Abstract
This study aimed to determine whether bisphosphonates exert an effect on preventing femoral head collapse after osteonecrosis of the femoral head (ONFH) in an animal model and in clinical trials. A systematic literature search was performed for studies published up to January 2017. Twenty-three articles (16 animal studies, seven clinical trials) were included in the meta-analysis. We found that the bisphosphonate group obtained significant improvement in epiphyseal quotients (MD = 15.32; 95% CI, 9.25–21.39) and provided better performance on bone volume (SMD = 1.57; 95% CI, 0.94–2.20), trabecular number (SMD = 1.30; 95% CI, 0.80–1.79), trabecular thickness (SMD = 0.77; 95% CI, 0.10–1.43) and trabecular separation (SMD = −1.44; 95% CI, −1.70 to −0.58) in the animal model. However, the bisphosphonate group did not achieve better results in pain score, Harris score, the occurrence rate of femoral head collapse, or total hip arthroplasty in the clinical trials. In conclusion, despite bisphosphonates significantly improving bone remodeling outcomes in animal models, no significant efficacy was observed in the treatment of ONFH in the clinical studies. Further studies are required to solve the discordant outcomes between the animal and clinical studies.
Introduction
Osteonecrosis of the femoral head (ONFH) is a common debilitating disease that occurs in young and middle-aged adults1,2. In fact, children also suffer from ONFH with an incidence of 8.5–21 per 100 000, but in this population, it is called Perthes disease3,4. Although the progressions of adult ONFH and Perthes disease differ, both conditions result in femoral head deformity or collapse. Therefore, preventing femoral head collapse is a significant treatment goal5,6. The pathogenesis of ONFH remains unclear, but an imbalance of bone metabolism is considered one of the most important causes7. When ONFH occurs, bone formation fails to keep pace with bone resorption, resulting in low bone mineral density in the femoral head and the progression to collapse8. Therefore, clinicians must take measures to reduce bone resorption and improve osteogenesis when treating ONFH.
Bisphosphonates are a class of drugs that can bind to the bone and inhibit osteoclast activity by reducing bone resorption9–11. They are usually used to treat diseases involving bone resorption progression, such as osteoporosis, Paget’s disease, and fibrous dysplasia11–13. Bisphosphonates has also been considered a promising medication for early ONFH and preventing femoral head collapse14–17. However, the efficiency of this kind of drug in both animal studies18,19 and clinical trials20,21 remains controversial. Furthermore, a meta-analysis of a small number of clinical studies reported that the use of bisphosphonates cannot prevent femoral head collapse or delay total hip replacement after ONFH22. Consequently, the use of bisphosphonates in the early stage of ONFH seems to involve some challenges.
To evaluate the effect of bisphosphonates on preventing femoral head collapse after osteonecrosis, we identified all related animal studies and clinical trials from the electronic database and conducted this meta-analysis comprehensively to judge whether bisphosphonates should be recommended to ONFH patients and are worthy of further study.
Materials and Methods
This meta-analysis conformed to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement23.
Search strategy
The study’s search protocol was developed on December 20, 2016. An electronic search was conducted online to identify relevant studies published up to January 2017 in PubMed; Ovid MEDLINE(R) (1946 to present with daily update); all EBM reviews; the ISI Web of Science; Academic Search Premier, and MEDLINE in EBSCO; Cochrane Library databases; CBM; and CNKI databases using the following terms: (ibandronate or alendronate or bisphosphonate or zoledronate or pamidronate or clodronate) AND (osteonecrosis of the femoral head or femoral head necrosis or Perthes disease) in all fields. In addition, the reference lists of the retrieved articles were manually searched for further pertinent studies. Furthermore, we contacted the study authors for the raw data and to complete the search strategy whenever possible. The two investigators independently selected potential eligible studies and any discrepancy between them was resolved by consensus.
Data extraction
The data collection was conducted by two investigators independently and the result was checked by a third investigator. Discrepancies were settled by group discussion. Collected data included the first author’s surname, publication year, study location, study design, sample size, type of medicine or dose range, and route of medication delivery. For the animal studies, we extracted the characteristics of the animal models including species, animal age, weight, and sex; and outcomes including mean epiphyseal quotients (EQ), trabecular bone volume (BV), trabecular separation (TS), trabecular thickness (TT), and trabecular number (TN). When various methods were presented and more than two groups were analyzed in a single study, the data were assessed as two comparisons of those exposed to bisphosphonates if necessary. For clinical trials, we recorded pain scores, Harris scores, and femoral head collapse and THA occurrence rates.
Assessment of methodological quality
Two reviewers independently assessed the methodology of the included animal studies using updated Stroke Therapy Academic Industry Roundtable recommendations24 and that of the clinic studies using the modified Jadad scale25. The methodological quality of each individual study was scored against the following criteria: sample size calculation; inclusion and exclusion criteria; randomization; allocation concealment; reporting of objects excluded from the analysis; blinded assessment of the outcomes; and report of potential conflicts of interest and study funding. Each item was allocated one point for a quantitative appraisal of overall quality of the individual studies. Each animal study was given a quality score out of a possible total of seven points and the group median was calculated. The modified Jadad scale evaluated the clinical studies in terms of randomization (2 points); concealment of allocation (2 points); double blinding (2 points); and total withdrawals and dropouts (1 point). Clinical studies achieving a score of >4 points were considered of high quality.
Statistical analysis
This meta-analysis was conducted using Review Manager Software (Revman 5.3, Cochrane Collaboration, Oxford, United Kingdom). For the animal studies, we divided them into Perthes model and mature ONFH model subgroups and analyzed the pooled results. The relative risk (RR) was used to measure the dichotomous outcomes, while the mean difference (MD) was used to analyze continuous outcomes, both with 95% confidence intervals (CI). When the parameter was measured using different methods, standard mean difference (SMD) was used to compare continuous outcomes (e.g. BV, TT, TN, and TS). Chi squared tests and the I2 statistic were used to evaluate statistical heterogeneity. An I2 > 50% was considered to indicate significantly statistical heterogeneity and the random-effect or fixed-effect model was used. Publication bias was visually examined using funnel plots. Values of P < 0.05 were considered statistically significant. The sensitivity analysis was performed to explore the impact of an individual study by the exclusion of one study each time. Publication bias was visually examined using funnel plots.
