sensing of microbial products induces IL-12 production by DCs.45 46 In support of this hypothesis, POI has explicitly been shown to be attenuated in mice lacking Toll-like receptor signalling,45 a prerequisite for microbial sensing. As CD103+CD11b+ DCs abundantly express Toll-like receptors,46 47 sensing the microbiota, for example, by TLR4 or 5 may activate these DCs and induce POI. Our results do not prove that CD103+CD11b+ DCs directly sense the microbiome. It is also possible that another cell senses the microbiome and then stimulates CD103+CD11b+ DCs to produce IL-12, the mediator that starts the inflammatory cascade leading to POI.4 Answering this question might be possible with the use of mice in which only CD103+CD11b+ DCs lack MyD88. Generating these mice would require discovering a promoter specific for CD103+CD11b+ DCs.
Our present findings identify novel players in POI and thereby open new therapeutic opportunities. While inhibiting pathogenic players like IL-12 or iNOS might result in immunosuppressive side effects, modifying the microbiome in the intestine might represent a promising prophylaxis against POI through preventing the activation of CD103+CD11b+ DCs. Further studies are required to correlate the composition of the microbiota with the development of POI. Analysing the composition of the microbiota by sequencing the microbiome may indicate prognostic markers to identify patients of risk.
In conclusion, our study reveals a novel role of Irf4-dependent CD103+CD11b+ DCs in the intestine and differential requirements of monocytes and macrophages in the regulation of peristalsis along the gastrointestinal tract. We suggest that modifying the microbiome may be a promising prophylaxis against POI, as it might avoid inappropriate activation of CD103+CD11b+ DCs. Future studies are required to explore the clinical potential of this approach.
Materials and methods
Animals
Mice had been backcrossed >10 generations to C57BL/6 and were used at 8–14 weeks of age. Animals were bred and maintained under specific pathogen-free (SPF) conditions at the animal facilities of the University Clinic of Bonn and Essen.
Generation of bone marrow chimeras
Eight hours after lethal irradiation (9Gy) of 6-week-old CD45.1mice, 5*10(6) bone marrow cells were injected into the tail vein of recipient animals. Experiments were performed 8 weeks after bone marrow transfer. Ccr2−/− Cx3cr1GFP/+ were generated by crossing of Ccr2−/−48 with Cx3cr1GFP/GFP mice.49 Cd11c-Cre+ Irf4flox/flox mice were kindly provided by William Agace19 and bred to Cd11c-Cre– Irf4flox/flox mice. Germline deletion was tested as described previously.19 Cd11c-Cre+ Irf4flox/flox and Cd11c-Cre– Irf4flox/flox offspring as respective controls were used for experiments. Although loss of CD103+CD11b+ DCs does not alter commensal microbial communities,50 Cd11c-Cre+ Irf4flox/flox and Cd11c-Cre– Irf4flox/flox mice were cohoused to prevent the development of a different microbiota. Bone marrow chimeras were generated by lethally irradiating CD45.1 mice and reconstitution with Cx3cr1-Cre+ iDtrflox/+ bone marrow, which was kindly provided by Steffen Jung, Rehovot, Israel.
Intestinal manipulation
We performed standardised intestinal manipulation of the small or the large intestine as previously described.4 Briefly, after isoflurane narcosis, the peritoneal cavity was opened by a midline incision, and the jejunum or the colon was placed onto sterile moist gauze and manipulated by moist cotton applicators once from the oral to aboral direction. Then, two layers of running suture closed the laparotomy. Sham-operated mice underwent laparotomy without manipulation.
