Abstract
Sophoridine is an alkaloid extracted from Sophora alopecuroides that has extensive pharmacological actions. In the present study, the effect of sophoridine on cell growth of human medulloblastoma and its mechanism were investigated. Human medulloblastoma D283-Med cells were incubated with 0, 0.5, 1 or 2 mg/ml sophoridine for 24, 48 or 72 h. Cell proliferation and cytotoxicity were analyzed using MTT and lactate dehydrogenase assays, respectively. Next, analyses of cell apoptosis and caspase-3/8 activity were performed using flow cytometry or spectrophotometry, respectively. Lastly, the change in FoxM1, TrkB, BDNF, NF-κB and AP-1 expression was investigated using western blot analysis. In the present study, treatment with sophoridine significantly suppressed cell growth and induced apoptosis in human medulloblastoma cells. In addition, sophoridine significantly increased cytotoxicity and caspase-3/8 activity in human medulloblastoma. Finally, it was found that sophoridine suppresses the protein expression of FoxM1, TrkB, BDNF NF-κB and AP-1 in human medulloblastoma cells. The present study suggests that sophoridine suppresses cell growth of human medulloblastoma through the inhibition of the FoxM1, NF-κB and AP-1 signaling pathway.
Keywords: sophoridine, medulloblastoma, FoxM1, NF-κB, AP-1
Introduction
Cerebral tumors are the second most common tumor in pediatric patients, with morbidity rate of 3.3/10,0000 individuals in 2012 in China (1). Due to its poor prognosis, cerebral tumors are the leading cause of mortality in pediatric oncology (1). Medulloblastoma is a type of neuroepithelial tumor of the epencephalon, and is a common pediatric central nervous system tumor, with a morbidity rate of 25% and high paroxysmal age of 8 years (2). According to the reclassification of nervous system neoplasms by the World Health Organization in 2002, the types of medulloblastoma are classic medulloblastoma, pro-fibroplasia, maxicell/anaplasia and melanin. For classic medulloblastoma, tumor cells grow vigorously with little cytoplasm shown on histopathological examination (3). Statistics have shown that the survival time of 70% of patients reaches 5 years, while a certain high risk-patients have metastasis (4). Only 25% of these patients with metastasis have a survival time of 5 years (5).
Termed Forkhead box or winged helix domain, Forkhead box protein Ml (FOXM1) is an evolutionarily-conserved transcriptional regulatory factor family characterized by DNA binding domain (6). FOXM1 is a key regulator of the cell cycle. FOXM1 is expressed during the G1 and S phases in the cell cycle, and can regulate the transcription activity of numerous genes, including encoding cell division cycle (Cdc) 25A, Cdc25B, cyclin B, cyclin A, cyclin Dl, p21cip1, p27kipl, Aurora B kinase and Polo-like kinase 1. The loss of FOXM1 expression may lead to formation defects in the mitotic spindle and the delay of cell division, which may result in the failure of mitosis (7). FOXM1 is also associated with cell proliferation and apoptosis. A previous study has found that the signaling pathway of FOXM1 has an essential role in maintaining self-stability during cell developmental process (8). New evidence has indicated that FOXM1 has abnormal high expression in malignant cancers, such as lung cancer, spongioblastoma, prostatic cancer, hepatocellular carcinoma, primary breast carcinoma and pancreatic carcinoma (9). When altered using RNA interference, the expression of FOXM1 in the mammary gland and pancreatic cancer cells triggered the proliferation, metastasis, invasion, migration and inhibition of carcinoma cells (10).
As an important intranuclear transcription factor, the stress protein activator protein-1 (AP-1) directly participates in normal growth and carcinous conversion process while its functions in cells directly depend on cell types, the constituents of AP-1 and the relative proportion of different parts (11). The activity of AP-1 is closely associated with tumors. It can activate tumor-associated genes and promotes the occurrence and malignant evolution of tumors. AP-1 participates in cell proliferation, differentiation and conversion and has a fundamental role in tumor formation, metastasis and invasion (12).
Sophoridine (Fig. 1) is an alkaloid that occurs in high levels in Sophora alopecuroides (13). Previous studies have identified that sophoridine reduced inflammatory responses caused by LPS (14,15). It has pharmacological functions such as antitumor and anti-arrhythmia and affects the immune and central nervous systems (12). When used at an antitumor dosage, sophoridine can lower the heart rates of tested animals without affecting other indicators of the electrocardiogram or breathing (13). Therefore, the present study assessed the effect of Sophoridine on the suppression of cell growth in human medulloblastoma, which may provide an important biological basis for the pathogenesis of this disease.
Figure 1.
