Abstract
Recently, genome-wide association studies (GWAS) have identified 11 loci associated with adipose-related traits across different populations. However, their functional roles still remain largely unknown. In this study, we aimed to explore the splicing regulation of these GWAS signals in a tissue-specific fashion. For adipose-related GWAS signals, we selected six adipose-related tissues (adipose subcutaneous, artery tibial, blood, heart left ventricle, muscle-skeletal, and thyroid) with the sample size greater than 80 for splicing quantitative trait loci (QTL) analysis using GTEx released datasets. We integrated GWAS summary statistics of nine adipose-related traits (an average of 2.6 million SNPs per GWAS), and splicing QTLs from 6 GTEx tissues with an average of 337,900 splicing QTL SNPs, and 684,859 junctions. Our filtering process generated an average of 86,549 SNPs and 162,841 exon-exon links (junctions) for each tissue. A total of seven exon-exon junctions in four genes (AKTIP, DTNBP1, FTO and UBE2E1) were found to be significantly associated with four SNPs that showed genome-wide significance with body fat distribution (rs17817288, rs7206790, rs11710420 and rs2237199). These splicing events might contribute to the causal effect on the regulation of ectopic-fat, which warrants further experimental validation.
Introduction
Adiposity is one of the leading risk factors for type 2 diabetes1, hypertension and cardiovascular disease2. Recent studies have suggested that body fat distribution, above and beyond generalized adiposity, is an important metric of metabolic health, as specific fat compartments are associated with the corresponding metabolic risk3. Adiposity is highly heritable: the heritability of subcutaneous adipose tissue and visceral adipose tissue was previously estimated to be 57% and 36%, respectively4,5.
Recently, genome-wide association studies (GWAS) of nine traits related to body fat distribution were conducted in up to 9,594 women and 8,738 men of European, African, Hispanic and Chinese ancestries, with and without sex stratification6. A total of 11 loci have been identified from 27 genomic scans and 7 loci are new. To translate the new discoveries into clinical care, the exact physiological mechanisms underlying each genotype-phenotype association remains to be disentangled. Importantly, all the 11 loci are not in the protein-coding regions (Supplementary Table S1). Further analysis of their regulatory roles will be an important task to decipher their genetic susceptibility to body fat.
Alternative splicing is the process by which different splice sites in precursor messenger RNA are selected to produce multiple distinct messenger RNA isoforms. Splicing plays the lead role in shaping the post-transcriptional landscape and is the main mechanism governing protein diversity. Alternative splicing normally affects multi-exon genes often in a cell type-, condition-, tissue- or species-specific fashion7, and modulates phenotypic changes in a flexible and dynamic way8. Disease is often found to be initiated when alternative splicing undergoes dysfunction9. RNA-sequencing (RNA-seq) data is mainly used to study gene level expression. However, it can be used for the quantification of expression splicing variants, such as splice junctions. We refer to the quantitative measurement of isoforms and splicing junctions associated with the genetic variation as splicing QTLs. Splicing QTLs will help identify SNPs that are associated with variation in the expression levels of exon-exon junctions.
In this study, we systematically investigated the events of slicing QTLs of the significant GWAS SNPs for body fat distribution in order to gain a deep understanding of potential mechanisms of the regulatory roles. Augmented with the largest splicing QTLs from the GTEx project10, we identified 7 splicing events involving the SNPs that were significantly associated with 3 body fat distribution phenotypes.
Results
Summary statistics and integration
Twenty-seven GWAS summary statistics datasets for 9 adipose traits with an average of 2.6 million (standard deviation (SD) = 0.2 million; range: 2.4–2.9 million) SNPs for each trait were used in this study. These nine traits are subcutaneous adipose tissue volume (SAT), visceral adipose tissue volume (VAT), visceral adipose tissue volume adjusted for BMI (VATadjBMI), pericardial adipose tissue volume (PAT), pericardial adipose tissue volume adjusted for height and weight (PATadjHtWt), subcutaneous adipose tissue attenuation (SATHU), visceral adipose tissue attenuation (VATHU), ratio of visceral-to-subcutaneous adipose tissue volume (VATSAT), and ratio of visceral-to-subcutaneous adipose tissue volume adjusted for BMI (VATSATadjBMI). For each trait, GWAS was conducted for all samples and for male and female samples respectively, resulting in 3 sets of GWAS summary statistics. Splicing QTL for 10 tissues from GTEx Pilot V3 were downloaded. We used data for six tissues that are related to adipose traits (see Methods). Specifically, 337,900 (SD = 78,241; range: 216,059–414,058) unique SNPs for each tissue from the GTEx Pilot V3 release were included, involving 684,859 (SD = 157,957; range 448,085–830,970) exon-exon links (junctions). By integrating the two datasets, we identified 86,549 (SD = 20,146; range: 51,882–111,640) overlapped SNPs. The overlapped SNPs are involved in 162,841 (SD = 37,547; range: 100,259–210,213) junctions for each tissue. The work flow is summarized in Fig. 1.