Functional studies
We measured gastrointestinal fluorescein isothiocyanate (FITC)-dextran transit 24 hours after intestinal manipulation. FITC-dextran was applied via gavage into the stomach. After 90 min, we sacrificed the animals and divided the gastrointestinal tract into 15 pieces (stomach, ten equal parts from the jejunum, caecum and three equal parts of the colon) and measured the FITC-fluorescence intensity in each intestinal segment by fluorometry. We calculated the geometric centre of FITC-dextran ((sum (% FITC-fluorescence per segment × segment number))/100) and displayed the result as bar graphs. The colonic transit measurement was performed 24 hours after intestinal manipulation by insertion of a 2 mm glass ball retrograde 2.5 cm deep into the colon and measuring the excretion time. Additional information on these techniques has been reviewed recently.51
Antibiotic treatment
Antibiotic treatment was performed as previously described.5 Shortly, mice were treated for 1–5 weeks before intestinal manipulation with drinking water containing 1 g/L of streptomycin, metronidazole, vancomycin and ampicillin. Water bottles were protected from light and drinking solution was exchanged every 4 days.
Isolation of intestinal immune cells
We separated the muscularis from the mucosa by slipping 10 cm length sections from the intestine or colon over a glass rod and stripped the muscularis using cotton applicators. We cut the muscularis into 5 cm segments and digested these for 45 min at 37°C with 100 U/mL collagenase type VIII and 50 U/mL DNase I in RPMI containing 10% fetal calf serum (FCS) and 0.1% sodium azide. Then samples were homogenised by vigorously pipetting up and down 10 times using 1000 µL tips; afterwards supernatants were passed through a 100 µm nylon mesh.
Flow cytometry
We used the following monoclonal antibody clones: CD103 (PE, M290, BD); CD45 (APC-Cy7, APC and FITC, 30-F11, BD); F4/80 (PE-Cy7, BM8, BioLegend); CD11c (BV421 and BV605, N418, BioLegend); CD64 (PE, X54-5/7.1, BioLegend); MHC class II (APC and PE-Cy7, M5/114.15.2, BioLegend); Ly6C (PerCP-Cy5.5, AL-21, BD); and CD11b (APC and PE, M1/70, BD). Unspecific Fc receptor binding was blocked using human immune globulin, 10% liquid (Privigen) diluted 1:66. We determined absolute cell numbers by adding fixed numbers of CaliBRITE APC-beads (6 µm) (BD Biosciences) before measurement as internal reference, excluding dead cells with Hoechst-33258 (Invitrogen). Additionally, muscularis was weighed before digestion and cell numbers were normalised to 100 mg muscularis. Flow cytometry was performed on a BD LSRFortessa II (cytometer configuration is listed on https://cores.ukb.uni-bonn.de/fccf/wp-content/uploads/sites/11/2017/05/Filter-und-Fluorochrome-Fortessa-BUV.pdf) and data were analysed with Flow-Jo software (Tristar). Experiments including fluorescence activated cell sorting were performed on a BD Aria with a 70 µm nozzle (configuration: https://cores.ukb.uni-bonn.de/fccf/wp-content/uploads/sites/11/2017/02/Filter-und-Fluorochrome-Aria.pdf).
Enzyme-linked immunosorbent assay
ELISA on muscularis homogenate was performed as described previously.4 Shortly, purified muscularis was homogenised in phosphate-buffered saline (PBS) solution containing Roche protease inhibitor and supernatant of centrifuged homogenate was analysed on either IFNγ or IL-12p70 ELISA (DuoSet, eBioscience, Heidelberg, Germany).
RNA extraction and real-time PCR
All reagents, if not otherwise specified, were from Applied Biosystems. Cells for flow cytometric cell sorting were prepared as described above and RNA was extracted using NucleoSpin RNA XS kit (Macherey Nagel). Cells were directly sorted into the supplied lysis buffer RA1. RNA extraction was performed according to the supplier’s instructions. RNA was eluted in 10 µL RNAse-free water and directly transcribed into cDNA with the High Capacity Reverse Transcription kit according to manufacturer’s description. For whole tissue analysis purified muscularis was snap-frozen in liquid nitrogen and extracted using NucleoSpin RNA II kit according to manufacturer’s description. High-capacity cDNA kit was used for reverse transcription according to manufacturer’s description. Qiagen supplied QuantiTect Nos2 primer, primers (5’-TCATTGAATGCTTGGCGCTG-3’, 5’- AGGAACTGGCAAAAGGATGGT-3’) were used for Ifnγ, Hprt (5’- GTCCCAGCGTCGTGATTAGCGAT-3’, 5’-GGGCCACAATGTGATGGCCTCC-3’) was used as a standard. PCR was performed on a Roche LightCycler using SYBR Green Master Mix according to manufacturer’s description. Dissociation of the PCR products by a melting curve analysis was used to exclude samples showing multiple or aberrant melting peaks.
