Abstract
Polychlorinated biphenyls (PCBs), a group of 209 congeners that differ in the number and position of chlorines on the biphenyl ring, are anthropogenic chemicals that belong to the persistent organic pollutants (POPs). For many years, PCBs have been a topic of interest because of their bioaccumulation in the food-chain and their environmental persistence. PCBs with fewer chlorine atoms however are less persistent and more susceptible to metabolic attack, giving rise to chemicals characterized by the addition of one or more hydroxyl groups to the chlorinated biphenyl skeleton, collectively known as hydroxylated PCBs (OH-PCBs). In animals and plants this biotransformation of PCBs to OH-PCBs is primarily carried out by cytochrome P-450-dependent monooxygenases. One of the reasons for infrequent detection of lower chlorinated PCBs in serum and other biological matrices is their shorter half-lives, and their metabolic transformation, resulting in OH-PCBs or their conjugates, such as sulfates and glucuronides, or macromolecule adducts. Recent biomonitoring studies have reported the presence of OH-PCBs in human serum. The occurrence of OH-PCBs, the size of this group (there are 837 mono-hydroxyl PCBs alone), their wide spectra of physical characteristics (pKa’s and log P’s ranging over 5 to 6 orders of magnitude), give rise to a multiplicity of biological effects. Among those are: bioactivation to electrophilic metabolites that can form covalent adducts with DNA and other macromolecules, interference with hormonal signaling, inhibition of enzymes that regulate cellular concentrations of active hormones, and interference with the transport of hormones. New information creates an urgent need for this perspective.
Keywords: PCBs, Hydroxylated PCBs, Phenolic PCBs, Metabolites, Endocrine Disruption, Persistent Organic Pollutants, Biotransformation
INTRODUCTION
Polychlorinated biphenyls (PCBs) are synthetic organic compounds with the empirical formula C12H10-xClx (x=1–10). There are 209 congeners of PCBs, numbered PCB 1 to PCB 209 based on the number and position of chlorine atoms around the biphenyl rings (Ballschmiter & Zell 1980). Commercial production of PCBs began in the early 1920s, and PCBs were marketed in the US under the trade name Aroclor and with various trade names in other countries (Silberhorn et al. 1990). When added to a material, PCBs impart plastic and fire retardant properties. They are also very good coolants and lubricating agents. For these properties, PCBs were valuable chemicals in the industrial development of the twentieth century. Examples of PCB-containing products are transformers, capacitors, microwave ovens, air conditioners, fluid-cooled motors and electromagnets, electrical light ballasts, hydraulic and heat transfer fluids, switches, voltage regulators, circuit breakers, vacuum pumps, electric cables, inks, lubricants, waxes, flame retardants, adhesives, electrical and thermal insulating materials, pesticides, dyes, paints, asphalts, caulks, sealants and many more (AIHA 2013, ATSDR 2000, 2011, IARC 2013). The commercial production of PCBs was banned in many countries since the late 1970s. However, PCB legacy materials and new materials containing PCBs from pigments continue to be a source of environmental release even today (Hu & Hornbuckle 2010, Hu et al. 2010, Shanahan et al. 2015).
PCBs as a group are persistent organic pollutants (POPs) because they are not readily degraded by the physical and biologic processes in the environment. PCBs are also more soluble in oils, less in water. As a result, they tend to associate with organic materials and to bioaccumulate in fatty tissues of animals in the food web (Janssen et al. 2011). Very high levels of PCBs have been reported in the Inuit people who eat predominately fish and sea mammals (Laird et al. 2013). Average body burden of PCBs in adults living in a community with a history of PCB contamination is as high as 528 ng/g of serum lipid (Pavuk et al. 2014). Although a fish and meat-based diet has been recognized as the primary source of exposure to PCBs, an estimated 6–64% of body burden of PCBs occurs through inhalation exposures (Currado & Harrad 1998). PCBs with four or fewer chlorine atoms are found in many indoor and outdoor environments as a result of temperature-dependent slow evaporation and transport from PCB containing materials (Hu & Hornbuckle 2010, Hu et al. 2010). Other atmospheric sources of PCBs are landfills, electronic waste recycling, and open incineration of plastic wastes (Tue et al. 2013). Generation of atmospheric PCB 11, which was not found in Aroclor mixtures, was a strong indication that newly manufactured materials, like dyes, pigments and paints can also contribute PCBs (Hu et al. 2008). Cities like Chicago and Cleveland (Persoon et al. 2010), and contaminated sites like New Bedford Harbor (Martinez et al. 2017) are major sources of airborne PCBs. PCBs are transported with facility among strata (sediment, water and air). Fluxes of PCBs from sediment to the overlying water (4 kg per year), and from water to air (7 kg per year) have been described for an industrial harbor in East Chicago (Martinez et al 2010b). This site alone contributes about 44 kg per year to Lake Michigan. Airborne PCBs are in dynamic equilibrium and depending on the atmospheric conditions, the flux of PCBs may be from Chicago to Lake Michigan, or the reverse (Hu et al. 2010, Martinez et al. 2010a, Martinez et al. 2010b).
Because PCBs are considered non-degradable chemicals, human biomonitoring for PCB exposure is targeted primarily on the identification of parent PCBs. NHANES surveys still use serum measurement of 42 congeners (PCB 28 and higher chlorinated ones) as an indicator of PCB body burden (CDC 2013). However, a recent human biomonitoring study of mothers and children in Iowa reports the detection of many PCBs including ones that are less chlorinated than PCB 28 (2,4,4’-trichlorobiphenyl) (Koh et al. 2015). In addition, a plethora of metabolism studies now indicate that PCBs in fact are bio-transformed into a variety of metabolites by many life forms including animals, plants and microorganisms.
Since there is no longer intentional commercial production of PCBs, and there is a continuous, but slow biotransformation of many parent PCBs, it is likely that the global stock of PCBs may gradually shift into metabolic forms, although it will be a very slow process. Evidence of PCB metabolism suggests that OH-PCBs are environmentally stable and may be more toxic by some measures.
Physical Properties of OH-PCBs
OH-PCBs are characterized by the addition of one or more hydroxyl groups to the chlorinated biphenyl skeleton. Although 837 mono-hydroxyl PCBs are possible, a much smaller number have been synthesized, characterized and determined in environmental samples. The acid-base dissociation constant (pKa) of all possible mono-OH-PCBs have been calculated with in silico methods and found to range from about 4 to 10 (Grimm et al. 2015b). On this basis, only OH-PCBs pentachloro- and higher would exist as anions in biological systems (Figure 1). Surprisingly, substitution with a hydroxyl group does not greatly alter the lipophilicity (log P) of the PCB (Table 1). OH-PCBs have mean Log P values ranging from 4 to 8, very similar to those of the parent PCBs (Table 1).
Figure 1. Change in pKa and Log P vs. Chlorine number of hydroxylated polychlorinated biphenyls (PCBs).