Figure 1.
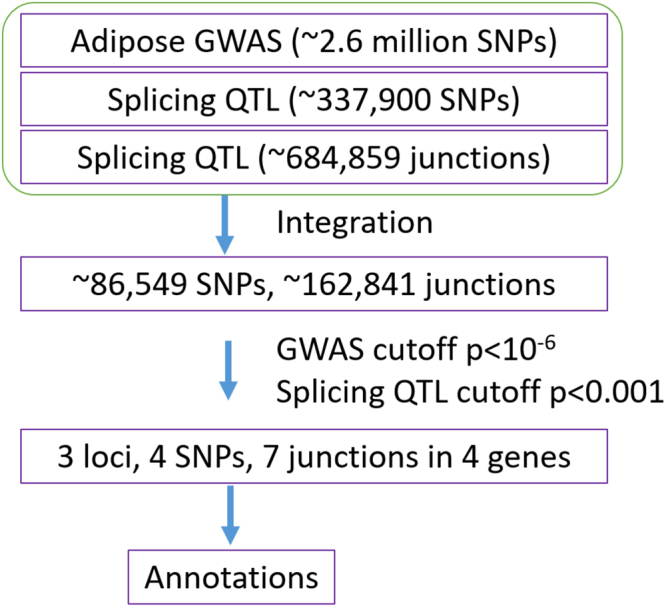
Flow chart for splicing junction identification. Note: ~, the numbers of SNPs or junctions were based on the average from multiple datasets.
Splicing junctions in overall samples
To identify the functional body fat distribution related junctions, summary statistics of nine adipose traits GWAS and 6 GTEx tissues’ splicing QTLs were integrated. At the genome-wide significant level, which is p-value < 5.78 × 10−7 considering 86,549 SNPs, we did not find any significant splicing variants. Considering that this is a stringent multiple test correction, we chose a less stringent threshold to explore the splicing junctions. Specifically, we chose GWAS SNPs with p < 10–6 and splicing QTL with p < 0.001 for exploring the potential causal junctions that are related to adipose GWAS signals. As shown in Fig. 2 and Supplementary Table S2, for overall samples (men and women), we found two SAT GWAS SNPs (rs17817288 [p(GWAS) = 9.50 × 10−9] and rs7206790 [p(GWAS) = 4.20 × 10−9]) and one VATSAT GWAS SNP (rs11710420 [p(GWAS) = 3.20 × 10−8]) were significantly associated with specific expressed junctions in adipose-related tissues. (1) SNP rs17817288 was significantly associated with splicing events of exon 5 - exon 8 junction of FTO gene in adipose subcutaneous (p(splicing QTL) = 6.92 × 10−4). (2) SNP rs17817288 was significantly associated with splicing events of truncated exon 5 - exon 10 junction of AKTIP (AKT interacting protein) gene in artery tibial (p(splicing QTL) = 5.52 × 10−5). (3) SNP rs7206790 was significantly associated with splicing events of truncated and normal exon 6 – exon 9 junctions of AKTIP gene in thyroid (p(splicing QTL) = 7.31 × 10−4 and p(splicing QTL) = 5.73 × 10−4, respectively). (4) SNP rs11710420 was significantly associated with splicing events of exon 2 – exon 4 junction (p(splicing QTL) = 9.19 × 10−4) and exon 3 – exon 4 junction (p(splicing QTL) = 4.09 × 10−4) of UBE2E1 gene (encoding ubiquitin conjugating enzyme E2 E1) in whole blood.
Figure 2.
