Fig. 2.
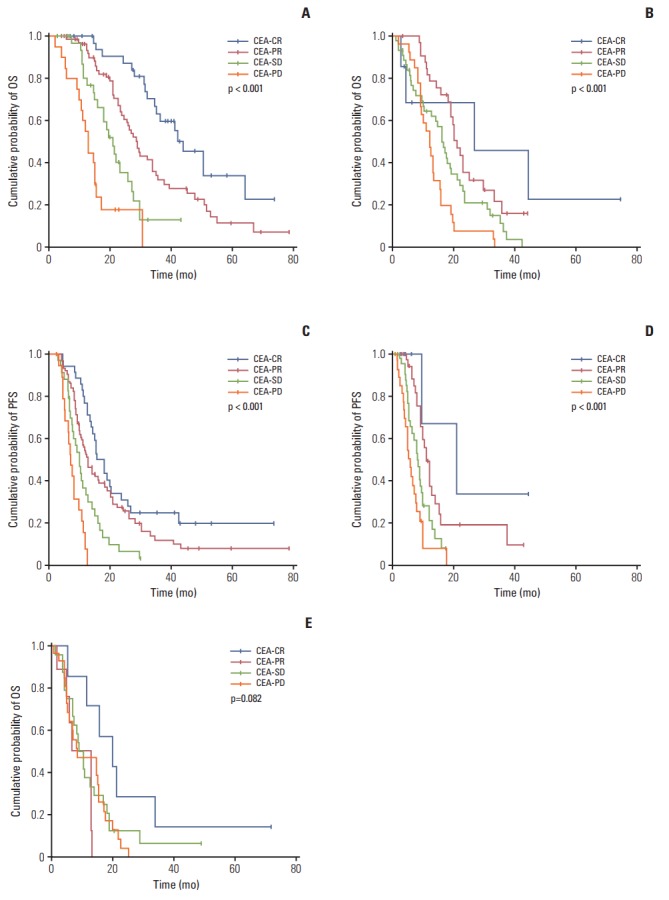
Cumulative survival rates according to CEA-response in patients with different RECIST evaluations. In patients with RECIST-PR (A and C), a discordant CEA-response (CEA-PD/SD) showed poorer survival than CEA-CR/PR (median OS and PFS: 44.0±5.9 and 15.4±1.9 months [CEA-CR], 28.9±1.8 and 12.5±1.3 [CEA-PR], 21.0±2.1 and 9.8±1.0 [CEA-SD], and 13.0±1.1 and 7.0±0.8 [CEA-PD], respectively; all p < 0.001). In patients with RECIST-SD (B and D), a more favorable CEA-response demonstrated better OS and PFS (median OS and PFS: 26.8±19.6 and 21.0±9.3 months [CEA-CR], 21.0±1.4 and 11.0±0.8 [CEA-PR], 16.1±1.4 and 8.2±0.8 [CEA-SD], and 12.2±1.1 and 6.0±0.7 [CEA-PD], respectively; all p < 0.001). In patients with RECIST-PD (E), there was no significant difference in OS according to CEA-response (median OS: 20.1±5.8 months [CEA-CR], 13.0±4.8 [CEA-PR], 9.0±1.4 [CEA-SD], and 8.7±4.6 [CEA-PD]; p=0.082). (A) OS in RECIST-PR patients, (B) OS in RECIST-SD patients, (C) PFS in RECIST-PR patients, (D) PFS in RECIST-SD patients, (E) OS in RECST-PD patients. CEA, carcinoembryonic antigen; CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease; RECIST, Response Evaluation Criteria in Solid Tumors; OS, overall survival; PFS, progression-free survival.