Mean pKa and mean Log P values of mono-hydroxyl PCBs were calculated from data provided for each group (please see reference Grimm et al 2015b)
Table 1. Number of isomers, pKa and Log P values of mono-hydroxyl PCBs.
Log P values of the parent PCBs are presented for comparison. The original sources of pKa and Log P are compiled from (Grimm et al. 2015b). The Log P values of parent PCBs are compiled from (Brodsky & Ballschmiter 1988)
| Phenolic Metabolites of | Number of Isomers |
pKa OH-PCBs (Mean)1 |
Log P OH-PCBs (Mean)1 |
Log P of parent PCBs (Mean)2 |
|---|---|---|---|---|
| Monochlorobiphenyls | 19 | 9.47 | 4.03 | 4.51 |
| Dichlorobiphenyls | 64 | 8.97 | 4.77 | 5.02 |
| Trichlorobiphenyls | 136 | 8.52 | 5.19 | 5.45 |
| Tetrachlorobiphenyls | 198 | 8.04 | 5.89 | 5.81 |
| Pentachlorobiphenyls | 198 | 7.55 | 6.55 | 6.28 |
| Hexachlorobiphenyls | 136 | 7.05 | 7.14 | 6.64 |
| Heptachlorobiphenyls | 64 | 6.58 | 7.63 | 6.99 |
| Octachlorobiphenyls | 19 | 6.06 | 8.09 | 7.36 |
| Nonachlorobiphenyls | 3 | 5.39 | 8.23 | 7.83 |
| Decachlorobiphenyl | 0 | - | - | 8.38 |
| Total | 837 |
Data compiled from (Grimm et al. 2015b)
Data compiled from (Brodsky & Ballschmiter 1988)
Chirality in the OH-PCBs
Nineteen PCB congeners with three or four ortho chlorines form atropisomers stable to racemization under physiological conditions. This was demonstrated by isolation of PCB enantiomers (atropisomers) through semi-preparative enrichment, by liquid chromatography and synthesis of diasteriomeric intermediates (Haglund 1996a, b, Püttmann et al. 1989, Püttmann et al. 1986). All nineteen chiral PCBs are present in technical PCB mixtures (Kania-Korwel & Lehmler 2016b), and large quantities of these congeners have been produced across the world (Kania-Korwel & Lehmler 2016b). As a result of their release into the environment, many chiral PCBs are present in wildlife and humans [for in-depth reviews of chiral PCBs see (Kania-Korwel & Lehmler 2016b, a, Lehmler & Robertson 2001, Lehmler et al. 2010)]. Chiral PCBs undergo enantiomeric enrichment in wildlife and humans due to enantioselective biotransformation and other biological processes, a fact that has been used to study the environmental fate and transport of PCBs (Lehmler et al. 2010). Moreover, exposure to chiral PCBs enantioselectively alters the expression of drug metabolizing enzymes in the liver (Püttmann et al. 1990, Püttmann et al. 1989, Rodman et al. 1991) and modulates endpoints in the developmental neurotoxicity of PCBs (Lehmler et al. 2005, Pessah et al. 2009, Yang et al. 2014). Although this has not been shown experimentally, it is likely that the atropisomers of OH-PCBs also cause toxicity in an atropselective manner. Assuming that an OH substituent offers a barrier to rotation approximately equal to that of a chlorine atom in the ortho position, there would be 110 stable mono-hydroxyl PCBs pairs (Table 2). Indeed several of these have been found to be formed (see the discussion below).
Table 2. All possible mono-OH-PCBs that are chiral.
An assumption is that OH hinders rotation/racemization of the PCB atropisomers. This table is based on a similar approach employed by (Püttmann et al. 1989, Püttmann et al. 1986) for PCB atropisomers. The shorthand for the structures is that used in Table 2 of Grimm et al, 2015b. The yellow highlighted panels are duplicates.
| Ring Substitution | −2,3,6 | −2,3,4,6 |
|---|---|---|
| −2 | 6'-0H-PCB 16, 6'-0H-PCB 18, 3'-0H-PCB 19, 2'-0H-PCB 24 | 6-0H-PCB 41, 4-0H-PCB 45, 6-0H-PCB 48, 3-0H-PCB 50, 2'-0H-PCB 62 |
| −2,3 | 6-0H-PCB 40, 4'-0H-PCB 44, 3'-0H-PCB 45, 3'-0H-PCB 46, 2'-0H-PCB 59 | 6-0H-PCB 82, 4-0H-PCB 84, 3'-0H-PCB 88, 6'-0H-PCB 97, 3'-0H-PCB 98, 2'-0H-PCB 109 |
| −2,4 | 6-0H-PCB 42, 4'-0H-PCB 45, 6'-0H-PCB 49, 3'-0H-PCB 51, 2'-0H-PCB 64 | 6-0H-PCB 85, 4'-0H-PCB 88, 4-0H-PCB 91, 6-0H-PCB 99, 3-0H-PCB 100, 2'-0H-PCB 115 |
| −2,5 | 6-0H-PCB 44, 5'-0H-PCB 45, 6-0H-PCB 52, 3'-0H-PCB 53, 6'-0H-PCB 59 | 6-0H-PCB 87, 5'-0H-PCB 88, 4-0H-PCB 95, 6-0H-PCB 101, 3-0H-PCB 103, 6'-0H-PCB 109 |
| −2,3,4 | 6'-0H-PCB 82,4'-0H-PCB 84, 6'-0H-PCB 87, 3'-0H-PCB 89, 3'-0H-PCB 91, 2'-0H-PCB 110 | 6-0H-PCB 128, 4'-0H-PCB 131, 4'-0H-PCB 132, 6'-0H-PCB 138, 3'-0H-PCB 139, 3'-0H-PCB 140, 2'-0H-PCB 158 |
| −2,3,5 | 6'-0H-PCB 83, 5'-0H-PCB 84, 6'-0H-PCB 92, 3'-0H-PCB 94, 3'-0H-PCB 95, 2'-0H-PCB 113 | 6-0H-PCB 130, 5'-0H-PCB 131, 4'-0H-PCB 135, 3'-0H-PCB 144, 6'-0H-PCB 146, 3'-0H-PCB 148, 2'-0H-PCB 161 |
| −2,3,6 | 6'-0H-PCB 84, 6'-0H-PCB 95, 3'-0H-PCB 96 | 6'-0H-PCB 131, 6-0H-PCB 132, 4-0H-PCB 136, 6'-0H-PCB 144, 3'-0H-PCB 145, 6'-0H-PCB 149, 3'-0H-PCB 150 |
| −2,4,5 | 5'-0H-PCB 91, 4'-0H-PCB 95, 6-0H-PCB 97, 6'-0H-PCB 101, 3'-0H-PCB 102, 6'-0H-PCB 110 | 6-0H-PCB 138, 5'-0H-PCB 139, 4'-0H-PCB 144, 4-0H-PCB 149, 6-0H-PCB 153, 3'-0H-PCB 154, 6'-0H-PCB 158 |
| −2,3,4,5 | 6'-0H-PCB 129, 5-0H-PCB 132, 4-0H-PCB 135, 6'-0H-PCB 141, 3'-0H-PCB 143, 3'-0H-PCB 149, 2'-0H-PCB 164 | 6'-0H-PCB 170, 5'-0H-PCB 171, 4'-0H-PCB 174, 4'-0H-PCB 175, 6'-0H-PCB 180, 3'-0H-PCB 182, 3'-0H-PCB 183, 2'-0H-PCB 191 |
| −2,3,4,6 | 6'-0H-PCB 131, 6-0H-PCB 132, 4-0H-PCB 136, 6'-0H-PCB 144, 3'-0H-PCB 145, 6'-0H-PCB 149, 3'-0H-PCB 150 | 6'-0H-PCB 171, 4'-0H-PCB 176, 6'-0H-PCB 183, 3'-0H-PCB 184 |