Reagents
Clodronate liposomes were obtained from www.clodronateliposomes.org and 200 µL were applied intravenously 12 hours before manipulation, at the time point of manipulation and 6 hours after manipulation if not indicated otherwise. 10 ng DT (Sigma Aldrich, Germany) per gram mouse was applied intravenously 12 hours before manipulation and at the time point of manipulation.
Statistical analysis
Appropriate assumptions of data (eg, normal distribution or similar variation between experimental groups) were examined before statistical tests were conducted. The sample size was analysed by G*Power analysis to ensure adequate power to detect a prespecified effect size. The number of experiments and the amount of mice per group are given in the legends to the figures. Student’s t-tests were used whenever two groups were compared, and one-way and two-way analyses of variance were done wherever necessary to determine significance for all other data. In all experiments, statistical significance was determined between the groups, which underwent SIM. Results are given as mean±SEM; *p<0.05; **p<0.01; ***p<0.001.
gutjnl-2017-313856supp006.pdf (198.8KB, pdf)
Acknowledgments
The authors acknowledge support from the Central Animal Facilities of the Medical Faculty Bonn and Essen and Lars Franken at Ayoxxa for measuring proteins in low volume samples. We also acknowledge the Imaging Core Facility in Essen (IMCES) and the flow cytometry Core Facility in Bonn.
Footnotes
Contributors: DRE and CK conceived and supervised the study and wrote the manuscript. SG, JMP, CK and DRE designed the experiments. JMP, SG, ST and JKV performed the experiments. WWA, SJ, MG, LKH and DZ provided crucial ideas and reagents and discussed the data. All authors discussed and interpreted the results.
Funding: This work was supported by grants from the Deutsche Forschungsgemeinschaft to DRE and CK (EN984/1-1, EN984/5-1, EN984/6-1 and SFBTR57), Mercur (An-2015-0066), the Marga und Walter-Boll Stiftung, EU Consortium RELENT and intramural research funds from the Medical Faculty of the University Duisburg-Essen (IFORES to DRE). CK is a member of the Excellence Cluster ImmunoSensation in Bonn.
Competing interests: None declared.
Ethics approval: All procedures involving mice were approved by the governmental review board (Bezirksregierung Köln, Landesamt für Natur, Umwelt und Verbraucherschutz NRW in Recklinghausen, Germany).
Provenance and peer review: Not commissioned; externally peer reviewed.
Correction notice: This article has been corrected since it published Online First. The last two author positions have been corrected.