Schematic view of the 7 junctions in 3 adipose GWAS loci. (A) rs17817288 was associated with junc5.8 of FTO in the combo of SAT_OVERALL GWAS Adipose Subcutaneous, and juncTrunc5.10 of AKTIP in the combo of SAT_OVERALL GWAS and Artery Tibial. SNP rs7206790 was associated with junc6.9 and juncTrunc6.9 of AKTIP in the combo of SAT_OVERALL GWAS and Thyroid. (B) SNP rs11710420 was associated with junc2.4 and junc3.4 of UBE2E1 in the combo of VATSAT_OVERALL GWAS and Whole Blood. (C) SNP rs2237199 was associated with junc4.6 of DTNBP1 in the combo of SATHU_MEN GWAS and Artery Tibial. Rec box: genomic locus in chromosome; green box: exon; purple box: truncated exon. Solid green line: SNP location; solid blue line: intron; and dash line: multiple exons and introns. Black arrow, distance from SNP to junction. Junc: exon-exon junction; juncTrunc: truncated exon-exon junction.
Gender-specific splicing junctions
Gender fat pattern in total and regional body composition is apparent in adults, with men having greater lean tissue mass and a more central fat pattern compared with the more peripheral fat distribution typically observed in adult women11. To identify the gender-specific splicing events that are associated with the GWAS loci for body fat distribution, we used the same pipeline as the overall samples for the 9 traits. We found that SNP rs2237199, a male-specific GWAS signal that was associated with SATHU (p(GWAS) = 1.40 × 10–8), was significantly associated with the splicing event of exon 4 – exon 6 junction of DTNBP1 gene (encoding dystrobrevin binding protein 1) (p = 8.30 × 10–4) (Fig. 2 and Supplementary Table S2). The splicing results of the 7 junctions across the 27 GWAS summary statistics datasets for body fat distribution traits were shown in Supplementary Figure S1 and Supplementary Table S3.
Linkage disequilibrium (LD) of the splicing QTL and top SNPs in GWAS
Considering that three of the four identified GWAS SNPs were missing in the GTEx Pilot V3 splicing QTL, LD was estimated using European population by LDlink (https://analysistools.nci.nih.gov/LDlink/?tab=home). We found the LDs between the identified splicing QTLs and the 11 GWAS top SNPs were strong. The LD values of splicing QTLs (rs17817288 and rs7206790) and GWAS genome-wide significant SNP rs7185735 (chr16:53822651, hg19) were: D′ = 0.937 and 1; r 2 = 0.604 and 0.628, respectively. Moreover, the LD values of the splicing QTLs (rs11710420) and GWAS top signal rs7374732 (chr3:23203454, hg19) were: D′ = 0.937 and r 2 = 0.661. Our examination found one of the four identified SNP, rs2237199, was among the 11 GWAS top hits (Supplementary Tables S1 and S2).
Discussion
Recent genome-wide association studies have identified 11 loci (i.e. SNPs) associated with fat tissue distributions. However, until most recently, little has known about the molecular function for most of the genome-wide significant SNPs with the phenotype in examination. In this study, by integrating GWAS summary statistics of nine body fat distribution traits with splicing QTL data from six adipose metabolism related tissues, we detected that 7 splicing events in four genes (AKTIP, DTNBP1, FTO, and UBE2E1) were significantly associated with four GWAS top signals for body fat distribution (rs17817288, rs7206790, rs11710420 and rs2237199; all were genome-wide significant). Our finding, although needs further extensive validation, promoted our understanding of the underlying regulatory mechanism of body fat distribution.
Fat tissue distribution has been reported to be strongly related to the development of type 2 diabetes1, coronary artery disease2, blood, and serum lipids12. The more recent hypothesis-free GWAS of those traits have systematically evaluated the predisposed genomic loci by using more than 10,000 human subjects (Supplementary Table S4). For adipose tissue distribution assessment to be clinically useful, the ideal adiposity phenotype should provide a single risk estimate that captures the separate ‘effects’ of diverse adiposity traits. The currently available largest studies have the greatest statistical power, and allow direct comparison to other related traits with available GWAS data (cross-trait analysis). Supplementary Table S4 showed that rs7206790 and rs17817288 reached genome-wide significant level for Type 2 diabetes among other 6 adipose-related traits, which indicated that the two SNPs shared genetic contributions to both adipose tissue distribution and type 2 diabetes (i.e. pleiotropy), while rs11710420 is likely a unique marker for adipose tissue distribution. The genetic heritability should be considered as a variable for differentiated splicing regulations.