Oxidation of PCB congeners by cytochrome P-450 monooxygenases leads to production of OH-PCBs
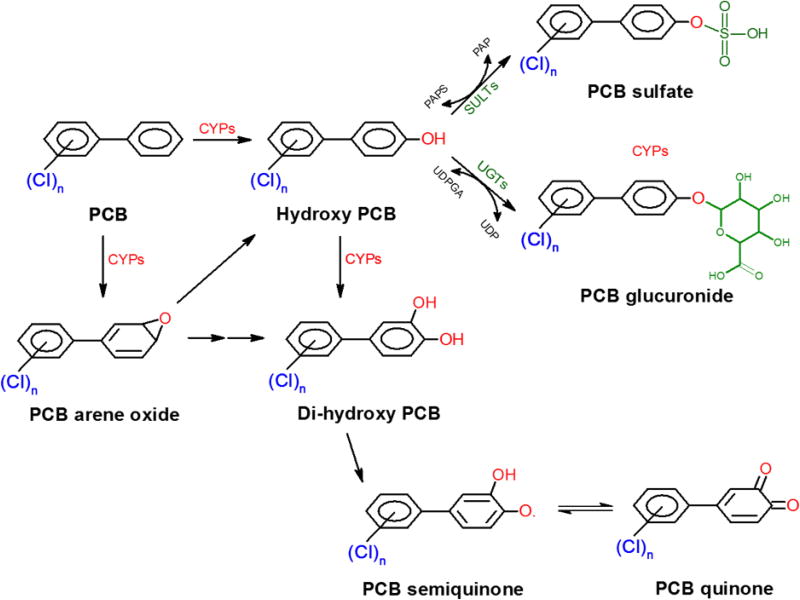
Cytochrome P-450 monooxygenases are ubiquitous enzymes that are expressed in plants and animals. In humans cytochrome P-450 families 1 and 2 contain major inducible isoforms involved in biotransformation of PCBs. Liver is the main organ for biotransformation reactions, although expression of cytochrome P-450 isoforms has been documented in other organs such as the intestine, brain, and lungs (Stamou et al. 2014, Thelen & Dressman 2009). To understand the mechanism of cytochrome P-450 enzymatic reactions with PCBs, early studies focused on the mono-chlorinated PCBs as substrates (Block & Cornish 1959, Wyndham & Safe 1978). These studies led to the discovery of two oxidative processes cytochrome P-450 enzymes exert. First, the direct process involves the insertion of a hydroxyl group into the PCB. Second, in a more indirect process, an arene oxide intermediate is formed which rearranges to form the OH-PCB (Figure 2). The intermediacy of an arene oxide may allow the movement of neighboring substituents, a process called an NIH (or the 1,2) shift (McLean et al. 1996). Through profiling of all urinary metabolites of PCB 3 (4-monochlorobiphenyl) after in vivo exposure in rats, Dhakal et al. showed that the formation of arene oxide intermediates is as important as direct insertion because several mercapturic acid metabolites were detected in urine as end products of PCB 3 arene oxide reaction with glutathione (Dhakal et al. 2012). The 4’OH-PCB 3 is the most favorable and major oxidation product of PCB 3 (McLean et al. 1996, Wyndham & Safe 1978). Hydroxylation at the 4’ para position is in fact the major OH-PCB congener detected in human serum (Koh et al. 2015, Marek et al. 2014, Marek et al. 2013). Incubation of PCB 3 with liver microsomes resulted in the formation of five mono- and three di-hydroxylated PCB 3 metabolites of PCB 3 (McLean et al. 1996). A similar mechanism of the formation of OH-PCBs can be anticipated for higher chlorinated PCBs, although factors like ortho-substitution in PCB 126 or isomerism in PCB 95 and PCB 136 may give rise to different OH-PCB forms. Biotransformation of PCBs to OH-PCBs through cytochrome P-450-catalzyed oxidation has also been documented in lower organisms (Kamata et al. 2015) and in plants (Ma et al. 2016, Zhai et al. 2010a, b, 2011b, 2013a).
Figure 2. Metabolism of hydroxylated PCBs.
The parent PCBs are oxidized in reactions catalyzed by cytochrome P-450 mono-oxygenases (CYPs) to form hydroxylated PCBs. The hydroxylated PCBs may then be conjugated to form PCB sulfates or PCB glucuronides, or metabolized into reactive semi-quinones or quinones.
Chirality in the cytochrome P-450 metabolism of PCBs
Chiral OH-PCBs can be formed by the cytochrome P-450 mediated metabolism of either chiral or prochiral PCBs, thus resulting in 110 possible chiral OH-PCB metabolites (Table 2). Studies examining chiral OH-PCB formation used several model systems including recombinant cytochrome P-450 enzymes (Lai et al. 2011, Lu et al. 2013, Waller et al. 1999a, Warner et al. 2009), liver microsomes (Kania-Korwel et al. 2011b, Kania-Korwel & Lehmler 2013, Schnellmann et al. 1983, Uwimana et al. 2016, Wu et al. 2014a, Wu et al. 2011b), primary rat liver hepatocytes (Vickers et al. 1986) and precision cut liver tissue slices (Wu et al. 2013a, Wu et al. 2013c). These studies demonstrate that chiral PCB congeners, in particular PCB congeners with a 2,3,6-trichloro substitution pattern in one phenyl ring, are metabolized in a congener-specific manner by mammalian cytochrome P-450 enzymes to chiral OH-PCB metabolites. In mammals, CYP2B isoforms, such as rat CYP2B1 (Lu et al. 2013, Waller et al. 1999a, Warner et al. 2009), human CYP2B6 (Warner et al. 2009) and dog CYP2B11 (Waller et al. 1999a), oxidize chiral PCBs with a 2,3,6-trichloro substitution pattern in meta position. Based on a recent study with PCB 52 (2,2’,5,5’-tetrachlorobiphenyl)(Shimada et al. 2016), human CYP2A6 is likely responsible for the formation of chiral para hydroxylated metabolites from chiral PCBs. In addition to the formation of chiral OH-PCBs from chiral parent PCBs, we recently demonstrated that axially prochiral PCB congeners can be metabolized to chiral OH-PCB metabolites (Uwimana et al. 2017). For example, oxidation of the 2,6-dichlorophenyl ring in PCB 51 by CYP2B enzymes in the meta position breaks the symmetry of this phenyl ring, thus resulting in a chiral OH-PCB metabolite. The oxidation of both prochiral and chiral PCBs to chiral OH-PCBs is species and congener dependent (Uwimana et al. 2017, Waller et al. 1999b, Wu et al. 2014b). This observation is not surprising given the diversity of expression of cytochrome P-450 enzymes across species. In vivo studies also reveal the formation of chiral OH-PCB metabolites in rats (Kania-Korwel et al. 2008c) and mice (Kania-Korwel et al. 2015, Kania-Korwel et al. 2012, Kania-Korwel et al. 2017, Wu et al. 2015) exposed orally or intraperitoneally to a racemic PCB congener. There is also evidence that plants, such as poplar plants, metabolize chiral PCBs to chiral OH-PCB metabolites (Ma et al. 2016, Zhai et al. 2011a).