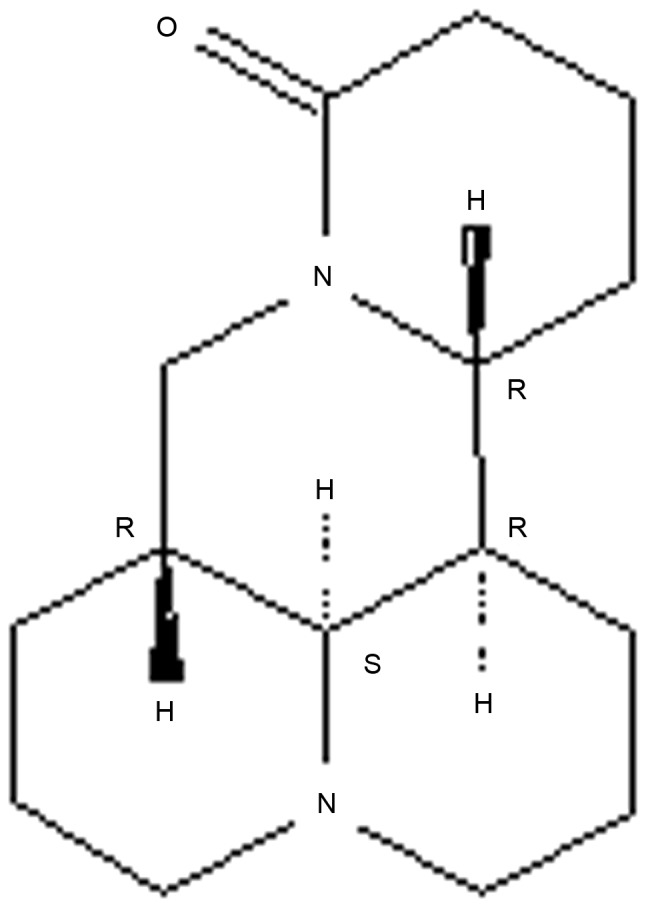
The chemical structure of sophoridine.
Materials and methods
Cell culture
The human medulloblastoma D283-Med cell line was obtained directly from the Central Laboratory of Tianjin Medical University Cancer Institute and Hospital (Tianjin, China) and maintained in DMEM-Nutrient Mixture F-12 (DMEM/F12; Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA) with human recombinant epidermal growth factor (20 ng/ml), B-27 supplement (2%; Invitrogen; Thermo Fisher Scientific, Inc.), supplemented with 10% fetal bovine serum (Invitrogen; Thermo Fisher Scientific, Inc.) and 1% 100 µg/ml penicillin/streptomycin solution at 37°C in a 5% CO2 atmosphere.
Cell proliferation and cytotoxicity analysis
D283-Med cells (5×104/ml; 200 µl) were added to a 96-well plate and divided into groups treated with 0, 0.5, 1 and 2 mg/ml sophoridine (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) for 24, 48 and 72 h. Subsequently, 50 µl MTT (5 mg/ml; Invitrogen; Thermo Fisher Scientific, Inc.) was added to the D283-Med cells and incubated for 4 h at 37°C in a 5% CO2 atmosphere. MTT was removed and 150 µl dimethyl sulfoxide was added and the plate was agitated for 20 min. Lactate dehydrogenase (LDH; 60 µl; Thermo Fisher Scientific, Inc.) was added, and the plate was incubated for 30 min at 37°C in darkness. Cell proliferation and cytotoxicity were analyzed using a spectrophotometer (Tecan Sunrise Rainbow; Tecan Japan Co., Ltd., Tokyo, Japan) at 490 nm.
Cell apoptosis analysis
D283-Med cells (1×108/ml; 2 ml) were added to a 6-well plate and divided into groups treated with 0, 0.5, 1 and 2 mg/ml sophoridine for 48 h. The D283-Med cells were incubated with 5 µl annexin V-fluorescein isothiocyanate (Caltag Laboratories, Burlingame, CA, USA) and 5 µl of propidium iodide (Caltag Laboratories) for 30 min at room temperature. Cells apoptosis was analyzed using the Guava easyCyte 5HT Flow Cytometer (Merck KGaA, Darmstadt, Germany).
Caspase-3/8 activity
D283-Med cells (1×108/ml; 2 ml) were added to a 6-well plate and divided into groups treated with 0, 0.5, 1 and 2 mg/ml sophoridine for 48 h. D283-Med cells were lysed on ice with 100 ml pre-cooled cell lysis buffer (Beyotime Institute of Biotechnology, Haimen, China) for 30 min. The protein concentration was determined by the bicinchoninic acid method (NE-PER; Thermo Fisher Scientific, Inc.). The substrates 10 µl of Ac-DEVD-pNA (caspase-3; 2 mM; Beyotime Institute of Biotechnology) and 10 µl of Ac-IETD-pNA (caspase-8; 2 mM; Beyotime Institute of Biotechnology) were added to each well for 1 h at 37°C. The activity of caspase-3/8 was analyzed using a spectrophotometer (Tecan Sunrise Rainbow; Tecan Japan Co., Ltd.) at 450 nm.