In addition to body fat distribution traits, rs17817288 was also reported to be associated with predisposition to obesity in multiple populations13–15. And the associations with the combined samples were mostly contributed by the Caucasian group due to great genetic differentiation16. And this SNP was also reported to be associated with cancer17. SNP rs7206790 was reported to be associated with risk of obesity18 and breast cancer risk19. No other traits were reported with their susceptibilities from SNPs rs11710420 and rs2237199.
In this study, 6 adipose-related tissues (adipose subcutaneous, artery tibial, blood, heart left ventricle, muscle-skeletal and thyroid) were selected for splicing QTL investigation that are related to body fat distribution GWAS signals. Obesity is one of the most pervasive, chronic diseases, which is defined as excess adipose tissue. It is a significant independent risk factor for cardiovascular disease and other co-morbidities20. Adipose tissue is considered the best indicator of long-term essential fatty acid intake, but other tissues may prove equally valid such as whole blood21. Ectopic-fat depots are associated with cardio metabolic risk and cardiovascular events22. Pericardial adipose tissue represents distinct ectopic-fat deposition around the heart. The maintenance of energy balance is regulated by complex homeostatic mechanisms, including those emanating from adipose tissue. The main function of the adipose tissue is to store the excess of metabolic energy in the form of fat. Thyroid hormone regulates metabolic processes essential for regulating metabolism in the adult23. Thyroid dysfunction has a great impact on lipids as well as a number of other cardiovascular risk factors.
In this study, splicing events within the four genes were identified to be strongly associated with adipose GWAS. FTO (fat mass- and obesity-associated) is the most investigated gene in obesity and has complex molecular mechanisms that are yet to be elucidated. The genetic variation in the FTO gene was associated with metabolisms traits such as increased body weight, total fat mass, lean body mass, increased insulin secretion, and reduced insulin sensitivity24. In mouse models, inactivation of the FTO gene results in lean phenotype, whereas overexpression of FTO leads to increased food intake and obesity. The function of genes AKTIP and UBE2E1 (encoding ubiquitin conjugating enzyme E2 E1) is still unclear. DTNBP1 is one of most studied genes that contributes to the risk of mental health disorders. It was speculated to be involved in the modulation of glutamatergic neurotransmission in the brain25.
Traditional expression QTLs mainly focused on gene-level, but each gene can produce a multitude of splicing variants or isoforms. Furthermore, different isoform may be expressed in different tissues or at different developmental periods and, thus, exert unique biological functions. The accumulation of RNA-seq data especially in different tissues and disease conditions will further consolidate the tissue-specificity of some of these junctions and their corresponding isoforms. We identified 7 splicing junctions that represented a specific cluster of transcripts for the genes, which might be the underlying effect factors, considering that each gene typically has multiple transcripts. Interestingly, no significant eQTLs were found for the four SNPs in all tissues based on GTEx Portal (https://www.gtexportal.org/home/), suggesting the subtle and specific regulation of potential genetic isoforms in the adipose developmental process. There are a few limitations in the current study. First, LD trimming was not performed when examining overlapped SNPs between GWAS and sQTLs. The LD values were estimated when splicing SNPs were identified to be related to adipose-associated variations. The 7 identified junctions could not survive after multiple test corrections if we applied stringent Bonferroni correction method for the total of 162,841 junctions (Bonferroni or false discovery rate corrected significant cutoff p-value = 3.07 × 10–7). The experimental validation for the isoforms is also needed considering the complexity of the traits. For example, nominal significance was found for NRG3 isoforms26 and NRG1 isoforms27. And replications should be performed when other adipose RNA-seq samples are available. Importantly, the junctions we identified here are based on low SNP density, and until fine-mapping and functional investigations are completed it remains unclear whether their association with body fat distribution is driven by other causal variants in the region. Furthermore, current splicing QTLs provide an incomplete picture of splicing variants involved in body fat distribution, and additional adipose genetic transcript-specific junctions will be discovered as sample size increases.
To the best of our knowledge, this is the first systematical investigation of splicing architecture using genome-wide loci associated with the body fat distribution. We refined and identified 7 splicing junctions in AKTIP, DTNBP1, FTO, and UBE2E1, thereby providing a window into the biological processes that cause the fat tissue distribution traits. Future studies are necessary to validate these 7 junctions, and the full-length transcript clusters that cover those junctions should be determined and further explored for their mechanisms on how these transcripts affect adipocyte biology and how their perturbations contribute to systemic metabolic disease.