Similar to the parent compounds, chiral OH-PCBs can be separated using both liquid (Pham-Tuan et al. 2005, Zhai et al. 2013b) and gas chromatographic (GC) techniques (Kania-Korwel et al. 2011a, Kania-Korwel et al. 2008c, Uwimana et al. 2017). To date, enantioselective LC analyses appear to be limited to a smaller number of chiral OH-PCBs. Enantioselective GC analyses using different commercial capillary GC columns containing modified β- or γ-cyclodextrins require the derivatization of the OH-PCBs to the corresponding methylated derivatives, but allow the separation of a considerable number of OH-PCB atropisomer derivatives. Enantioselective GC methods have been used to study the enantioselective metabolism of PCBs to chiral OH-PCBs in vitro and in vivo. The enantioselective metabolism of PCBs by mammalian cytochrome P-450 enzymes to OH-PCBs is both congener and species dependent (Kania-Korwel et al. 2008b, Uwimana et al. 2017, Wu et al. 2013a, Wu et al. 2014a, Wu et al. 2013c, Wu et al. 2011b). The enantiomeric enrichment of OH-PCBs is not only dependent on their atropselective formation by cytochrome P-450 enzymes, but is also modulated by their further oxidation to dihydroxylated and, most likely, other PCB metabolites. For example, racemic 2,2′,3,5′,6-pentachlorobiphenyl-4-ol and 2,2′,3,5′,6-pentachlorobiphenyl-5-ol, two metabolites of PCB 95, are enantioselectively metabolized by rat CYP2B1 to 3,4-dihydroxy-2,2′,5,5′,6-pentachlorobiphenyl, a chiral catechol metabolite of PCB 95 (Lu et al. 2013).
Only limited information about the absolute configuration of chiral PCB congeners and their metabolites is currently available (Pham-Tuan et al. 2005, Toda et al. 2012). The identification of OH-PCB atropisomers (as methylated derivatives) using GC methods is therefore primarily based on their elution order on the respective enantioselective GC column. Metabolism studies with pure atropisomers of PCB 136 allowed the identification of which OH-PCB atropisomer is formed from which PCB 136 atropisomer in incubations with rat liver microsomes (Figure 3) (Wu et al. 2011a). Based on an analysis of the Chirasil-Dex column, the first (E1) and second (E2) eluting atropisomers of 2,2’,3,3’,6,6’-hexachlorobiphenyl-5-ol (5-OH-136) are formed from (−)-and (+)-PCB 136, respectively. The atropisomers of the minor metabolite, 2,2’,3,3’,6,6’-hexachlorobiphenyl-4-ol (4-OH-136), eluting first and second on the Cyclosil-B column, are also formed from (−)- and (+)-PCB 136, respectively. E2-5-OH-136, formed from (+)-PCB 136, was the major metabolite formed in these rat liver incubations, which is consistent with a significant enrichment of (−)-PCB 136 in these experiments. Similarly, the more rapid oxidation of (+)-PCB 136 to E2-5-OH-136 is consistent with the enrichment of (−)-PCB 136 observed in incubations of racemic PCB 136 in precision-cut liver tissue slices from phenobarbital pre-treated male and female rats (Wu et al. 2013b). E2-5-OH-136 is also enriched in the liver of male and female rats exposed intraperitoneally to racemic PCB 136. In contrast, E1-5-HO-PCB 136 and E1-4-HO-PCB 136 are formed preferentially in incubations of racemic PCB 136 with liver microsomes or precision-cut liver tissue slices from mice (Wu et al. 2013a, Wu et al. 2014a). In agreement with this finding, mice exposed to racemic PCBs display an enrichment of (+)-PCB 136 in tissues (Kania-Korwel et al. 2010, Kania-Korwel et al. 2008a, Kania-Korwel et al. 2007, Kania-Korwel et al. 2008d, Milanowski et al. 2010a).
Figure 3.
The atropisomer of 5-OH PCB 136 eluting second on the Chirasil-Dex column (E2-5-OH-PCB 136) and the atropisomer of 4-OH PCB 136 eluting first on the Cyclosil-B column (E1-4-OH-PCB) are formed preferentially in incubations of racemic PCB 136 with liver microsomes from rats pretreated with different inducers of cytochrome P-450 enzymes. Based on incubations with pure PCB 136 atropisomers, the first and second eluting atropisomer of both 5-OH-PCB 136 and 4-OH-PCB 136 are formed from (−)-PCB 136 and (+)-PCB 136, respectively. Enantioselective gas chromatograms of (A) racemic 5-OH-PCB 136 and 4-OH-PCB 136 standards; (B) E1-5-OH-PCB 136 and E1-4-OH-PCB 136 formed from (−)-PCB 136; (C) E2-5-OH-PCB 136 and E2-4-OH-PCB 136 formed from (+)-PCB 136; and chiral signature of 5-OH-PCB 136 and 4-OH-PCB 136 formed by liver microsomes from rats treated with (D) PB, (E) DEX or (F) VEH. All OH-PCB metabolites were methylated with diazomethane prior to GC analysis. Reprinted with permission from (Wu et al. 2011a). Copyright 2011 American Chemical Society.