Western blot analysis
D283-Med cells (1×108/ml; 2 ml) were added to a 6-well plate and divided into groups treated with 0, 0.5, 1 and 2 mg/ml sophoridine for 48 h. D283-Med cells were lysed on ice with 100 ml pre-cooled cell lysis buffer for 30 min. The protein concentration was determined by the bicinchoninic acid method (NE-PER; Thermo Fisher Scientific, Inc.). Protein samples were separated by SDS-PAGE on a 8–12% gel and then transferred onto a polyvinylidene difluoride membrane (GE Healthcare, Little Chalfont, UK). After blocking with 5% non-fat milk for 1 h, membranes were incubated with primary anti-FoxM1 (cat. no. sc-502; 1:500; Santa Cruz Biotechnology, Inc., Dallas, TX, USA), anti-TrkB (cat. no. sc-118; 1:500; Santa Cruz Biotechnology, Inc.), anti-BDNF (cat. no. sc-20981; 1:500; Santa Cruz Biotechnology, Inc.), anti-NF-κB (cat. no. sc-7178; 1:500; Santa Cruz Biotechnology, Inc.), anti-AP-1 (cat. no. ab21981; 1:1,000; Abcam, Cambridge, MA, USA) and anti-β-actin (cat. no. sc-7210; 1:500; Santa Cruz Biotechnology, Inc.) antibodies at 4°C overnight. After washing in Tris-buffered saline with Tween-20 (20 mM Tris-HCl, 150 mM NaCl and 0.05% Tween-20), the membranes were incubated with goat anti-rabbit IgG-horseradish peroxidase secondary antibodies (cat. no. sc-2004; 1:5,000; Santa Cruz Biotechnology, Inc.) at 37°C for 2 h. Subsequent to this incubation, the membranes were visualized using a BeyoECL Plus (Beyotime Institute of Biotechnology).
Statistical analysis
The experimental data were presented as the mean ± standard deviation and analyzed with SPSS 13.0 software (SPSS, Inc., Chicago, IL, USA). Statistical significance was determined by one-way analysis of variance followed by Tukey post hoc analysis. P<0.05 was considered to indicate a statistically significant difference.
Results
Sophoridine suppresses cell growth of human medulloblastoma
To determine the effect of sophoridine on the growth of D283-Med cells, cell proliferation was measured using an MTT assay. As shown in Fig. 2, the proliferation of D283-Med cells was suppressed by treatment with sophoridine in a dose- and time-dependent manner. In particular, 2 mg/ml sophoridine significantly suppressed cell proliferation subsequent to treatment for 24, 48 and 72 h, 1 mg/ml sophoridine significantly suppressed cell proliferation subsequent to treatment for 72 h, and 0.5 mg/ml sophoridine significantly suppressed cell proliferation subsequent to treatment for 72 h in D283-Med cell.
Figure 2.
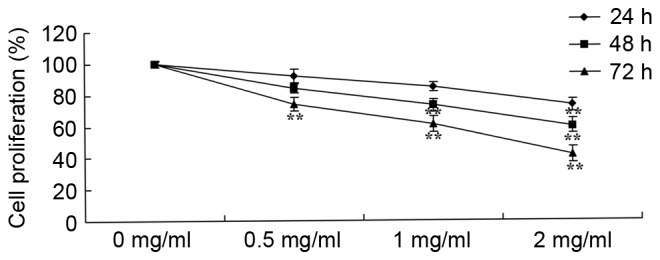
Sophoridine suppresses cell growth of human medulloblastoma cells. **P<0.01 vs. D283-Med cells incubated with 0 µM sophoridine.
Sophoridine is cytotoxic in human medulloblastoma
To study the effect of sophoridine on cytotoxicity of human medulloblastoma, a LDH assay was used to analyze the cytotoxicity of sophoridine on D283-Med cells. The results showed that treatment with 1 or 2 mg/ml sophoridine significantly induced cytotoxicity of D283-Med cells (Fig. 3).
Figure 3.
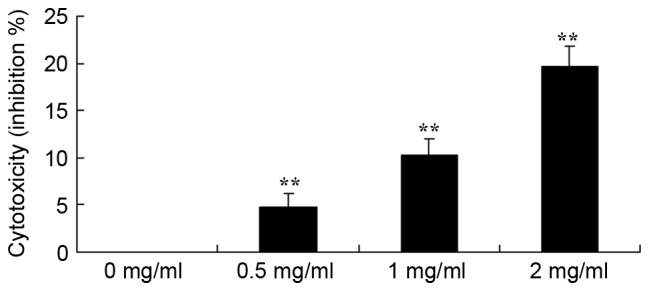
Sophoridine is cytotoxic in human medulloblastoma cells. **P<0.01 vs. D283-Med cells incubated with 0 µM sophoridine.
Sophoridine induces apoptosis in human medulloblastoma
To further study the effect of sophoridine on the apoptosis of human medulloblastoma cells, apoptosis rates were detected using flow cytometry. Fig. 4 showed that treatment with 1 or 2 mg/ml sophoridine significantly induced apoptosis in D283-Med cells, compared with control cells (0 mg/ml sophoridine).
Figure 4.
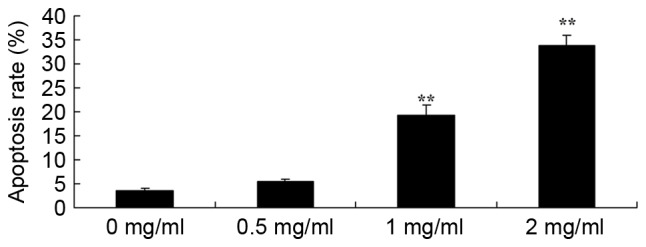
Sophoridine induces apoptosis in human medulloblastoma cells. **P<0.01 vs. D283-Med cells incubated with 0 µM sophoridine.