Methods
Splicing QTL datasets
Splicing QTL datasets were downloaded from GTEx Portal in section GTEx Analysis Pilot V3 (https://www.gtexportal.org/home/datasets)10. GTEx provides splicing QTLs for 10 tissues. In our study, we chose six GTEx tissues that were likely related to adipose-related phenotypes: adipose subcutaneous (n = 94), artery tibial (n = 112), blood (n = 156), heart left ventricle (n = 83), muscle-skeletal (n = 138), and thyroid (n = 105). The genotyping data for these tissue samples were generated by Illumina’s Human Omni5-Quad and InfiniumExomeChip. After imputation, a total of 6,820,471 autosomal SNPs were used for splicing QTL analysis. Splicing QTLs were calculated for each of the 6 tissues that had sufficient sample size (>80 donors) for all SNPs within ±1 Mb of the transcription start site of each gene10.
GWAS datasets
GWAS summary statistics data from meta-analyses were downloaded from the VATGen Consortium6 for the following traits: SAT, VAT, VATadjBMI, PAT, PATadjHtWt, SATHU, VATHU, VATSAT, and VATSATadjBMI. The original genotyping data were generated on multiple platforms and imputation was performed based on the HapMap Phase 2 data, resulting in ~2.6 million SNPs.
Integration and annotation
All data analyses were performed using R software with version 3.4.1. The SNP and gene coordinates were based on human reference genome hg19, and reference transcripts were referred to UCSC Genome Browser and NCBI RefSeqGene (Homo sapiens Release 108, 2016-06-07).
Electronic supplementary material
Acknowledgements
We thank the investigators of Genotype-Tissue Expression (GTEx) Project and those who generated the GWASs for sharing the data. Z.Z. was partially supported by National Institutes of Health grant (R01LM012806). The funders did not participate in the study design, data analysis, decision to publish, or preparation of the manuscript.
Author Contributions
L.M., P.J. and Z.Z. conceived the study. L.M. collected the data, conducted the bioinformatics. All authors contributed to the writing and editing of the manuscript. All authors read and approved the final manuscript.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-017-18767-z.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Meisinger C, Doring A, Thorand B, Heier M, Lowel H. Body fat distribution and risk of type 2 diabetes in the general population: are there differences between men and women? The MONICA/KORA Augsburg cohort study. Am J Clin Nutr. 2006;84:483–489. doi: 10.1093/ajcn/84.3.483. [DOI] [PubMed] [Google Scholar]
- 2.Canoy D. Distribution of body fat and risk of coronary heart disease in men and women. Curr Opin Cardiol. 2008;23:591–598. doi: 10.1097/HCO.0b013e328313133a. [DOI] [PubMed] [Google Scholar]
- 3.Poirier P, Despres JP. Waist circumference, visceral obesity, and cardiovascular risk. J Cardiopulm Rehabil. 2003;23:161–169. doi: 10.1097/00008483-200305000-00001. [DOI] [PubMed] [Google Scholar]
- 4.Fox CS, et al. Genome-wide association for abdominal subcutaneous and visceral adipose reveals a novel locus for visceral fat in women. PLoS Genet. 2012;8:e1002695. doi: 10.1371/journal.pgen.1002695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fox CS, et al. Abdominal visceral and subcutaneous adipose tissue compartments: association with metabolic risk factors in the Framingham Heart Study. Circulation. 2007;116:39–48. doi: 10.1161/CIRCULATIONAHA.106.675355. [DOI] [PubMed] [Google Scholar]
- 6.Chu AY, et al. Multiethnic genome-wide meta-analysis of ectopic fat depots identifies loci associated with adipocyte development and differentiation. Nat Genet. 2017;49:125–130. doi: 10.1038/ng.3738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wang ET, et al. Alternative isoform regulation in human tissue transcriptomes. Nature. 2008;456:470–476. doi: 10.1038/nature07509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Braunschweig U, Gueroussov S, Plocik AM, Graveley BR, Blencowe BJ. Dynamic integration of splicing within gene regulatory pathways. Cell. 2013;152:1252–1269. doi: 10.1016/j.cell.2013.02.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lara-Pezzi E, Gomez-Salinero J, Gatto A, Garcia-Pavia P. The alternative heart: impact of alternative splicing in heart disease. J Cardiovasc Transl Res. 2013;6:945–955. doi: 10.1007/s12265-013-9482-z. [DOI] [PubMed] [Google Scholar]