Conjugation of OH-PCBs and the biological implications
Biotransformation of xenobiotics often involves conversion of lipophilic compounds to more polar forms. OH-PCBs are Phase I biotransformation products of PCBs. In Phase II biotransformation, phenols are conjugated with functional groups that may result in more polar products that facilitate excretion. The most common conjugation reactions for phenols are glucuronidation and sulfation. A glucuronide metabolite is formed by transfer of a glucuronosyl group from uridine-5’-diphospho-α-D-glucuronic acid (UDPGA) to a phenol in an enzymatic reaction catalyzed by the microsomal UDP-glucuronosyltransferases (UGTs). Similarly, mammalian cytosolic sulfotransferases (SULTs) catalyze formation of sulfate metabolites by transfer of a sulfuryl group from 3’-phosphoadenosine-5’-phosphosulfate (PAPS). OH-PCBs are good substrates for SULT and UGT isoforms in vitro (Ekuase et al. 2011, Kester et al. 2000, Liu et al. 2006, Liu et al. 2009, Sacco & James 2005, Tampal et al. 2002). However, some congeners with the phenolic hydroxyl group in an ortho-position with respect to the biphenyl ring-junction have been found to hinder conjugation reactions (Ekuase et al. 2011, Tampal et al. 2002). Several phenolic PCBs have been reported to be selective inhibitors of sulfotransferases and UDP-glucuronosyltransferases, and thereby may reduce the sulfation or glucuronidation of other xenobiotics that is required for their elimination (Sacco et al. 2008, van den Hurk et al. 2002, Wang et al. 2006). The reported presence of OH-PCBs in human urine, blood and milk, and their potential inhibitory effects on the sulfotransferases and glucuronosyltransferases, has also been suggested to cause metabolic, endocrine, and developmental defects (James & Ambadapadi 2013, Kester et al. 2000)
Incubation of OH-PCBs with hepatic cytosolic fractions derived from polar bear (Ursus maritimus) resulted in formation of sulfate conjugates for 4’-hydroxy-3,3’,4,5’-tetrachlorobiphenyl (4’OH-PCB 79), 4’-hydroxy-2,3,3’,4,5,5’-hexachlorobiphenyl (4’-OH-PCB 159), 4’-hydroxy-2,3,3’,5,5’,6-hexachlorobiphenyl (4’-OH-PCB 165) (Sacco & James 2005). There are limited metabolism studies devoted to in vivo phase II biotransformations of OH-PCBs. A metabolism study carried out with rats has reported that a majority of PCB 3 metabolites excreted via the urine after inhalation exposure were sulfates (Dhakal et al. 2014). Following intraperitoneal injection of PCB 3 in the rat, some glucuronide metabolite was detected in the urine and bile, but it was minor as compared to either sulfation or glutathione conjugation (Dhakal et al. 2012). More recently, 4-PCB 11 sulfate was detected in the serum of humans who were known to have been exposed to environments containing PCB 11 and other airborne PCBs (Grimm et al. 2017). This provides evidence for both initial hydroxylation of the PCB and subsequent sulfation of the resulting OH-PCB in humans. Interestingly, plants, including poplar trees, can also form sulfated metabolites after exposure to PCBs through their roots (Zhai et al. 2010a). These findings suggest that conjugated metabolites of PCBs are formed in a broad range of species, although the stability and fate of conjugated metabolites of OH-PCBs in the environment have not yet been fully explored.
Although phenolic conjugates provide evidence for the intermediacy of the phenols, it is also clear that sulfatases and β-glucuronidases are capable of catalyzing hydrolysis of sulfate and glucuronide conjugates and would thus provide the basis for a dynamic interchange between OH-PCBs and their conjugates (Parkinson et al. 2012). For example, bisphenol A is a substrate for the estrogen sulfotransferase and the product sulfate ester is a substrate for steroid sulfatase (Stowell et al. 2006). Interestingly, when 4-PCB 11 sulfate was administered to rats by intravenous injection, OH-PCB 11 was detected in urine, suggesting that conversion of this conjugated metabolite of PCB 11 to free phenol was occurring in vivo (Grimm et al. 2015a). Following urinary excretion, however, PCB sulfates may be quite stable. PCB 11 sulfate was stable in rat urine for 24 h at 25 °C (Grimm et al. 2013). Some PCB sulfates were found to be stable in human urine for several days at 25 °C and for more than six weeks at −20 °C (Dhakal et al. 2013). In summary, evidence from various in vivo and in vitro studies shows that both PCB congeners and their respective sulfate and glucuronide metabolites are converted into OH-PCBs.
OH-PCBs are bioactivated to electrophilic metabolites
Following exposure, PCBs are transferred into the blood circulation and distributed to all organs and tissues (Hu et al. 2014, Milanowski et al. 2010b, Pereg et al. 2001). In the liver and possibly other organs, PCBs are oxidized by cytochrome P-450 with the generation of reactive intermediates, PCB arene oxides. These arene oxides are short lived, very strong electrophiles which may potentially react with macromolecules that have nitrogen and sulfur nucleophilic centers (Amaro et al. 1996). Arene oxides may be detoxified by conjugation with glutathione through the action of glutathione transferases (Dhakal et al. 2012), or disproportionate to the OH-PCB. Alternatively, arene oxides may be detoxified by epoxide hydrolase to a dihydrodiol that can be further metabolized by dihydrodiol dehydrogenase (AKR1C) to the diOH-PCB (McLean et al. 1996). OH-PCBs undergo phase II metabolism by glucuronosyl transferases or sulfotransferases (see above). Alternatively, the OH-PCBs may undergo a second oxidation and the formation of a diOH-PCB which may oxidize to a semiquinone and further to a quinonoid species. PCB quinones (PCB-Q) are another form of bioactivated metabolites. Formation of PCB-Q is favored by the substitution of hydroxyl groups in ortho or para positions, and are carried out enzymatically by peroxidases (Amaro et al. 1996, Wangpradit et al. 2009). These different metabolites display diverse toxic effects with strong differences in potency depending on the parent PCB structure. In addition, different tissues with their diverse metabolizing enzyme activities may favor distinct metabolism pathways for PCBs. With the current level of knowledge it is therefore not possible to deduce which pathway is the most predominant leading to a toxic effect.
Toxicity, Genotoxicity and Carcinogenicity of OH-PCBs
PCBs are classified by IARC as Class 1, human carcinogens (Lauby-Secretan et al. 2015). An important question is whether OH-PCBs could play a role in this carcinogenicity, as initiators, promoters or co-carcinogens.
The first step in carcinogenesis is initiation, a genotoxic event. Very few studies exist that evaluated the initiating activity/genotoxicity, of OH-PCBs. Espandiari and coworkers observed that PCB 3, PCB 15, PCB 52 and PCB 77, but not PCB 12 and PCB38, were positive in a modified rat liver foci assay that determines initiating activity of compounds (Espandiari et al. 2003). Using three mono-OH, three di-OH, and two quinone metabolites of PCB 3, they further discovered that only the 4-OH-PCB 3 and the 3,4-quinone of PCB 3 also exhibited this activity (Espandiari et al. 2004). This suggested that PCB 3 and the 4-OH metabolite could be proximate carcinogens and the 3,4-quinone the ultimate carcinogen. The initiating activity of PCB 3 and 4-OH-PCB 3 was further analyzed using the transgenic BigBlue rat model. PCB 3 significantly increased the number of mutations of the lac I reporter gene (Lehmann et al. 2007). 4OH-PCB 3 caused a non-significant doubling of the mutant frequency. The PCB 3 induced mutations were mostly GC→ TA transversions and frameshift mutations, suggesting DNA adduction or oxidative DNA damage as underlying mechanisms (Lehmann et al. 2007). This suggests that an arene-oxide, the precursor of OH-PCBs, could be the genotoxic species, but does not exclude that OH-PCBs are a precursor for a second ultimate carcinogenic metabolite.