Sophoridine induces caspase-3/8 activity in human medulloblastoma
A Business kit was used to evaluate the effect of sophoridine on caspase-3/8 activity of human medulloblastoma in vitro subsequent to treatment for 48 h. The representative results are shown in Fig. 5. Sophoridine (1 or 2 mg/ml) significantly induced caspase-3/8 activity in D283-Med cells, compared with control cells (0 mg/ml sophoridine).
Figure 5.
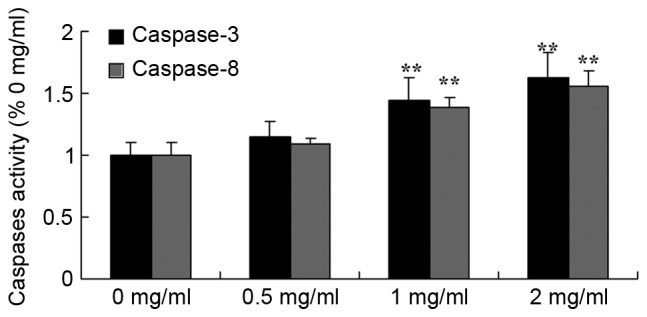
Sophoridine induces caspase-3/8 activity in human medulloblastoma cells. **P<0.01 vs. D283-Med cells incubated with 0 µM sophoridine.
Sophoridine suppresses FoxM1 protein expression in human medulloblastoma
To evaluate the effect of sophoridine on FoxM1 signal in human medulloblastoma, FoxM1 protein expression was recorded using western blot analysis. Sophoridine concentrations of 1 and 2 mg/ml concentrations were found to significantly suppress FoxM1 protein expression in D283-Med cells, compared with control cells (0 mg/ml sophoridine; Fig. 6).
Figure 6.
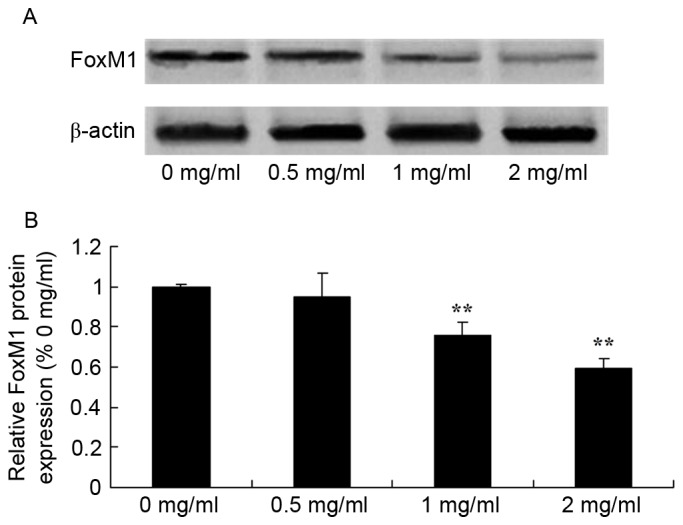
Sophoridine suppresses FoxM1 protein expression in human medulloblastoma cells, as shown using (A) western blot analysis and (B) statistical analysis of FoxM1 protein expression in human medulloblastoma D283-Med cells. **P<0.01 vs. D283-Med cells incubated with 0 µM of sophoridine.
Sophoridine suppresses TrkB protein expression in human medulloblastoma
To further evaluate the effect of sophoridine on TrkB signaling in human medulloblastoma, western blot analysis was used to detect TrkB protein expression in D283-Med cells. TrkB protein expression of D283-Med cells was significantly inhibited by 1 and 2 mg/ml of sophoridine, compared with control cells (0 mg/ml sophoridine; Fig. 7).
Figure 7.
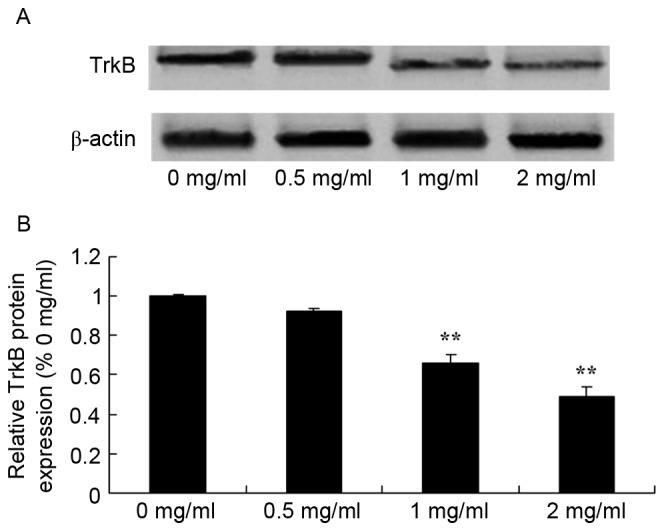
Sophoridine suppresses TrkB protein expression in human medulloblastoma cells, as shown using (A) western blot analysis and (B) statistical analysis of FoxM1 protein expression in human medulloblastoma D283-Med cells. **P<0.01 vs. D283-Med cells incubated with 0 µM of sophoridine.