References
- 1. Cerovic V, Bain CC, Mowat AM, et al. . Intestinal macrophages and dendritic cells: what’s the difference? Trends Immunol 2014;35:270–7. 10.1016/j.it.2014.04.003 [DOI] [PubMed] [Google Scholar]
- 2. Mildner A, Jung S. Development and function of dendritic cell subsets. Immunity 2014;40:642–56. 10.1016/j.immuni.2014.04.016 [DOI] [PubMed] [Google Scholar]
- 3. Gordon S, Taylor PR. Monocyte and macrophage heterogeneity. Nat Rev Immunol 2005;5:953–64. 10.1038/nri1733 [DOI] [PubMed] [Google Scholar]
- 4. Engel DR, Koscielny A, Wehner S, et al. . T helper type 1 memory cells disseminate postoperative ileus over the entire intestinal tract. Nat Med 2010;16:1407–13. 10.1038/nm.2255 [DOI] [PubMed] [Google Scholar]
- 5. Muller PA, Koscsó B, Rajani GM, et al. . Crosstalk between muscularis macrophages and enteric neurons regulates gastrointestinal motility. Cell 2014;158:300–13. 10.1016/j.cell.2014.04.050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Anitha M, Vijay-Kumar M, Sitaraman SV, et al. . Gut microbial products regulate murine gastrointestinal motility via Toll-like receptor 4 signaling. Gastroenterology 2012;143:1006–16. 10.1053/j.gastro.2012.06.034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Wehner S, Engel DR. Resident macrophages in the healthy and inflamed intestinal muscularis externa. Pflugers Arch 2017;469:541–52. 10.1007/s00424-017-1948-4 [DOI] [PubMed] [Google Scholar]
- 8. Bain CC, Scott CL, Uronen-Hansson H, et al. . Resident and pro-inflammatory macrophages in the Colon represent alternative context-dependent fates of the same Ly6Chi monocyte precursors. Mucosal Immunol 2013;6:498–510. 10.1038/mi.2012.89 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Wehner S, Behrendt FF, Lyutenski BN, et al. . Inhibition of macrophage function prevents intestinal inflammation and postoperative ileus in rodents. Gut 2007;56:176–85. 10.1136/gut.2005.089615 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Zigmond E, Varol C, Farache J, et al. . Ly6C hi monocytes in the inflamed Colon give rise to proinflammatory effector cells and migratory antigen-presenting cells. Immunity 2012;37:1076–90. 10.1016/j.immuni.2012.08.026 [DOI] [PubMed] [Google Scholar]
- 11. Tilg H, Moschen AR, Food MAR. Food, immunity, and the microbiome. Gastroenterology 2015;148:1107–19. 10.1053/j.gastro.2014.12.036 [DOI] [PubMed] [Google Scholar]
- 12. Schwarz NT, Kalff JC, Türler A, et al. . Selective jejunal manipulation causes postoperative pan-enteric inflammation and dysmotility. Gastroenterology 2004;126:159–69. 10.1053/j.gastro.2003.10.060 [DOI] [PubMed] [Google Scholar]
- 13. Gross M, Salame TM, Jung S. Guardians of the gut - Murine intestinal macrophages and dendritic cells. Front Immunol 2015;6:254 10.3389/fimmu.2015.00254 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Edelson BT, Kc W, Juang R, et al. . Peripheral CD103+ dendritic cells form a unified subset developmentally related to CD8alpha+ conventional dendritic cells. J Exp Med 2010;207:823–36. 10.1084/jem.20091627 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Hildner K, Edelson BT, Purtha WE, et al. . Batf3 deficiency reveals a critical role for CD8alpha+ dendritic cells in cytotoxic T cell immunity. Science 2008;322:1097–100. 10.1126/science.1164206 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Kurts C, Robinson BW, Knolle PA. Cross-priming in health and disease. Nat Rev Immunol 2010;10:403–14. 10.1038/nri2780 [DOI] [PubMed] [Google Scholar]
- 17. Varol C, Vallon-Eberhard A, Elinav E, et al. . Intestinal Lamina propria dendritic cell subsets have different origin and functions. Immunity 2009;31:502–12. 10.1016/j.immuni.2009.06.025 [DOI] [PubMed] [Google Scholar]
- 18. Bogunovic M, Ginhoux F, Helft J, et al. . Origin of the Lamina propria dendritic cell network. Immunity 2009;31:513–25. 10.1016/j.immuni.2009.08.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Persson EK, Uronen-Hansson H, Semmrich M, et al. . IRF4 transcription-factor-dependent CD103(+)CD11b(+) dendritic cells drive mucosal T helper 17 cell differentiation. Immunity 2013;38:958–69. 10.1016/j.immuni.2013.03.009 [DOI] [PubMed] [Google Scholar]