To gain insight into the genotoxic activity of the various metabolic products, PCB 3 and seven synthetic metabolites were tested in several genotoxicity assays. Using a V79 cell line which does not contain cytochrome P-450 enzymes, PCB 3 was negative in all assays. Only the quinones induced point mutations, only the 3,4-diOH PCB 3 increased sister chromatid exchanges (SCE), and only the 2,5-diOH PCB 3 produced polyploid cells (Zettner et al. 2007). 4-OH PCB 3 and both the diOH- and the quinonoid metabolites induced chromosome breaks, with an efficiency that increased from 4-OH < diOH- < quinones (Table 3), suggesting that the quinones may be the ultimate clastogenic metabolites. This was confirmed in human promyelocytic leukemia cells, where 2,5-diOH PCB 3 produced DNA strand breaks only if the cellular myeloperoxidase (MPx) was active, while the 2,5-quinone produced strand breaks independently of MPx (Xie et al. 2010). Surprisingly, all three OH-PCB 3s produced chromosome loss in V79 cells although with 10-fold lower efficacy than the diOH and quinoid metabolites (Table 3) (Zettner et al. 2007). This suggests protein binding, but the reactive intermediate is unknown. In fact, PCB 3 and PCB 77 both produced protein adducts in liver nuclei of rats and to a much smaller degree also DNA adducts (Pereg et al. 2001). It is noteworthy that non-genotoxic concentrations of PCB 77 combined with PCB 52, which alone did not produce chromosome damage in human lymphocytes in vitro, caused a vastly enhanced cytogenetic effect if combined, and this synergistic effect was also observed in rat bone marrow cells in vivo (Meisner et al. 1992, Sargent et al. 1991, Sargent et al. 1989). Also, several mono- and di-chlorinated PCBs were shown to very efficiently induce micronuclei formation and hprt mutations in hamster V79 cells that express CYP 2E1 and surprisingly the most active congeners were some non-planar tri- and tetra-chlorinated congeners (Liu et al. 2016, Wang et al. 2017, Zhang et al. 2016). V79 cells without CYP or other forms of P-450 were protected from PCB genotoxicity demonstrating that CYP 2E1 may be a predominant enzyme bioactivating PCBs with 1 to 4 chlorines to genotoxins.
Table 3. Genotoxicity endpoints for PCB 3 and its metabolites.
Genotoxic profile of PCB 3 and metabolites, 2-OH, 3-OH and 4-OH PCB 3, 3,4- and 2,5 di OH PCB 3, and 3,4- and 2,5 PCB 3 quinones. Endpoints measured were point mutations, chromosome loss, chromosome breaks, polyploidization, sister chromatid exchanges (SCE) and DNA strand breaks (COMET). Numbers are LOELs (micromolar concentrations). MPx=myeloperoxidase. Table was modified from (Robertson & Ludewig 2011).
| Compound | Point mutations |
Chromosome loss |
Chromosome breaks |
Polyploi- dization |
SCE | DNA strand break (COMET) |
|---|---|---|---|---|---|---|
| PCB 3 | - | - | - | - | - | |
| 2-OH | - | 50 | - | |||
| 3-OH | - | 100 | - | |||
| 4-OH | - | 75 | 75 | |||
| 3,4-diOH | - | 15 | 25 | - | 5 | |
| 2,5-diOH | - | 2.5 | 5 | 7.5 | - | + (MPx dependent) |
| 3,4-quinone | 0.6 | 5 | 15 | - | - | |
| 2,5-quinone | 0.5 | 2.5 | 1 | - | - | + (MPx independent |
The second step in carcinogenesis is promotion. Inhibition of gap junction intercellular communication (GJIC) is believed to be one mechanism of promotion and all non-dioxin-like PCBs and OH-PCBs tested were shown to be positive in an in vitro GJIC assay (Hamers et al. 2011, Machala et al. 2004). Hydrophobicity (log P) and molecular volume were found to significantly correlate with potency to down-regulate GJIC. Another marker of tumor-promoting activity, induction of proliferation of confluent liver cells, was positively correlated with the ability of dioxin-like PCBs and OH-PCBs to activate the AhR (Vondracek et al. 2005). Also, lower chlorinated OH-PCBs were shown to have significant estrogenic activity while anti-estrogenic activity was observed with persistent, higher chlorinated and a few lower chlorinated OH-PCB and was negatively correlated with hydrophobicity and molecular volume (Machala et al. 2004). Estrogenic activity was associated with the induction of cervicovaginal tract carcinoma in neonatal female mice exposed to OH-PCB 30 or OH-PCB 61, similar to the effect of DES (Martinez et al. 2005). Thus, lower chlorinated OH-PCBs may be cancer promoters by several mechanisms, inhibition of GJIC, loss of contact inhibition, and estrogenicity. OH-PCBs can readily transfer through the placenta and are found at higher levels in human fetal blood than in maternal blood (Park et al. 2008), raising serious concerns about possible carcinogenic activity of OH-PCBs in humans in utero.
OH-PCBs may also act as co-carcinogens by changing the bioavailability of other ultimate carcinogens. Many OH-PCBs are potent activators of the AhR, surpassing the efficacy of the parent PCB (Kamata et al. 2009). Since AhR activation is associated with carcinogenicity, these OH-PCBs may be more dangerous than their parent compounds. OH-PCBs were also shown to inhibit phase II metabolism. Specifically, the inhibition of glucuronosyl transferase by five different OH-PCBs was shown to increase the formation of DNA adducts by benzo(a)pyrene 7,8-dihydrodiol in isolated intact livers (James et al. 2004).
The sheer number of PCBs and their metabolites (837 mono-OH-PCBs alone!) makes it impossible to test even a small percentage of all these compounds for their toxic activity. Efforts are therefore made to use existing data for the development of predictive QSAR models (Niu et al. 2007). Applying such an approach, Ruiz calculated that the probability of mutagenicity was higher for lower chlorinated mono- and di-OH PCBs than with higher chlorinated metabolites (Ruiz et al. 2008). This group also examined the estrogenic activity of OH-PCBs (Ruiz et al. 2016, Ruiz et al. 2013). Analyzing the data of a large multi-center study with 17 in vitro assays and 20 ultra-pure non-dioxin-like PCBs by multivariant toxicity profiling and QSAR modeling revealed that generally smaller, ortho-substituted congeners (5 of 10 with <5 chlorines) were most active, while the second group with less active PCBs contained mostly higher chlorinated congeners (8 of 10 with >4 chlorines), further highlighting the potential toxicological importance of this usually overlooked group of PCBs, many of which are parent compounds of OH-PCBs (Stenberg et al. 2011).