Sophoridine suppresses BDNF protein expression in human medulloblastoma
To study the effect of sophoridine on BDNF signaling in human medulloblastoma, BDNF protein expression in D283-Med cells was measured using western blot analysis. The results showed that treatment with 1 and 2 mg/ml sophoridine significantly reduced BDNF protein expression in D283-Med cells, compared with control cells (0 mg/ml sophoridine; Fig. 8).
Figure 8.
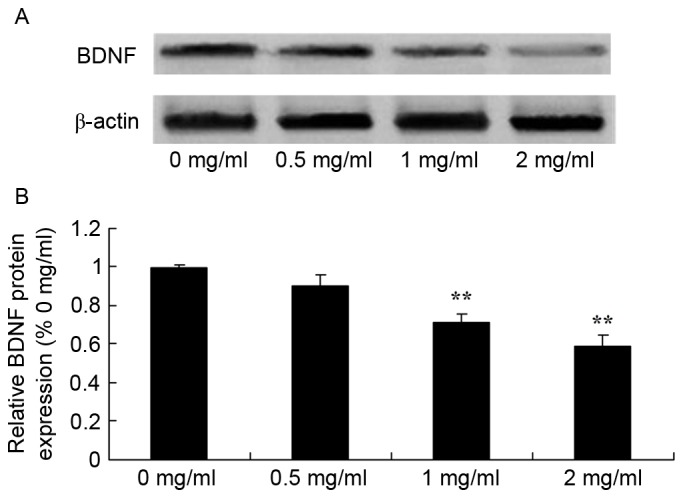
Sophoridine suppresses BDNF protein expression in human medulloblastoma cells, as shown using (A) western blot analysis and (B) statistical analysis of BDNF protein expression in human medulloblastoma D283-Med cells. **P<0.01 vs. D283-Med cells incubated with 0 µM sophoridine.
Sophoridine suppresses NF-κB protein expression in human medulloblastoma
To further study the mechanism of sophoridine on cell growth of human medulloblastoma, changes in NF-κB protein expression were examined using western blot analysis. The present study also found that NF-κB protein expression in D283-Med cells was significantly suppressed by treatment with 1 and 2 mg/ml sophoridine, compared with control cells (0 mg/ml sophoridine; Fig. 9).
Figure 9.
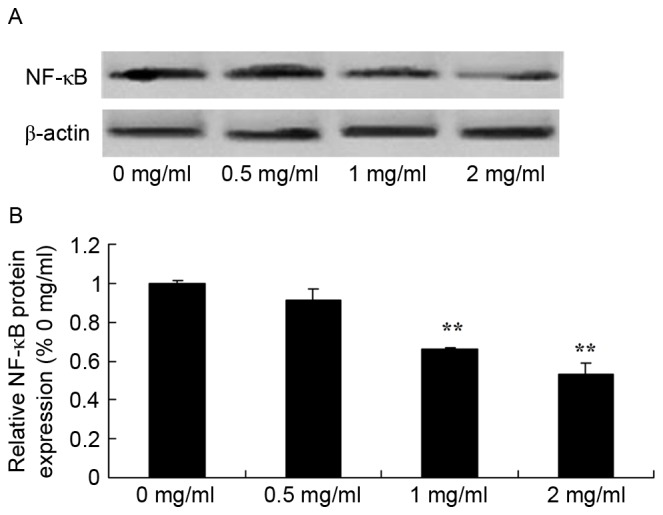
Sophoridine suppresses NF-κB protein expression in human medulloblastoma cells, as shown using (A) western blot analysis and (B) statistical analysis of NF-κB protein expression (B) of human medulloblastoma. **P<0.01 vs. D283-Med cells incubated with 0 µM of sophoridine.
Sophoridine suppresses AP-1 protein expression in human medulloblastoma
Since one of the main mechanisms of sophoridine is its effect on AP-1 signaling in human medulloblastoma, the present study evaluated whether sophoridine affects AP-1 protein expression in D283-Med cells. As shown in Fig. 10, 1 and 2 mg/ml sophoridine significantly inhibited AP-1 protein expression in human D283-Med cells, compared with control cells (0 mg/ml sophoridine; Fig. 10).
Figure 10.
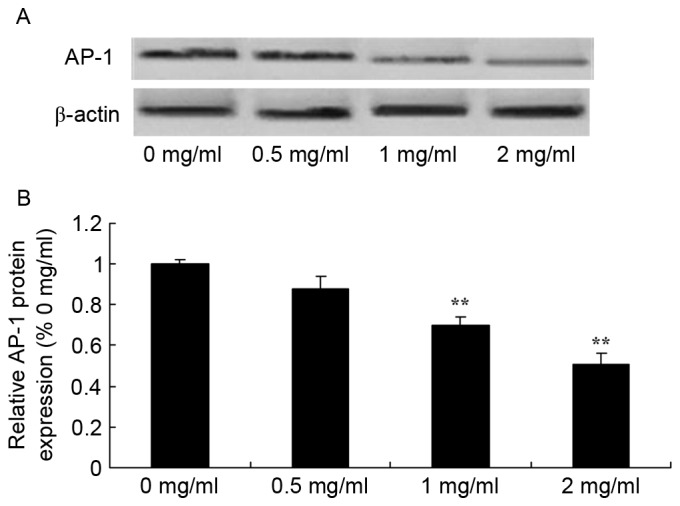
Sophoridine suppresses AP-1 protein expression in human medulloblastoma, as shown using (A) western blot analysis and (B) statistical analysis of AP-1 protein expression in human medulloblastoma. **P<0.01 vs. D283-Med cells incubated with 0 µM sophoridine.