Results
Search results and study characteristics
As shown in Fig. 1, a total of 508 potentially relevant articles were identified from the databases. Of them, 279 were screened. After a title and abstract screen, 230 were excluded. A total of 49 full-text articles were assessed for eligibility, but 26 were excluded for different reasons (10 review articles, eight studies lacked a control group, and data for eight studies were unavailable). The remaining 23 articles including 16 animal studies16–19,26–37 and seven human studies14,15,20,21,38–40 were passed for synthetic evaluation for this meta-analysis. A summary of selected studies is shown in Table 1, while the basic techniques of the included studies are shown in Table 2. As Tables 3 and 4 present, the median quality score of the reported animal studies was 4 (range, 3–6), while the median of modified Jadad score of the clinical studies was 5 (range, 4–7).
Figure 1.
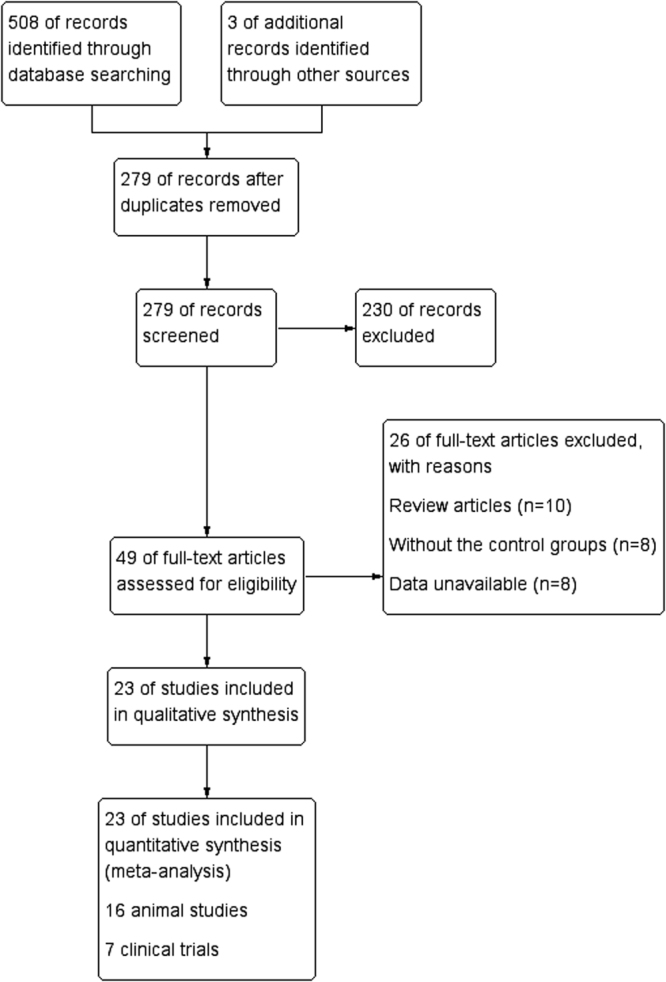
Flow chart of study selection.
Table 1.
Summary of the basic information.
| Author | Year | Country | Study Object | SG (n/hip) | Age | Sex,M/F | CG (n/hip) | Age | Sex,M/F |
|---|---|---|---|---|---|---|---|---|---|
| Animal studies | |||||||||
| Kim HK(1) | 2005 | USA | piglet | 8 | 4–5 w | male | 8 | 4–5 w | male |
| Aya-ay J | 2007 | Germany | piglet | 5 | 6–9 w | male | 4 | 6–9 w | male |
| Vandermeer JS | 2011 | USA | piglet | 5 | 6–8 w | male | 5 | 6–8 w | male |
| Kim HK (2) | 2014 | USA | piglet | 6 | 6–8 w | male | 6 | 6–8 w | male |
| Cheng TL | 2014 | Australia | piglet | 4 | 6–8 w | male | 4 | 6–8 w | male |
| Zou Y | 2015 | USA | piglet | 6 | 2–3 w | male | 9 | 2–3 w | male |
| Aruwajoye OO | 2016 | USA | Piglet | 10 | 5–8 w | male | 10 | 5–8 w | Male |
| Little DG(1) | 2003 | Australia | Rat | 16 | 14 w | female | 8 | 14 w | Female |
| Little DG(2) | 2005 | Australia | Rat | 32 | 4 w | female | 16 | 4 w | Female |
| Fan M | 2012 | China | Rat | 8 | NR | male | 8 | NR | Male |
| Xu XL | 2015 | China | Rat | 10 | 8 w | male | 10 | 8 w | male |
| Hofstaetter JG | 2009 | USA | Rabbit | 15 | 10–12 m | male | 15 | 10–12 m | Male |
| Xu XL | 2015 | China | Rat | 10 | 8 w | male | 10 | 8 w | male |
| Xie XW | 2013 | China | Rat | 30 | 8 w | unlimited | 30 | 8 w | unlimited |
| Tan G | 2013 | China | Rat | 60 | 8 w | unlimited | 60 | 8 w | unlimited |
| Xin DS(1) | 2014 | China | Rat | 15 | NR | male | 15 | NR | male |
| Xin DS(2) | 2014 | China | Rat | 15 | NR | male | 15 | NR | male |
| Clinical studies | |||||||||
| Wang CJ | 2008 | Taiwan | Human | 23/30 | 35.7 ± 4.7y | 13/10 | 25/30 | 38.6 ± 12.6y | 20/5 |
| Chen CH | 2012 | Taiwan | Human | 26/32 | 48.4 ± 11.4y | 22/4 | 26/33 | 44.2 ± 9.2y | 19/7 |
| Nishii T | 2006 | Japan | Human | 14/20 | 48y | 07/7 | Aug-13 | 36y | 07/1 |
| Lai KA | 2005 | Taiwan | Human | 20/29 | 42.6y | NR | 20/25 | 42.4y | NR |
| Lee YK | 2015 | Korea | Human | 55/55 | 43.8 ± 11.8y | 39/1 | 55/55 | 45.2 ± 11.6y | 41/1 |
| Kang P | 2012 | China | Human | 39/55 | 43.8y | 6 | 40/52 | 45.3y | 4 |
| Gao yan | 2016 | China | Human | 32/32 | 23–55y | 37/18 | 32/32 | 23–55y | 35/17 |
| 21/11 | 22/10 | ||||||||
SG: study group or bisphosphonates group; CG: control group. unlimited means animal gender is not limited. n/hip: animal number/hip number. w- weeks. y: years old. m: month. NR: not report.
Table 2.