- 10.GTEx_Consortium. The Genotype-Tissue Expression (GTEx) pilot analysis: Multitissue gene regulation in humans. Science348, 648–660 (2015). [DOI] [PMC free article] [PubMed]
- 11.Kissebah AH, Krakower GR. Regional adiposity and morbidity. Physiol Rev. 1994;74:761–811. doi: 10.1152/physrev.1994.74.4.761. [DOI] [PubMed] [Google Scholar]
- 12.Anderson AJ, et al. Body fat distribution, plasma lipids, and lipoproteins. Arteriosclerosis. 1988;8:88–94. doi: 10.1161/01.ATV.8.1.88. [DOI] [PubMed] [Google Scholar]
- 13.Apalasamy YD, Ming MF, Rampal S, Bulgiba A, Mohamed Z. Genetic association of SNPs in the FTO gene and predisposition to obesity in Malaysian Malays. Braz J Med Biol Res. 2012;45:1119–1126. doi: 10.1590/S0100-879X2012007500134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang G, et al. Common SNPs in FTO gene are associated with obesity related anthropometric traits in an island population from the eastern Adriatic coast of Croatia. PLoS One. 2010;5:e10375. doi: 10.1371/journal.pone.0010375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Karns R, et al. Common variants in FTO are not significantly associated with obesity-related phenotypes among Samoans of Polynesia. Ann Hum Genet. 2012;76:17–24. doi: 10.1111/j.1469-1809.2011.00686.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tan LJ, et al. Replication of 6 obesity genes in a meta-analysis of genome-wide association studies from diverse ancestries. PLoS One. 2014;9:e96149. doi: 10.1371/journal.pone.0096149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Figlioli G, et al. A Comprehensive Meta-analysis of Case-Control Association Studies to Evaluate Polymorphisms Associated with the Risk of Differentiated Thyroid Carcinoma. Cancer Epidemiol Biomarkers Prev. 2016;25:700–713. doi: 10.1158/1055-9965.EPI-15-0652. [DOI] [PubMed] [Google Scholar]
- 18.Xu Y, et al. Rs7206790 and rs11644943 in FTO gene are associated with risk of obesity in Chinese school-age population. PLoS One. 2014;9:e108050. doi: 10.1371/journal.pone.0108050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kaklamani V, et al. The role of the fat mass and obesity associated gene (FTO) in breast cancer risk. BMC Med Genet. 2011;12:52. doi: 10.1186/1471-2350-12-52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pi-Sunyer X. The medical risks of obesity. Postgrad Med. 2009;121:21–33. doi: 10.3810/pgm.2009.11.2074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Baylin A, et al. Fasting whole blood as a biomarker of essential fatty acid intake in epidemiologic studies: comparison with adipose tissue and plasma. Am J Epidemiol. 2005;162:373–381. doi: 10.1093/aje/kwi213. [DOI] [PubMed] [Google Scholar]
- 22.Britton KA, et al. Body fat distribution, incident cardiovascular disease, cancer, and all-cause mortality. J Am Coll Cardiol. 2013;62:921–925. doi: 10.1016/j.jacc.2013.06.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Brent GA. Mechanisms of thyroid hormone action. J Clin Invest. 2012;122:3035–3043. doi: 10.1172/JCI60047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Loos RJ, Yeo GS. The bigger picture of FTO: the first GWAS-identified obesity gene. Nat Rev Endocrinol. 2014;10:51–61. doi: 10.1038/nrendo.2013.227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Guo AY, et al. The dystrobrevin-binding protein 1 gene: features and networks. Mol Psychiatry. 2009;14:18–29. doi: 10.1038/mp.2008.88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Paterson C, et al. Temporal, Diagnostic, and Tissue-Specific Regulation of NRG3 Isoform Expression in Human Brain Development and Affective Disorders. Am J Psychiatry. 2017;174:256–265. doi: 10.1176/appi.ajp.2016.16060721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Paterson C, Wang Y, Kleinman JE, Law AJ. Effects of schizophrenia risk variation in the NRG1 gene on NRG1-IV splicing during fetal and early postnatal human neocortical development. Am J Psychiatry. 2014;171:979–989. doi: 10.1176/appi.ajp.2014.13111518. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.