Discussion
Medulloblastoma is an epithelial tumor of cerebellar neurocytes and is the most common central nervous system tumor in children (16). The morbidity of medulloblastoma accounts for 25% of pediatric brain tumors (17). The most common age is 8 years, and it mostly appears in patients >20 years old. According to statistics, the 5-year survival rate of patients is 70%, but certain high-risk patients (<3 years old) often present with metastasis, and the 5-year survival rate is 25% in 2012 in China (18). The present study showed that sophoridine suppresses cell growth, induces cytotoxicity and apoptosis and increased caspase-3/8 activity of human medulloblastoma. Wang et al (19) reported that sophoridine induces mitochondrial apoptosis through suppression of β-catenin/survivin signaling in cisplatin-resistant non-small cell lung cancer cells.
FOXM1 is a key regulator of the cell cycle and expressed during the G1, S and mitotic phases (9). A previous study suggested that FOXM1 could significantly downregulate the expression of P2cipi and P27kipi (7). During the transitional phase between G2 and M, two downstream target genes of FOXM1 were found to promote NF-κB to participate in the progression of neurodegenerative diseases, such as Alzheimer's disease and Parkinson's disease (20). In the present study, it was found that sophoridine significantly suppressed FOXM1 protein expression in D283-Med cells. Wang et al (13) reported that sophoridine suppresses cell proliferation of human glioma U87MG cell line by upregulating the expression of caspase-3/8, NF-κB and AP-1.
The nuclear transcription factor NF-κB is contains κBα. In normal cells, NF-κB is located in the cytoplasm (21). Stimulated by extracellular functions, κBα can be degraded, which results in the activation of p65. The activation of NF-κB induces the expression of certain genes, including inflammatory factors, proliferation or pro-apoptosis factors (22). In the present study, it was also found that sophoridine significantly decreases the protein expression of NF-κB in D283-Med cells. Li et al (23) reported that sophoridine inhibits cell proliferation through regulation of the NF-κB signaling pathway in castration-resistant prostate cancer cells.
As target proteins of the TrkB/BDNF signaling pathway, NF-κB and FKHR are associated with FOXM1 (24). The functions of these proteins are associated with the survival, proliferation, invasion and metastasis (25). The interaction of FOXM1 with the TrkB/BDNF signaling pathway affects the biological behaviors of multiple myeloma cells. Previous studies have indicated that there is abnormal expression of TrkB/BDNF and FOXM1 in medulloblastoma, and the expression levels of these proteins are positively associated (26). The interaction between FOXM1 and TrkB/BDNF affects the apoptosis, cell cycle progression and proliferation of multiple myeloma cells (27). The present results showed that sophoridine suppressed TrkB and BDNF protein expression in D283-Med cells. Kan et al (28) reported that sophoridine protects neuro-axon from inflammation-induced injury through BDNF.
The AP-1 compound exerts its function by combining with genetic promoters (29). AP-1 is essential to the biological function of oncogenes, particularly participates in metastasis mediated by oncogenes. By importing TAM67 to inhibit the activity of AP-1, the invasiveness of squamous cell carcinoma in rats and humans can be markedly reduced (30). The results of the present study suggested that sophoridine suppressed the protein expression of NF-κB and AP-1 in D283-Med cell. Wang et al (13) reported that sophoridine suppresses cell proliferation of human glioma U87MG cell line through upregulating the expression of caspase-3/8, NF-κB and AP-1.
To conclude, the present findings demonstrated that sophoridine suppresses cell growth, induces cytotoxicity and apoptosis, and increases caspase-3/8 activity in human medulloblastoma. In addition, it was concluded that the FOXM1, NF-κB and AP-1 pathway has an important function in intracellular signaling in response to the anticancer effect of sophoridine in human medulloblastoma cells.