All these findings indicate that OH-PCBs themselves may not be direct acting carcinogens but may have unexpected toxic and carcinogenic activity through their metabolic precursor or secondary metabolites or by interference with regulatory mechanisms. Our ability to predict which PCB metabolites are active and with which targets awaits further information.
OH-PCBs are endocrine hormone disrupting chemicals by indirect mechanisms
In addition to the roles that PCBs and OH-PCBs play in genotoxicity and carcinogenesis, in part through their hormone-like activity, they have also been shown to be endocrine and metabolic disrupting substances through indirect mechanisms (Gadupudi et al. 2015, Gadupudi et al. 2016a, Gadupudi et al. 2016b, Hamers et al. 2011, Heindel et al. 2015, Kawano et al. 2005, Liu et al. 2006, Ludewig et al. 2008, Machala et al. 2004, Quinete et al. 2014, Robertson & Ludewig 2011, Thayer et al. 2012). Epidemiological studies have shown that children exposed to PCBs in their early life have lower IQ than unexposed children, which may be due to endocrine disruption (Jacobson & Jacobson 1996). While a definitive link between OH-PCBs and these neurodevelopmental effects has not yet been established, several mechanisms for neurotoxicity of selected OH-PCBs have been investigated in vitro (Amano et al. 2010, Londono et al. 2010, Sethi et al. 2017, Shimokawa et al. 2006). Endocrine hormones are essential in growth, development, and sexual orientation. Phenolic metabolites of PCBs have been known as endocrine disrupting chemicals (EDCs) for some time (Kester et al. 2000, Lans et al. 1993, Lans et al. 1994). The effect of a subtle change in the level of hormones during development is very difficult to measure. Therefore, researchers have utilized various in vitro models to understand molecular mechanism of toxicity due to EDCs (Borgert et al. 2011). Endocrine hormones such as estradiol, thyroxine and triiodothyronine are small molecules which remain in the circulation bound to carrier proteins. OH-PCBs have been found to interact with and alter the effects of these hormones through various mechanisms. Using porcine pre-pubertal follicle culture, Ptak et al., have reported that there were significant alternations in the secretion of sex hormones by these ovary follicles when exposed to either PCB 3 or OH-PCB 3 (Ptak et al. 2005). Many OH-PCBs are inhibitors of mammalian cytosolic sulfotransferases (SULTs), enzymes that are important in the regulation of cellular concentrations of active estrogens (Kester et al. 2000). In addition to inhibition, some bioactivated OH-PCBs such as biphenyl quinones can modify the catalytic activity of human hydroxysteroid sulfotransferase hSULT2A1, which is responsible for the sulfation of hydroxysteroids such as dehydroepiandrosterone (Qin et al. 2013). Certain hydroxylated metabolites of commonly found non-dioxin-like PCBs have been reported to inhibit aromatase (CYP19) that is necessary for the conversion of androgens to estrogens (Antunes-Fernandes et al. 2011). In vitro studies using a yeast-glucocorticoid screening assay have shown that OH-PCBs exhibit anti-glucocorticoid properties by the inhibition of the glucocorticoid receptor (Antunes-Fernandes et al. 2011). OH-PCBs also have the potential to disrupt thyroid hormone homeostasis by inhibition of thyroid hormone sulfation (Schuur et al. 1999, Schuur et al. 1998) and by interfering with the binding of thyroxine to transthyretin (Grimm et al. 2013, Gutleb et al. 2010, Lans et al. 1993). PCB sulfates derived from OH-PCBs also bind with high affinity to transthyretin (Grimm et al., 2015c).
Both OH-PCBs and their sulfated forms in circulation can serve as ligands for human serum albumin, a protein that also functions in the retention and transport of thyroxine with additional functions in binding selected drugs and other molecules (Rodriguez et al. 2016). While the binding of a OH-PCB metabolite to serum albumin may potentially displace drugs, hormones, or other ligands, it is also important to note that the protein may serve as a reservoir for the PCB metabolite and/or facilitate its transfer to tissues that would be subject to toxic effects of the OH-PCBs.
CONCLUSIONS
PCBs are an important class of persistent environmental chemicals which were commercially synthesized for various industrial purposes in the twentieth century. The legacy of PCB pollution has left numerous health questions and concerns. PCBs are converted to OH-PCBs which tend to persist and bioaccumulate in trophic levels. The overall transformation of PCBs into phenols is facilitated by enzymatic biotransformation in animals, plants and other organisms, and potential environmental hydrolysis of sulfated and glucuronidated metabolites back to OH-PCBs. OH-PCBs may exert toxicities through a variety of mechanisms. Among those investigated so far include bioactivation to electrophilic metabolites that can form covalent adducts with DNA and other macromolecules, interference with hormonal signaling through modulations of transcriptional activity of hormone nuclear receptors, inhibition of enzymes that regulate cellular concentrations of of active hormones, and interference with the transport of hormones, through binding to circulating carrier proteins such as transthyretin and albumin. With the imminent global trends in slow biotransformation of bio-accumulated parent PCBs, further research on the mechanisms underlying health effects of OH-PCBs and their metabolites is warranted.
Acknowledgments
The authors are/were supported by a grant from the National Institute of Environmental Health Sciences (P42 ES013661). K.D. and G.S.G. recognize the Training Core of the Iowa Superfund Research Center for training and financial support. Opinions are those of the authors.