Detail information of the research technique.
| Author | Kind of ONFH | Study design | Drug delivery | Dosage | Study period |
|---|---|---|---|---|---|
| Animal studies | |||||
| Kim HK(1) | Traumatic | Ibandronate VS placebo | Subcutaneous | 44.4 μg/kg,3 × per/w × 6 w | 8 weeks |
| Aya-ay J | Traumatic | Ibandronate VS placebo | intraosseous | 0.28 mg or 0.56 mg, once | 7 weeks |
| Vandermeer JS | Traumatic | Ibandronate VS placebo | intraosseous | 0.56 mg, once | 8 weeks |
| Kim HK (2) | Traumatic | Ibandronate+BMP VS BMP | intraosseous | 0.6 mg, once | 8 weeks |
| Cheng TL | Traumatic | Zoledronic+BMP VS BMP | intraosseous | 0.25 mg, once | 8 weeks |
| Zou Y | Traumatic | Clodronate VS placebo Clodronate + Sim VS Sim | Intraosseous | 2.2 mg, once | 6 weeks |
| Aruwajoye OO | Traumatic | Ibandronate VS placebo BMP+Ibandronate VS BMP Zoledronic acid VS placebo | Intraosseous | 0.6 mg, once | 8 weeks |
| Little DG(1) | Traumatic | Zoledronic acid VS placebo | Subcutaneous | 0.1 mg/kg/w × 3 w | 6 weeks |
| Little DG(2) | Traumatic | Zoledronic acid VS placebo | Subcutaneous | 0.15 mg/kg in total | 15 weeks |
| Fan M | Traumatic | PLGA+zoledronate VS PLGA | Subcutaneous | 0.1 mg/kg,once | 5 weeks |
| Xu XL | Traumatic | Alendronate VS placebo | Implanted | 30 μg, once | 6 weeks |
| Hofstaetter JG | Traumatic | alendronate+Lova VS Lova | Subcutaneous | 150 μg/kg, 3 × per/w × 2 w | 24 weeks |
| Xie XW | Steroid | alendronate VS control | Oral | 150 μg/(kg.d) × 4 w | 12 weeks |
| Tan G | Steroid | alendronate VS control | Oral | 150 μg/(kg.d) × 4 w | 12 weeks |
| Xin DS(1) | Traumatic | alendronate VS control | Intragastric | 1 mg/(kg.d) × 2 w | 5 weeks |
| Xin DS(2) | Traumatic | alendronate VS control | Intragastric | 1 mg/(kg.d) 2 w | 5 weeks |
| Clinical studies | |||||
| Wang CJ | Alcohol, steroid | ESWT+alendronate VS ESWT | Oral | 70 mg/w for 1 year | 25 months |
| Chen CH | Alcohol, steroid | Alendronate +Ca+vitD VS Ca+vit D | Oral | 70 mg/w for 2 year | 24 months |
| Nishii T | Alcohol,steroid, Idiopathic | Alendronate VS placebo | Oral | 5 mg/day for 1 year | 12 months |
| Lai KA | Alcohol,steroid, Idiopathic | Alendronate VS placebo | Oral | 70 mg/w for 25 week | 24 months |
| Lee YK | Alcohol,steroid, Idiopathic | Zoledronate+Ca+vitD VS Placebo+Ca+vit D | Intravenous | 5 mg/year for 2 year | 24 months |
| Kang P | Alcohol,steroid, Idiopathic | Alendronate+MD+Ca+vitD VS PlaceboMD+Ca+vit D | Oral | 10 mg/day or 70 mg/w for 24 weeks | 63 months |
| Gao Y | steroid | alendronate+Ca VS placebo +Ca | Oral | 70 mg/week for 6 month | 6 months |
BMP: Bone Morphogenetic Protein; PLGA: Poly (lactic-co-glycolic acid); sim: Simvastatin; Lova: Lovastatin; ESWT: extracorporeal shockwave therapy.
Table 3.
The methodological quality of individual study.
| Authors | (1) | (2) | (3) | (4) | (5) | (6) | (7) | Total score |
|---|---|---|---|---|---|---|---|---|
| Animal studies | ||||||||
| Kim HK(1) | * | * | * | * | 4 | |||
| Aya-ay J | * | * | * | * | 4 | |||
| Vandermeer JS | * | * | * | 3 | ||||
| Kim HK (2) | * | * | * | * | 4 | |||
| Cheng TL | * | * | * | * | * | 5 | ||
| Zou Y | * | * | * | * | 4 | |||
| Aruwajoye OO | * | * | * | * | 4 | |||
| Little DG(1) | * | * | * | * | 4 | |||
| Little DG(2) | * | * | * | * | 4 | |||
| Fan M | * | * | * | * | * | 5 | ||
| Xu XL | * | * | * | * | 4 | |||
| Hofstaetter JG | * | * | * | * | 4 | |||
| Xie XW | * | * | * | * | 4 | |||
| Tan G | * | * | * | * | 4 | |||
| Xin DS(1) | * | * | * | * | 4 | |||
| Xin DS(2) | * | * | * | * | 4 | |||
*One score means the studies fulfilling one of the criteria of (1) sample size calculation; (2) inclusion and exclusion criteria; (3) randomisation; (4) allocation concealment; (5) reporting of objects excluded from analysis; (6) blinded assessment; (7) reporting potential conflicts of interest and study funding.
Table 4.
Modified Jadad Score for clinical trials.
| Study | Randomization | Concealment of allocation | Double blinding | Total Withdrawals and dropouts | Total |
|---|---|---|---|---|---|
| Wang CJ | ** | ** | * | 5 | |
| Chen CH | ** | ** | ** | * | 7 |
| Nishii T | * | * | * | * | 4 |
| Lai KA | ** | ** | * | 5 | |
| Lee YK | ** | ** | ** | * | 7 |
| Kang P | ** | * | ** | * | 6 |
| Gao Y | ** | ** | * | 5 |
Each asterisk represents one point. Modified Jadad score is used to evaluate the quality of articles and studies achieving a score of ≥4 points were considered to be of high quality.