References
- 1.Robinson GW, Orr BA, Wu G, Gururangan S, Lin T, Qaddoumi I, Packer RJ, Goldman S, Prados MD, Desjardins A, et al. Vismodegib exerts targeted efficacy against recurrent sonic hedgehog-subgroup medulloblastoma: Results from phase ii pediatric brain tumor consortium studies PBTC-025B and PBTC-032. J Clin Oncol. 2015;33:2646–2654. doi: 10.1200/JCO.2014.60.1591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Deutsch M, Thomas PR, Krischer J, Boyett JM, Albright L, Aronin P, Langston J, Allen JC, Packer RJ, Linggood R, et al. Results of a prospective randomized trial comparing standard dose neuraxis irradiation (3,600 cGy/20) with reduced neuraxis irradiation (2,340 cGy/13) in patients with low-stage medulloblastoma. A combined children's cancer group-pediatric oncology group study. Pediatr Neurosurg. 1996;24:167–177. doi: 10.1159/000121042. [DOI] [PubMed] [Google Scholar]
- 3.Allen J, Donahue B, Mehta M, Miller DC, Rorke LB, Jakacki R, Robertson P, Sposto R, Holmes E, Vezina G, et al. A phase II study of preradiotherapy chemotherapy followed by hyperfractionated radiotherapy for newly diagnosed high-risk medulloblastoma/primitive neuroectodermal tumor: A report from the children's oncology group (CCG 9931) Int J Radiat Oncol Biol Phys. 2009;74:1006–1011. doi: 10.1016/j.ijrobp.2008.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cefalo G, Massimino M, Ruggiero A, Barone G, Ridola V, Spreafico F, Potepan P, Abate ME, Mascarin M, Garrè ML, et al. Temozolomide is an active agent in children with recurrent medulloblastoma/primitive neuroectodermal tumor: An Italian multi-institutional phase II trial. Neuro Oncol. 2014;16:748–753. doi: 10.1093/neuonc/not320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dhall G, Grodman H, Ji L, Gardner S, Dunkel IJ, McCowage GB, Diez B, Allen JC, Gopalan A, Cornelius AS, et al. Outcome of children less than three years old at diagnosis with non-metastatic medulloblastoma treated with chemotherapy on the ‘Head Start’ I and II protocols. Pediatr Blood Cancer. 2008;50:1169–1175. doi: 10.1002/pbc.21525. [DOI] [PubMed] [Google Scholar]
- 6.Ito T, Kohashi K, Yamada Y, Maekawa A, Kuda M, Furue M, Oda Y. Prognostic significance of Forkhead box M1 (FOXM1) expression and antitumor effect of FOXM1 inhibition in melanoma. Histopathology. 2015;69:63–71. doi: 10.1111/his.12909. [DOI] [PubMed] [Google Scholar]
- 7.Dai J, Yang L, Wang J, Xiao Y, Ruan Q. Prognostic value of FOXM1 in patients with malignant solid tumor: A meta-analysis and system review. Dis Markers. 2015;2015:352478. doi: 10.1155/2015/352478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhao F, Lam EW. Role of the forkhead transcription factor FOXO-FOXM1 axis in cancer and drug resistance. Front Med. 2012;6:376–380. doi: 10.1007/s11684-012-0228-0. [DOI] [PubMed] [Google Scholar]
- 9.Quan M, Wang P, Cui J, Gao Y, Xie K. The roles of FOXM1 in pancreatic stem cells and carcinogenesis. Mol Cancer. 2013;12:159. doi: 10.1186/1476-4598-12-159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Shi M, Cui J, Xie K. Signaling of miRNAs-FOXM1 in cancer and potential targeted therapy. Curr Drug Targets. 2013;14:1192–1202. doi: 10.2174/13894501113149990192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chambers M, Kirkpatrick G, Evans M, Gorski G, Foster S, Borghaei RC. IL-4 inhibition of IL-1 induced Matrix metalloproteinase-3 (MMP-3) expression in human fibroblasts involves decreased AP-1 activation via negative crosstalk involving of Jun N-terminal kinase (JNK) Exp Cell Res. 2013;319:1398–1408. doi: 10.1016/j.yexcr.2013.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Uluckan O, Guinea-Viniegra J, Jimenez M, Wagner EF. Signalling in inflammatory skin disease by AP-1 (Fos/Jun) Clin Exp Rheumatol. 2015;33(4 Suppl 92):S44–S49. [PubMed] [Google Scholar]
- 13.Wang WX, Sun ZH, Chen HM, Xu BN, Wang FY. Role and mechanism of Sophoridine on proliferation inhibition in human glioma U87MG cell line. Int J Clin Exp Med. 2015;8:464–471. [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang B, Liu ZY, Li YY, Luo Y, Liu ML, Dong HY, Wang YX, Liu Y, Zhao PT, Jin FG, Li ZC. Antiinflammatory effects of matrine in LPS-induced acute lung injury in mice. Eur J Pharm Sci. 2011;44:573–579. doi: 10.1016/j.ejps.2011.09.020. [DOI] [PubMed] [Google Scholar]
- 15.Liou CJ, Lai YR, Chen YL, Chang YH, Li ZY, Huang WC. Matrine attenuates COX-2 and ICAM-1 Expressions in human lung epithelial cells and prevents acute lung injury in LPS-induced mice. Mediators Inflamm. 2016;2016:3630485. doi: 10.1155/2016/3630485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gessi M, von Bueren AO, Rutkowski S, Pietsch T. p53 expression predicts dismal outcome for medulloblastoma patients with metastatic disease. J Neurooncol. 2012;106:135–141. doi: 10.1007/s11060-011-0648-8. [DOI] [PubMed] [Google Scholar]