- 20. Scott CL, Bain CC, Wright PB, et al. . CCR2(+)CD103(-) intestinal dendritic cells develop from DC-committed precursors and induce interleukin-17 production by T cells. Mucosal Immunol 2015;8:327–39. 10.1038/mi.2014.70 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Cerovic V, Houston SA, Scott CL, et al. . Intestinal CD103(-) dendritic cells migrate in lymph and prime effector T cells. Mucosal Immunol 2013;6:104–13. 10.1038/mi.2012.53 [DOI] [PubMed] [Google Scholar]
- 22. Tamoutounour S, Henri S, Lelouard H, et al. . CD64 distinguishes macrophages from dendritic cells in the gut and reveals the Th1-inducing role of mesenteric lymph node macrophages during colitis. Eur J Immunol 2012;42:3150–66. 10.1002/eji.201242847 [DOI] [PubMed] [Google Scholar]
- 23. Gomez Perdiguero E, Klapproth K, Schulz C, et al. . Tissue-resident macrophages originate from yolk-sac-derived erythro-myeloid progenitors. Nature 2015;518:547–51. 10.1038/nature13989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Schulz C, Gomez Perdiguero E, Chorro L, et al. . A lineage of myeloid cells independent of myb and hematopoietic stem cells. Science 2012;336:86–90. 10.1126/science.1219179 [DOI] [PubMed] [Google Scholar]
- 25. Yona S, Kim KW, Wolf Y, et al. . Fate mapping reveals origins and dynamics of monocytes and tissue macrophages under homeostasis. Immunity 2013;38:79–91. 10.1016/j.immuni.2012.12.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Hashimoto D, Chow A, Noizat C, et al. . Tissue-resident macrophages self-maintain locally throughout adult life with minimal contribution from circulating monocytes. Immunity 2013;38:792–804. 10.1016/j.immuni.2013.04.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Jaensson E, Uronen-Hansson H, Pabst O, et al. . Small intestinal CD103+ dendritic cells display unique functional properties that are conserved between mice and humans. J Exp Med 2008;205:2139–49. 10.1084/jem.20080414 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Varol C, Landsman L, Fogg DK, et al. . Monocytes give rise to mucosal, but not splenic, conventional dendritic cells. J Exp Med 2007;204:171–80. 10.1084/jem.20061011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Zigmond E, Jung S. Intestinal macrophages: well educated exceptions from the rule. Trends Immunol 2013;34:162–8. 10.1016/j.it.2013.02.001 [DOI] [PubMed] [Google Scholar]
- 30. Wynn TA, Chawla A, Pollard JW. Macrophage biology in development, homeostasis and disease. Nature 2013;496:445–55. 10.1038/nature12034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Türler A, Kalff JC, Moore BA, et al. . Leukocyte-derived inducible nitric oxide synthase mediates murine postoperative ileus. Ann Surg 2006;244:220–9. 10.1097/01.sla.0000229963.37544.59 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Serbina NV, Pamer EG. Monocyte emigration from bone marrow during bacterial infection requires signals mediated by chemokine receptor CCR2. Nat Immunol 2006;7:311–7. 10.1038/ni1309 [DOI] [PubMed] [Google Scholar]
- 33. Geissmann F, Jung S, Littman DR. Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity 2003;19:71–82. 10.1016/S1074-7613(03)00174-2 [DOI] [PubMed] [Google Scholar]
- 34. Mowat AM, Agace WW. Regional specialization within the intestinal immune system. Nat Rev Immunol 2014;14:667–85. 10.1038/nri3738 [DOI] [PubMed] [Google Scholar]
- 35. Caballero S, Pamer EG. Microbiota-mediated inflammation and antimicrobial defense in the intestine. Annu Rev Immunol 2015;33:227–56. 10.1146/annurev-immunol-032713-120238 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Schulz O, Pabst O. Antigen sampling in the small intestine. Trends Immunol 2013;34:155–61. 10.1016/j.it.2012.09.006 [DOI] [PubMed] [Google Scholar]