Primary analysis of animal studies
Evaluation of femoral head sphericity
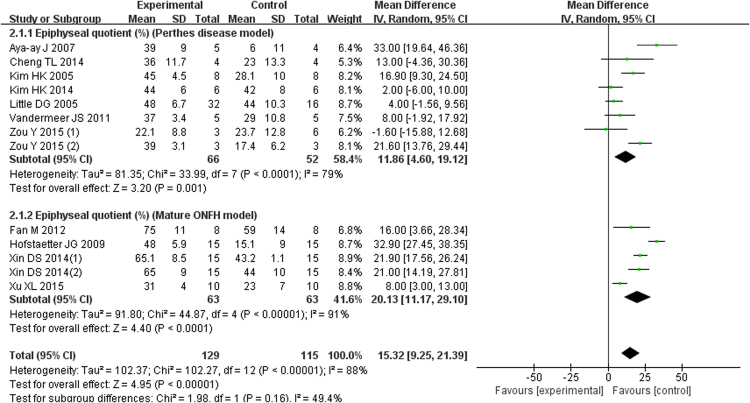
Outcome evaluation and measurement methods are shown in Table 5. We found that the included animal studies recorded similar outcomes in the summary. Sphericity measurements of the femoral head were derived using a modified EQ, which indicated the height at the center of the femoral head over the width. Seven studies of 118 Perthes disease animal models recorded the EQ, which was significantly increased in the experimental group using bisphosphonates compared with the control group (MD = 11.86; 95% CI, 4.60–19.12; Fig. 2). Five studies of a mature ONFH model recorded the EQ, and the result indicated that the bisphosphonates treatment had better outcomes (MD = 20.13; 95% CI, 11.17–29.10; Fig. 2). The pooled results of the two animal models were also significantly better in the experimental group (MD = 15.32; 95% CI, 9.25–21.39; Fig. 2).
Table 5.
Outcome evaluation and measurement method of animal studies.
| Study | Outcome evaluation | Measurement method |
|---|---|---|
| Animal studies | ||
| Kim HK(1) | EQ | X Radiographs |
| Trabecular parameters (BV,TT,TN,TS) | Histomorphometry | |
| Aya-ay J | EQ | X Radiographs |
| Trabecular parameters (BV,TT,TN,TS) | Histomorphometry | |
| Vandermeer JS | EQ | X Radiographs |
| Trabecular parameters (BV,TT,TN,TS) | Histomorphometry | |
| Kim HK (2) | EQ | X Radiographs |
| Trabecular parameters (BV,TT,TN,TS) | Histomorphometry | |
| Cheng TL | EQ | X Radiographs |
| Zou Y | EQ | X Radiographs |
| Trabecular parameters (BV,TT) | Micro-Quantitative-CT | |
| Aruwajoye OO | Trabecular parameters (BV) | Histomorphometry |
| Little DG(1) | Trabecular parameters (BV,TT,TN) | Histomorphometry |
| Little DG(2) | EQ | X Radiographs |
| Trabecular parameters (BV,TT,TN) | Histomomhometrv | |
| Fan M | EQ | X Radiographs |
| Trabecular parameters (BV,TT,TS) | Micro-Quantitative-CT | |
| Xu XL | EQ | X Radiographs |
| Trabecular parameters (BV,TT,TS) | Histomorphometry | |
| Hofstaetter JG | EQ | Micro- CT image |
| Trabecular parameters (TT) | Micro-Quantitative-CT | |
| Xie XW | Trabecular parameters (TT,TN,TS) | Micro-Quantitative-CT |
| Tan G | Trabecular parameters (TT,TN,TS) | Micro-Quantitative-CT |
| Xin DS(1) | EQ | X Radiographs |
| Trabecular parameters (BV,TT,TN,TS) | Micro-Quantitative-CT | |
| Xin DS(2) | EQ | X Radiographs |
| Trabecular parameters (BV,TT,TN,TS) | Micro-Quantitative-CT | |
EQ: Epiphyseal quotients; BV: bone volume; TT: Trabecular thickness; TN: trabecular number; TS: Trabecular separation.
Figure 2.
Forest plot showing Epiphyseal quotient.
Parametric analysis of bone trabeculae
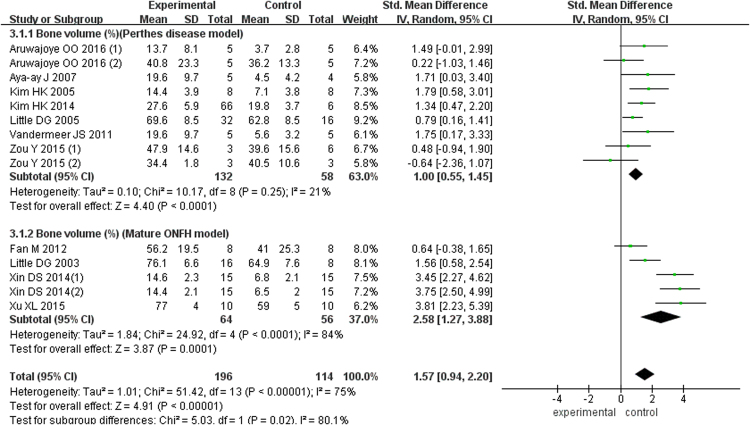
Trabecular bone volume (BV/TV) was presented in seven articles involving 190 animals with Perthes disease, and this meta-analysis demonstrated that animals in the bisphosphonate group had greater BV improvement (MD = 1.00; 95% CI, 0.55–1.45). Five studies of 120 mature ONFH models showed better BV/TV performance in the experimental group (SMD = 2.58; 95% CI, 1.27–3.88). The pooled outcomes were also higher in the bisphosphonate group (SMD = 1.57; 95% CI, 0.94–2.20) (Fig. 3).
Figure 3.
Forest plot showing bone volume.