- 17.Camara-Costa H, Resch A, Kieffer V, Lalande C, Poggi G, Kennedy C, Bull K, Calaminus G, Grill J, Doz F, et al. Neuropsychological outcome of children treated for standard risk Medulloblastoma in the PNET4 european randomized controlled trial of hyperfractionated versus standard radiation therapy and maintenance chemotherapy. Int J Radiat Oncol Biol Phys. 2015;92:978–985. doi: 10.1016/j.ijrobp.2015.04.023. [DOI] [PubMed] [Google Scholar]
- 18.Palmer SL, Leigh L, Ellison SC, Onar-Thomas A, Wu S, Qaddoumi I, Armstrong GT, Wright K, Wetmore C, Broniscer A, Gajjar A. Feasibility and efficacy of a computer-based intervention aimed at preventing reading decoding deficits among children undergoing active treatment for medulloblastoma: Results of a randomized trial. J Pediatr Psychol. 2014;39:450–458. doi: 10.1093/jpepsy/jst095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wang HQ, Jin JJ, Wang J. Matrine induces mitochondrial apoptosis in cisplatin-resistant non-small cell lung cancer cells via suppression of β-catenin/survivin signaling. Oncol Rep. 2015;33:2561–2566. doi: 10.3892/or.2015.3844. [DOI] [PubMed] [Google Scholar]
- 20.Arora R, Yates C, Gary BD, McClellan S, Tan M, Xi Y, Reed E, Piazza GA, Owen LB, Dean-Colomb W. Panepoxydone targets NF-kB and FOXM1 to inhibit proliferation, induce apoptosis and reverse epithelial to mesenchymal transition in breast cancer. PLoS One. 2014;9:e98370. doi: 10.1371/journal.pone.0098370. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 21.Arslan S, Korkmaz Ö, Özbilüm N, Berkan Ö. Association between NF-κBI and NF-κB BIA polymorphisms and coronary artery disease. Biomed Rep. 2015;3:736–740. doi: 10.3892/br.2015.499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Qian Y, Guan T, Huang M, Cao L, Li Y, Cheng H, Jin H, Yu D. Neuroprotection by the soy isoflavone, genistein, via inhibition of mitochondria-dependent apoptosis pathways and reactive oxygen induced-NF-κB activation in a cerebral ischemia mouse model. Neurochem Int. 2012;60:759–767. doi: 10.1016/j.neuint.2012.03.011. [DOI] [PubMed] [Google Scholar]
- 23.Li Q, Lai Y, Wang C, Xu G, He Z, Shang X, Sun Y, Zhang F, Liu L, Huang H. Matrine inhibits the proliferation, invasion and migration of castration-resistant prostate cancer cells through regulation of the NF-κB signaling pathway. Oncol Rep. 2016;35:375–381. doi: 10.3892/or.2015.4341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kasemeier-Kulesa JC, Morrison JA, Lefcort F, Kulesa PM. TrkB/BDNF signalling patterns the sympathetic nervous system. Nat Commun. 2015;6:8281. doi: 10.1038/ncomms9281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ghiglieri V, Sgobio C, Patassini S, Bagetta V, Fejtova A, Giampà C, Marinucci S, Heyden A, Gundelfinger ED, Fusco FR, et al. TrkB/BDNF-dependent striatal plasticity and behavior in a genetic model of epilepsy: Modulation by valproic acid. Neuropsychopharmacology. 2010;35:1531–1540. doi: 10.1038/npp.2010.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Turner BA, Sparrow J, Cai B, Monroe J, Mikawa T, Hempstead BL. TrkB/BDNF signaling regulates photoreceptor progenitor cell fate decisions. Dev Biol. 2006;299:455–465. doi: 10.1016/j.ydbio.2006.08.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Odate S, Nakamura K, Onishi H, Kojima M, Uchiyama A, Nakano K, Kato M, Tanaka M, Katano M. TrkB/BDNF signaling pathway is a potential therapeutic target for pulmonary large cell neuroendocrine carcinoma. Lung Cancer. 2013;79:205–214. doi: 10.1016/j.lungcan.2012.12.004. [DOI] [PubMed] [Google Scholar]
- 28.Kan QC, Lv P, Zhang XJ, Xu YM, Zhang GX, Zhu L. Matrine protects neuro-axon from CNS inflammation-induced injury. Exp Mol Pathol. 2015;98:124–130. doi: 10.1016/j.yexmp.2015.01.001. [DOI] [PubMed] [Google Scholar]
- 29.Xu L, Ning H, Gu L, Wang Q, Lu W, Peng H, Cui W, Ying B, Ross CR, Wilson GM, et al. Tristetraprolin induces cell cycle arrest in breast tumor cells through targeting AP-1/c-Jun and NF-κB pathway. Oncotarget. 2015;6:41679–41691. doi: 10.18632/oncotarget.6149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kuang H, Hua X, Zhou J, Yang R. Resolvin D1 and E1 alleviate the progress of hepatitis toward liver cancer in long-term concanavalin A-induced mice through inhibition of NF-κB activity. Oncol Rep. 2016;35:307–317. doi: 10.3892/or.2015.4389. [DOI] [PubMed] [Google Scholar]