- 37. Bain CC, Mowat AM. The monocyte-macrophage Axis in the intestine. Cell Immunol 2014;291:41–8. 10.1016/j.cellimm.2014.03.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Schlitzer A, McGovern N, Teo P, et al. . IRF4 transcription factor-dependent CD11b+ dendritic cells in human and mouse control mucosal IL-17 cytokine responses. Immunity 2013;38:970–83. 10.1016/j.immuni.2013.04.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Takada Y, Hisamatsu T, Kamada N, et al. . Monocyte chemoattractant protein-1 contributes to gut homeostasis and intestinal inflammation by composition of IL-10-producing regulatory macrophage subset. J Immunol 2010;184:2671–6. 10.4049/jimmunol.0804012 [DOI] [PubMed] [Google Scholar]
- 40. Rivollier A, He J, Kole A, et al. . Inflammation switches the differentiation program of Ly6Chi monocytes from antiinflammatory macrophages to inflammatory dendritic cells in the colon. J Exp Med 2012;209:139–55. 10.1084/jem.20101387 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Ginhoux F, Jung S. Monocytes and macrophages: developmental pathways and tissue homeostasis. Nat Rev Immunol 2014;14:392–404. 10.1038/nri3671 [DOI] [PubMed] [Google Scholar]
- 42. Denning TL, Norris BA, Medina-Contreras O, et al. . Functional specializations of intestinal dendritic cell and macrophage subsets that control Th17 and regulatory T cell responses are dependent on the T cell/APC ratio, source of mouse strain, and regional localization. J Immunol 2011;187:733–47. 10.4049/jimmunol.1002701 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Snoek SA, Dhawan S, van Bree SH, et al. . Mast cells trigger epithelial barrier dysfunction, bacterial translocation and postoperative ileus in a mouse model. Neurogastroenterol Motil 2012;24:172–e91. 10.1111/j.1365-2982.2011.01820.x [DOI] [PubMed] [Google Scholar]
- 44. Schwarz NT, Beer-Stolz D, Simmons RL, et al. . Pathogenesis of paralytic ileus: intestinal manipulation opens a transient pathway between the intestinal lumen and the leukocytic infiltrate of the jejunal muscularis. Ann Surg 2002;235:31–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Stoffels B, Hupa KJ, Snoek SA, et al. . Postoperative ileus involves interleukin-1 receptor signaling in enteric glia. Gastroenterology 2014;146:176–87. 10.1053/j.gastro.2013.09.030 [DOI] [PubMed] [Google Scholar]
- 46. Kinnebrew MA, Buffie CG, Diehl GE, et al. . Interleukin 23 production by intestinal CD103(+)CD11b(+) dendritic cells in response to bacterial flagellin enhances mucosal innate immune defense. Immunity 2012;36:276–87. 10.1016/j.immuni.2011.12.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Uematsu S, Fujimoto K, Jang MH, et al. . Regulation of humoral and cellular gut immunity by Lamina propria dendritic cells expressing Toll-like receptor 5. Nat Immunol 2008;9:769–76. 10.1038/ni.1622 [DOI] [PubMed] [Google Scholar]
- 48. Kuziel WA, Morgan SJ, Dawson TC, et al. . Severe reduction in leukocyte adhesion and monocyte extravasation in mice deficient in CC chemokine receptor 2. Proc Natl Acad Sci U S A 1997;94:12053–8. 10.1073/pnas.94.22.12053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Jung S, Aliberti J, Graemmel P, et al. . Analysis of fractalkine receptor CX(3)CR1 function by targeted deletion and green fluorescent protein reporter gene insertion. Mol Cell Biol 2000;20:4106–14. 10.1128/MCB.20.11.4106-4114.2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Welty NE, Staley C, Ghilardi N, et al. . Intestinal Lamina propria dendritic cells maintain T cell homeostasis but do not affect commensalism. J Exp Med 2013;210:2011–24. 10.1084/jem.20130728 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Wehner S, Koscielny A, Vilz TO, et al. . Measurement of gastrointestinal and colonic transit in mice. Protoc exch 2011. 10.1038/protex.2011.219 [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
gutjnl-2017-313856supp001.jpg (896.7KB, jpg)
gutjnl-2017-313856supp002.jpg (1.2MB, jpg)
gutjnl-2017-313856supp003.jpg (377.5KB, jpg)
gutjnl-2017-313856supp004.jpg (800.2KB, jpg)
gutjnl-2017-313856supp005.jpg (1.2MB, jpg)
gutjnl-2017-313856supp006.pdf (198.8KB, pdf)