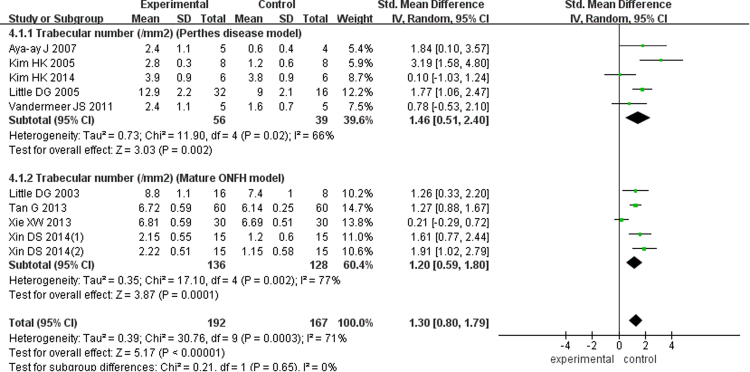
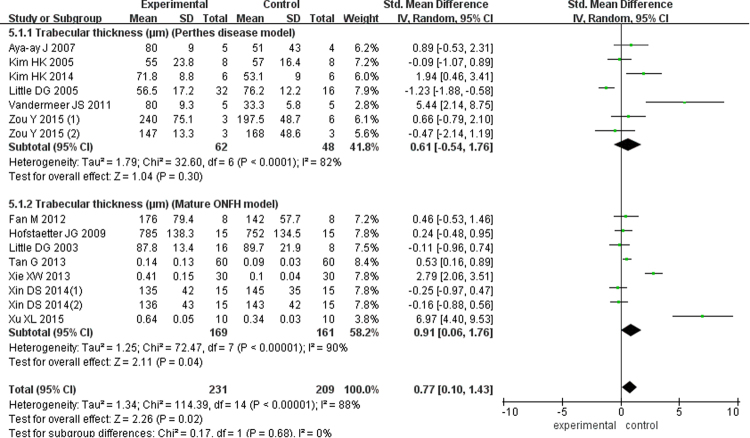
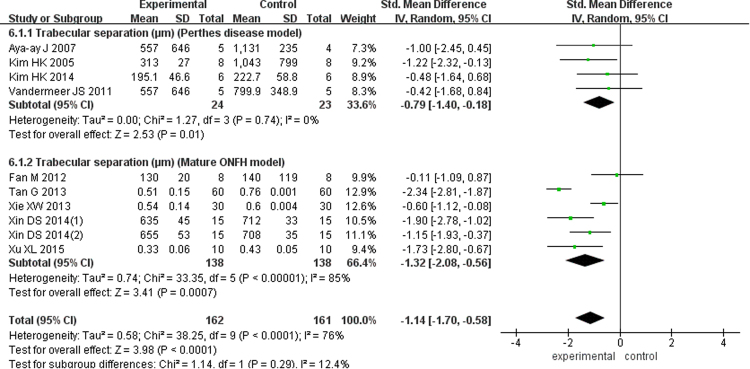
There were five studies with 95 Perthes disease models (SMD = 1.46; 95% CI, 0.51–2.40) and five studies of 264 mature ONFH models (SMD = 1.20; 95% CI, 0.59–1.80) showed the TN in this meta-analysis, and according to the pooled outcomes, the experimental group was superior to the control group (SMD = 1.30; 95% CI, 0.80–1.79; Fig. 4). Six studies of 110 Perthes disease models (SMD = 0.61; 95% CI, −0.54–1.76) and another eight studies of 330 mature ONFH models (SMD = 0.91; 95% CI, 0.06–1.76) analyzed the trabecular thickness and reported significantly better results in the bisphosphonate group (SMD = 0.77; 95% CI, 0.10–1.43; Fig. 5). At the same time, four articles examining 47 Perthes disease models (SMD = −0.79; 95% CI, −1.40 to −0.18) and six studies of 276 mature ONFH models (SMD = −1.32; 95% CI, −2.08 to −0.56) showed that the bisphosphonates improved the trabecular separation (SMD = −1.14; 95% CI, −1.70 to −0.58; Fig. 6).
Figure 4.
Forest plot showing trabecluar number.
Figure 5.
Forest plot showing trabecular thickness.
Figure 6.
Forest plot showing trabecular separation.
Outcomes of clinical trials
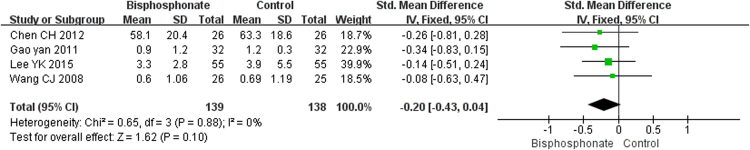
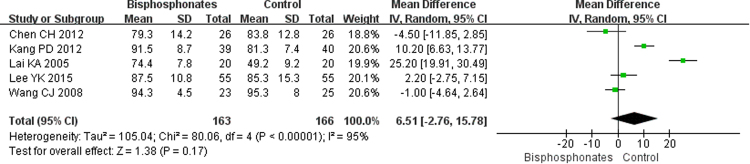
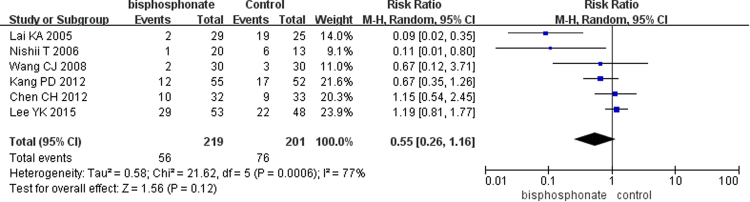
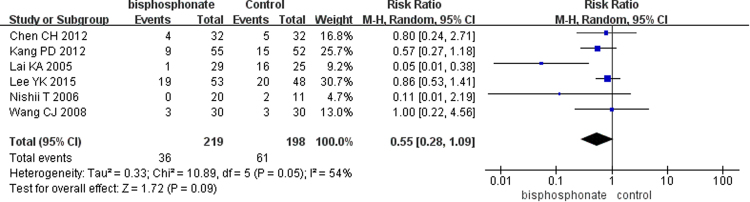
The pooled results of pain score with four studies of 277 patients and the hip Harris scores from five studies of 329 patients showed that bisphosphonate use achieved better pain scores (SMD = −0.20; 95% CI, −0.43–0.04; Fig. 7) and higher Harris scores (MD = 6.51; 95% CI, −2.76–5.78; Fig. 8); however, the differences were not statistically significant (p = 0.10 and p = 0.17, respectively). At the same time, the overall estimated proportion of patients who experienced progression to collapse in six studies involving 420 cases seemed to be reduced by bisphosphonate therapy (RR = 0.55; 95% CI, 0.26–1.16; Fig. 9), but it no significant difference was noted (p = 0.12). Likewise, the THA incidence tended to improve in patients treated with bisphosphonates (RR = 0.55; 95% CI, 0.28–1.09; Fig. 10), but the difference was not statistically significant (p = 0.09).
Figure 7.
Forest plot showing pain score.
Figure 8.
Forest plot showing Harris score.
Figure 9.
Forest plot showing collapse of the femoral head.
Figure 10.
Forest plot showing patients undergoing total hip arthroplasty.
Discussion
This meta-analysis aimed to determine whether bisphosphonates exerted effects on preventing femoral head collapse after osteonecrosis in animal models or clinical trials. Our results showed that bisphosphonate use significantly improved EQ indicative of femoral head sphericity as well as better BV, TN, trabecular separation, and trabecular thickness in the animal model. Unexpectedly, this finding is not supported by clinical studies, which showed no statistically significant differences in pain improvement, complications, or the need for THR. The animal studies and clinical trials seemed to present discordant outcomes.
There is a balance between osteoblast and osteoclast activity in the repair of ONFH 841,42. Osteoblasts are responsible for new bone formation, while osteoclasts are the bone resorptive cells. Although removal of the dead bone is also beneficial to the body, an excessively increased resorption rate leads to femoral head deformity and collapse. Some researchers have found that the osteoclasts become more active and develop a longer lifespan in the presence of osteonecrosis43,44. This may be the cause of the imbalance between osteogenesis and bone resorption. Bisphosphonates suppress the HMG-CoA reductase pathway and then inhibit osteoclast-mediated bone resorption, both of which accelerate osteoclast death32.
Although many animal studies16,17,27 and clinical trials14,15 have proven the efficiency of bisphosphonates in the treatment of ONFH, other researchers maintain different opinions. In clinical studies, Lee YK, et al. used zoledronate to treat patients with Steinberg stage I or II ONFH with a medium to large necrotic area, but their outcomes show that zoledronate does not prevent collapse of the femoral head or reduce the need for total hip arthroplasty39. Chen CH, et al. conducted a multicenter, prospective, randomized, double-blind, placebo-controlled study using alendronate to prevent femoral head collapse but concluded that alendronate had no obvious effects on decreasing the need for THA and cannot reduce disease progression or improve quality of life21. Moreover, the animal studies of Aruwajoye OO, et al.19 and Zou Y, et al.18 showed that the use of ibandronate alone did not obviously improve osteonecrosis, while the combination of ibandronate and other drugs such as BMP-2 or simvastatin could exert better protective effects. Likewise, Fan, et al.30 demonstrated that zoledronate could inhibit the formation of new vasculature, which may not benefit the repair process of ONFH. Accordingly, the current data was in controversy on the effectiveness of bisphosphonates.
Based on the outcomes of this meta-analysis, we found the EQ that stands for the height at center of the femoral head over the width, which used to evaluate the femoral head sphericity, was improved in the bisphosphonate group. This means that the using of bisphosphonates indeed exert effects on protecting the femoral head morphology. At the same time, the BV, TN, trabecular thickness, and trabecular separation factors used to assess bone mass of the femoral head in the animal model were all significantly improved by bisphosphonate use, a finding that was very encouraging. These results indicated that controlling the pathological activity of the osteoclasts helped repair the ONFH30. At the same time, some other studies17,45 reported that inhibiting osteoclast activity would help increase osteoblast action and, in turn, lead to a positive balance of bone formation. Resorption of dead compact bone during osteonecrosis repair may decrease the structural properties and mechanical support of the femoral head and may be partially responsible for the collapse in the late stages of osteonecrosis17,45. Thus, intervening in this process with bisphosphonates would make some difference just as our results showed. However, we also realized that those encouraging outcomes were drawn from the heterogeneous methods/models. Animal models of ONFH or Perthes disease are still too heterogeneous in terms of animal type, age, sex, interventionist strategy, and duration. The methods using bisphosphonates to treat ONFH also varied, including different bisphosphonate types, administration approaches, dosage, and durations, which may have influenced the outcomes. In summary, we observed improvement of the structural changes in terms of bone architecture and BV, but whether it could treat the ONFH in the animal model remains difficult to judge based on our outcomes alone.
Translating the animal results into clinical outcomes is always not easy, as discordant findings are usually present46,47. Although heterogeneous animal models suggest the improvement of some bone morphology in this meta-analysis, this is not supported by pooled clinical studies, which showed no statistical differences in pain improvement, complications, or the need for THR. In fact, the clinical studies also showed high heterogeneity, including different bisphosphonate drugs, administration approaches, and combination with many other procedures, all of which may contribute to different outcomes present in those studies and also influence the pooled outcomes in this meta-analysis. Selection bias in terms of patient case mix, sample bias, publication bias, or unintended bias due to the interpretation of post-randomization events could also have changed the outcomes46.
However, the obviously discordant findings in animal and human studies were still associated with some reasons. Animals and humans have different sensitivities to bisphosphonates. Animals were usually given adequate treatment doses throughout life, whereas humans were usually given low doses. It is easier to analyze the femoral head in animal models, both with radiographic films and specimen experiments. We only needed to analyze the desired outcomes while ignoring many other adverse events in animals. When we evaluated the patients, it was not until the femoral head had collapsed that we could obtain samples after the THA surgery, which means that the analytical methods were completely different and an effective evaluation method is lacking for human subjects. Besides, medication use in humans considers more consideration of safety, complications, and the overall body condition, all factors of which made the process differ from that of the animal experiment. More importantly, a poor methodology, publication bias, and inadequate animal models that simply do not reflect human disease are to blame for the discordant findings. Last but not least, it is difficult to know whether animal studies face similar problems regarding trial design. Clinical trials are usually powered to predict the number of patients likely to generate a statistical difference for the primary population. Additionally, to eliminate bias, patients are randomized and treatments are administered blindly by the researcher. In contrast, animal experiments are unlikely to be powered (usually concentrating on a static number per group according to laboratory preference) and rarely report crucial experimental design factors such as randomization and blinding46. To this end, the lack of bias elimination in preclinical studies is thought to contribute to a five-fold likelihood of showing a beneficial therapeutic effect48. Thus, using standardized animal models and standardized experiment methods may eliminate some of the differences between animal and human studies, enabling a greater degree of translation.
Conclusion
Bisphosphonates could improve bone architecture and BV in animal studies, but those results did not translate into either symptomatology or end-stage complications and management in the human studies. This might suggest that poor methodology, publication bias, and inadequate animal models that simply do not reflect human disease are to blame for the discordant findings. Thus, systematic reviews of animal studies are needed to ensure that the findings will be relevant to the design of clinical trials, while the use of standardized animal models and standardized experiment methods may eliminate some of the differences between animal and human studies to enable a greater degree of translation.
Acknowledgements
This study was supported by the National Natural Science Fund of China (81271976/H0605 and 81672165).
Author Contributions
Donghai Li contributed to search the database, extract the data, analyze the data and manuscript writing. Zhouyuan Yang helped to extract the data and review the manuscript. Zhun Wei searched the database and extracted the data. Pengde Kang is the correspondence author who responsible for the correspondence, topic identification and manuscript reviewing.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Hungerford DS, Jones LC. Asymptomatic osteonecrosis: should it be treated? Clin. Orthop. Relat. Res. 2004;429:124–130. doi: 10.1097/01.blo.0000150275.98701.4e. [DOI] [PubMed] [Google Scholar]
- 2.Mankin HJ. Nontraumatic necrosis of bone (osteonecrosis) N. Engl. J. Med. 1992;326:1473–1479. doi: 10.1056/NEJM199205283262206. [DOI] [PubMed] [Google Scholar]
- 3.Kealey WD, et al. Deprivation, urbanisation and Perthes’ disease in Northern Ireland. J. Bone. Joint. Surg. Br. 2000;82:167–171. doi: 10.1302/0301-620X.82B2.10033. [DOI] [PubMed] [Google Scholar]
- 4.Kim HK. Pathophysiology and new strategies for the treatment of legg-calve-perthes disease. J. Bone. Joint. Surg. 2012;94:659–669. doi: 10.2106/JBJS.J.01834. [DOI] [PubMed] [Google Scholar]
- 5.Stulberg SD, Cooperman DR, Wallensten R. The natural history of Legg-Calve-Perthes disease. J. Bone. Joint. Surg. Am. 1981;63:1095–1108. doi: 10.2106/00004623-198163070-00006. [DOI] [PubMed] [Google Scholar]
- 6.Kim SY, et al. Multiple drilling compared with core decompression for the treatment of osteonecrosis of the femoral head. J. Bone. Joint. Surg. Br. 2004;86:149. doi: 10.2106/00004623-200401000-00024. [DOI] [PubMed] [Google Scholar]
- 7.Chang CC, Greenspan A, Gershwin ME. Osteonecrosis: current perspectives on pathogenesis and treatment. Semin. Arthritis. Rheum. 1993;23:47–69. doi: 10.1016/S0049-0172(05)80026-5. [DOI] [PubMed] [Google Scholar]
- 8.Catterall A, et al. Perthes’ disease: is the epiphysial infarction complete? J. Bone. Joint. Surg. Br. 1982;64:276–281. doi: 10.1302/0301-620X.64B3.7096391. [DOI] [PubMed] [Google Scholar]
- 9.Allen MR, Burr DB. Bisphosphonate effects on bone turnover, microdamage, and mechanical properties: what we think we know and what we know that we don’t know. Bone. 2011;49:56–65. doi: 10.1016/j.bone.2010.10.159. [DOI] [PubMed] [Google Scholar]
- 10.Pazianas M, et al. Bisphosphonates and bone quality. BoneKey. Rep. 2014;3:1–8. doi: 10.1038/bonekey.2014.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Silverman S, Christiansen C. Individualizing osteoporosis therapy. Osteoporos. Int. 2012;23:797–809. doi: 10.1007/s00198-011-1775-y. [DOI] [PubMed] [Google Scholar]
- 12.Reid IR, et al. Biochemical and radiologic improvement in Paget’s disease of bone treated with alendronate: a randomized, placebo-controlled trial. Am. J. Med. 1996;101:341–348. doi: 10.1016/S0002-9343(96)00227-6. [DOI] [PubMed] [Google Scholar]
- 13.Lane JM, et al. Bisphosphonate therapy in fibrous dysplasia. Clin. Orthop. Relat. Res. 2001;382:6–12. doi: 10.1097/00003086-200101000-00003. [DOI] [PubMed] [Google Scholar]
- 14.Kang P, et al. Are the results of multiple drilling and alendronate for osteonecrosis of the femoral head better than those of multiple drilling? A pilot study. Joint. Bone. Spine. 2012;79:67–72. doi: 10.1016/j.jbspin.2011.02.020. [DOI] [PubMed] [Google Scholar]
- 15.Lai KA, et al. The use of alendronate to prevent early collapse of the femoral head in patients with nontraumatic osteonecrosis. A randomized clinical study. J. Bone. Joint. Surg. Am. 2005;87:2155–9. doi: 10.2106/00004623-200502000-00016. [DOI] [PubMed] [Google Scholar]
- 16.Little DG, et al. Zoledronic acid treatment results in retention of femoral head structure after traumatic osteonecrosis in young Wistar rats. J. Bone. Miner. Res. 2003;18:2016–22. doi: 10.1359/jbmr.2003.18.11.2016. [DOI] [PubMed] [Google Scholar]
- 17.Hofstaetter JG, et al. The effects of alendronate in the treatment of experimental osteonecrosis of the hip in adult rabbits. Osteoarthritis. Cartilage. 2009;17:362–70. doi: 10.1016/j.joca.2008.07.013. [DOI] [PubMed] [Google Scholar]
- 18.Zou Y, et al. Synergistic local drug delivery in a piglet model of ischemic osteonecrosis: a preliminary study. J. Pediatr. Orthop. B. 2015;24:483–92. doi: 10.1097/BPB.0000000000000213. [DOI] [PubMed] [Google Scholar]
- 19.Aruwajoye OO, Aswath PB, Kim HK. Material properties of bone in the femoral head treated with ibandronate and BMP-2 following ischemic osteonecrosis. J. Orthop. Res. 2017;35:1453–1460. doi: 10.1002/jor.23402. [DOI] [PubMed] [Google Scholar]
- 20.Wang CJ, et al. Treatment of osteonecrosis of the hip: comparison of extracorporeal shockwave with shockwave and alendronate. Arch. Orthop. Trauma. Surg. 2008;128:901–908. doi: 10.1007/s00402-007-0530-5. [DOI] [PubMed] [Google Scholar]
- 21.Chen CH, et al. Alendronate in the prevention of collapse of the femoral head in nontraumatic osteonecrosis: a two-year multicenter, prospective, randomized, double-blind, placebo-controlled study. Arthritis. Rheum. 2012;64:1572–1578. doi: 10.1002/art.33498. [DOI] [PubMed] [Google Scholar]
- 22.Yuan HF, Guo CA, Yan ZQ. The use of bisphosphonate in the treatment of osteonecrosis of the femoral head: a meta-analysis of randomized control trials. Osteoporos. Int. 2016;27:295–299. doi: 10.1007/s00198-015-3317-5. [DOI] [PubMed] [Google Scholar]
- 23.Moher D, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Plos. Med. 2009;6:e1000097. doi: 10.1371/journal.pmed.1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fisher M, et al. Update of the stroke therapy academic industry roundtable preclinical recommendations. Stroke. 2009;40:2244–2250. doi: 10.1161/STROKEAHA.108.541128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Oremus M, et al. Interrater reliability of the modified Jadad quality scale for systematic reviews of Alzheimer’s disease drug trials. Dement. Geriatr. Cogn. Disord. 2001;12:232–236. doi: 10.1159/000051263. [DOI] [PubMed] [Google Scholar]
- 26.Kim HK, et al. Ibandronate for prevention of femoral head deformity after ischemic necrosis of the capital femoral epiphysis in immature pigs. J. Bone. Joint. Surg. Am. 2005;87:550–557. doi: 10.2106/00004623-200503000-00011. [DOI] [PubMed] [Google Scholar]
- 27.Aya-ay J, et al. Retention, distribution, and effects of intraosseously administered ibandronate in the infarcted femoral head. J. Bone. Miner. Res. 2007;22:93–100. doi: 10.1359/jbmr.060817. [DOI] [PubMed] [Google Scholar]
- 28.Vandermeer JS, et al. Local administration of ibandronate and bone morphogenetic protein-2 after ischemic osteonecrosis of the immature femoral head: a combined therapy that stimulates bone formation and decreases femoral head deformity. J. Bone. Joint. Surg. Am. 2011;93:905–913. doi: 10.2106/JBJS.J.00716. [DOI] [PubMed] [Google Scholar]
- 29.Kim HK, et al. Local administration of bone morphogenetic protein-2 and bisphosphonate during non-weight-bearing treatment of ischemic osteonecrosis of the femoral head: an experimental investigation in immature pigs. J. Bone. Joint. Surg. Am. 2014;96:1515–1524. doi: 10.2106/JBJS.M.01361. [DOI] [PubMed] [Google Scholar]
- 30.Cheng TL, et al. Local delivery of recombinant human bone morphogenetic proteins and bisphosphonate via sucrose acetate isobutyrate can prevent femoral head collapse in Legg-Calve-Perthes disease: a pilot study in pigs. Int. Orthop. 2014;38:1527–1533. doi: 10.1007/s00264-013-2255-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Little DG, et al. Zoledronic acid improves femoral head sphericity in a rat model of perthes disease. J. Orthop. Res. 2005;23:862–868. doi: 10.1016/j.orthres.2004.11.015. [DOI] [PubMed] [Google Scholar]
- 32.Fan M, et al. Effect and mechanism of zoledronate on prevention of collapse in osteonecrosis of the femoral head. Zhongguo. Yi. Xue. Ke. Xue. Yuan. Xue. Bao. 2012;34:330–336. doi: 10.3881/j.issn.1000-503X.2012.04.004. [DOI] [PubMed] [Google Scholar]
- 33.Xu XL, et al. Prevention of femoral head collapse due to osteonecrosis in rats by local delivery of zoledronate via poly lactic-co-glycolic acid. Orthopedic. Journal. Of. China. 2015;23:843–849. [Google Scholar]
- 34.Xie XW, et al. Effects of alendronate and lovastatin in preventing early glucocorticoids-induced osteonecrosis of femoral head in rats by micro-CT. Orthopedic. Journal. of.China. 2013;21:82–86. [Google Scholar]
- 35.Tan G, et al. Micro-CT evaluation of alendronate in preventing glucocorticoids-induced osteonecrosis of femoral heads of rats. Chin. J. Joint. Surg. (Electronic Edition). 2013;7:534–538. [Google Scholar]
- 36.Xin DS, et al. Constructing a rat model of traumatic osteonecrosis of the femoral head with articular surface collapse and prevention mechanism of alendronate. Chinese. Journal. of.Tissue. Engineering. Research. 2014;18:5781–5787. [Google Scholar]
- 37.Xin DS, et al. Effects of strontium ranelate on the prevention of collapse in traumatic osteonecrosis of the femoral head in rats using micro-computed tomography. Orthopedic. Journal. of. China. 2014;15:1423–1429. [Google Scholar]
- 38.Nishii T, et al. Does alendronate prevent collapse in osteonecrosis of the femoral head? Clin. Orthop. Relat. Res. 2006;443:273–279. doi: 10.1097/01.blo.0000194078.32776.31. [DOI] [PubMed] [Google Scholar]
- 39.Lee YK, et al. Does Zoledronate Prevent Femoral Head Collapse from Osteonecrosis? A Prospective, Randomized, Open-Label, Multicenter Study. J. Bone. Joint. Surg. Am. 2015;97:1142–1148. doi: 10.2106/JBJS.N.01157. [DOI] [PubMed] [Google Scholar]
- 40.Gao Y. Clinical Observation of Early Steroid Secondary Osteonecrosis of Alendronate Therapy. Guide. Of. China. Medicine. 2016;21:20–21. [Google Scholar]
- 41.Terayama H, et al. Prevention of osteoneerosis by intravenous administration of human peripheral blood-derived CD34-positive cells in a rat osteonecrosis model. J. Tissue. Eng. Regen. Med. 2011;5:32–40. doi: 10.1002/term.285. [DOI] [PubMed] [Google Scholar]
- 42.Li W, et al. Distribution of TRAP-post-tire cells and expression of HIF-l alpha, VEGF, and FGF-2 in the reparative reaction in patients with osteonecrosis of the femoral head. J. Orthop. Res. 2009;27:694–700. doi: 10.1002/jor.20802. [DOI] [PubMed] [Google Scholar]
- 43.Weinstein RS, et al. Endogenous glucocorticoids decrease skeletal angiogenesis,vascularity,hydration,and strength in aged mice. Aging. Cell. 2010;9:147–161. doi: 10.1111/j.1474-9726.2009.00545.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Nagashima M, et al. Bisphosphonate (YM529) delays the repair of cortical bone defect after drill-hole injury by reducing terminal differentiation of osteoblasts in the mouse femur. Bone. 2005;36:502–511. doi: 10.1016/j.bone.2004.11.013. [DOI] [PubMed] [Google Scholar]
- 45.Hofstaetter JG, et al. Changes in bone microarchitecture and bone mineral density following experimental osteonecrosis of the hip in rabbits. Cells. Tissues. Organs. 2006;184:138–147. doi: 10.1159/000099620. [DOI] [PubMed] [Google Scholar]
- 46.Dyson A, Singer M. Animal models of sepsis: why does preclinical efficacy fail to translate to the clinical setting? Crit. Care. Med. 2009;37:S30–37. doi: 10.1097/CCM.0b013e3181922bd3. [DOI] [PubMed] [Google Scholar]
- 47.Krug N, Rabe KF. Animal models for human asthma: the perspective of a clinician. Curr. Drug. Targets. 2008;9:438–442. doi: 10.2174/138945008784533598. [DOI] [PubMed] [Google Scholar]
- 48.Perel P, Roberts I, Sena E, et al. Comparison of treatment effects between animal experiments and clinical trials: Systematic review. B.M.J. 2007;334:197. doi: 10.1136/bmj.39048.407928.BE. [DOI] [PMC free article] [PubMed] [Google Scholar]