References
- AIHA. Potential Hazards of PCBs in the Building Environment 2013 [Google Scholar]
- Amano I, Miyazaki W, Iwasaki T, Shimokawa N, Koibuchi N. The effect of hydroxylated polychlorinated biphenyl (OH-PCB) on thyroid hormone receptor (TR)-mediated transcription through native-thyroid hormone response element (TRE) Ind Health. 2010;48:115–8. doi: 10.2486/indhealth.48.115. [DOI] [PubMed] [Google Scholar]
- Amaro AR, Oakley GG, Bauer U, Spielmann HP, Robertson LW. Metabolic activation of PCBs to quinones: reactivity toward nitrogen and sulfur nucleophiles and influence of superoxide dismutase. Chem Res Toxicol. 1996;9:623–9. doi: 10.1021/tx950117e. [DOI] [PubMed] [Google Scholar]
- Antunes-Fernandes EC, Bovee TF, Daamen FE, Helsdingen RJ, van den Berg M, van Duursen MB. Some OH-PCBs are more potent inhibitors of aromatase activity and (anti-) glucocorticoids than non-dioxin like (NDL)-PCBs and MeSO(2)-PCBs. Toxicol Lett. 2011;206:158–65. doi: 10.1016/j.toxlet.2011.07.008. [DOI] [PubMed] [Google Scholar]
- ATSDR. Toxicological Profile for Polychlorinated Biphenyls (PCBs) Toxicological Profiles; Atlanta, GA: 2000. [PubMed] [Google Scholar]
- ATSDR. Addendum to the Toxicological Profile for Polychlorinated Biphenyls (PCBs) Atlanta, GA: 2011. [PubMed] [Google Scholar]
- Ballschmiter K, Zell M. Analysis of polychlorinated biphenyls (PCB) by glass capillary gas chromatography. Z. Anal. Chem. 1980;302:20–31. [Google Scholar]
- Block WD, Cornish HH. Metabolism of Biphenyl and 4-Chlorobiphenyl in the Rabbit. Journal of Biological Chemistry. 1959;234:3301–3302. [Google Scholar]
- Borgert CJ, Mihaich EM, Quill TF, Marty MS, Levine SL, Becker RA. Evaluation of EPA's Tier 1 Endocrine Screening Battery and recommendations for improving the interpretation of screening results. Regulatory toxicology and pharmacology : RTP. 2011;59:397–411. doi: 10.1016/j.yrtph.2011.01.003. [DOI] [PubMed] [Google Scholar]
- Brodsky J, Ballschmiter K. Reversed phase liquid chromatography of PCBs as a basis for the calculation of water solubility and log Kow for polychlorobiphenyls. Z. Anal. Chem. 1988;331:295–301. [Google Scholar]
- CDC. Exposure Reports and updated Tables (2013) Centers for Disease Control and Prevention; 2013. National Biomonitoring Program. [Google Scholar]
- Currado GM, Harrad S. Comparison of Polychlorinated Biphenyl Concentrations in Indoor and Outdoor Air and the Potential Significance of Inhalation as a Human Exposure Pathway. Environmental science & technology. 1998;32:3043–3047. [Google Scholar]
- Dhakal K, He X, Lehmler HJ, Teesch LM, Duffel MW, Robertson LW. Identification of sulfated metabolites of 4-chlorobiphenyl (PCB3) in the serum and urine of male rats. Chem Res Toxicol. 2012;25:2796–804. doi: 10.1021/tx300416v. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dhakal K, Adamcakova-Dodd A, Lehmler HJ, Thorne PS, Robertson LW. Sulfate conjugates are urinary markers of inhalation exposure to 4-chlorobiphenyl (PCB3) Chem Res Toxicol. 2013;26:853–5. doi: 10.1021/tx4001539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dhakal K, Uwimana E, Adamcakova-Dodd A, Thorne PS, Lehmler HJ, Robertson LW. Disposition of phenolic and sulfated metabolites after inhalation exposure to 4-chlorobiphenyl (PCB3) in female rats. Chem Res Toxicol. 2014;27:1411–20. doi: 10.1021/tx500150h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ekuase EJ, Liu Y, Lehmler HJ, Robertson LW, Duffel MW. Structure-activity relationships for hydroxylated polychlorinated biphenyls as inhibitors of the sulfation of dehydroepiandrosterone catalyzed by human hydroxysteroid sulfotransferase SULT2A1. Chem Res Toxicol. 2011;24:1720–8. doi: 10.1021/tx200260h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Espandiari P, Glauert HP, Lehmler HJ, Lee EY, Srinivasan C, Robertson LW. Polychlorinated biphenyls as initiators in liver carcinogenesis: resistant hepatocyte model. Toxicol Appl Pharmacol. 2003;186:55–62. doi: 10.1016/s0041-008x(02)00018-2. [DOI] [PubMed] [Google Scholar]
- Espandiari P, Glauert HP, Lehmler HJ, Lee EY, Srinivasan C, Robertson LW. Initiating activity of 4-chlorobiphenyl metabolites in the resistant hepatocyte model. Toxicol Sci. 2004;79:41–6. doi: 10.1093/toxsci/kfh097. [DOI] [PubMed] [Google Scholar]
- Gadupudi G, Gourronc FA, Ludewig G, Robertson LW, Klingelhutz AJ. PCB126 inhibits adipogenesis of human preadipocytes. Toxicology in vitro : an international journal published in association with BIBRA. 2015;29:132–41. doi: 10.1016/j.tiv.2014.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gadupudi GS, Klaren WD, Olivier AK, Klingelhutz AJ, Robertson LW. PCB126-Induced Disruption in Gluconeogenesis and Fatty Acid Oxidation Precedes Fatty Liver in Male Rats. Toxicol Sci. 2016a;149:98–110. doi: 10.1093/toxsci/kfv215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gadupudi GS, Klingelhutz AJ, Robertson LW. Diminished Phosphorylation of CREB Is a Key Event in the Dysregulation of Gluconeogenesis and Glycogenolysis in PCB126 Hepatotoxicity. Chem Res Toxicol. 2016b doi: 10.1021/acs.chemrestox.6b00172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grimm FA, Lehmler HJ, He X, Robertson LW, Duffel MW. Sulfated metabolites of polychlorinated biphenyls are high-affinity ligands for the thyroid hormone transport protein transthyretin. Environmental health perspectives. 2013;121:657–62. doi: 10.1289/ehp.1206198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grimm FA, He X, Teesch LM, Lehmler HJ, Robertson LW, Duffel MW. Tissue Distribution, Metabolism, and Excretion of 3,3'-Dichloro-4'-sulfooxy-biphenyl in the Rat. Environmental science & technology. 2015a;49:8087–95. doi: 10.1021/acs.est.5b01499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grimm FA, Hu D, Kania-Korwel I, Lehmler HJ, Ludewig G, Hornbuckle KC, Duffel MW, Bergman A, Robertson LW. Metabolism and metabolites of polychlorinated biphenyls. Critical reviews in toxicology. 2015b;45:245–72. doi: 10.3109/10408444.2014.999365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grimm FA, Lehmler HJ, Koh WX, DeWall J, Teesch LM, Hornbuckle KC, Thorne PS, Robertson LW, Duffel MW. Identification of a sulfate metabolite of PCB 11 in human serum. Environ Int. 2017;98:120–128. doi: 10.1016/j.envint.2016.10.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gutleb AC, Cenijn P, Velzen M, Lie E, Ropstad E, Skaare JU, Malmberg T, Bergman A, Gabrielsen GW, Legler J. In vitro assay shows that PCB metabolites completely saturate thyroid hormone transport capacity in blood of wild polar bears (Ursus maritimus) Environmental science & technology. 2010;44:3149–54. doi: 10.1021/es903029j. [DOI] [PubMed] [Google Scholar]
- Haglund P. Enantioselective separation of polychlorinated biphenyl atropisomers using chiral high-performance liquid chromatography. J Chromatogr A. 1996a;724:219–228. [Google Scholar]
- Haglund P. Isolation and characterisation of polychlorinated biphenyl (PCB) atropisomers. Chemosphere. 1996b;32:2133–2140. [Google Scholar]
- Hamers T, Kamstra JH, Cenijn PH, Pencikova K, Palkova L, Simeckova P, Vondracek J, Andersson PL, Stenberg M, Machala M. In vitro toxicity profiling of ultrapure non-dioxin-like polychlorinated biphenyl congeners and their relative toxic contribution to PCB mixtures in humans. Toxicol Sci. 2011;121:88–100. doi: 10.1093/toxsci/kfr043. [DOI] [PubMed] [Google Scholar]
- Heindel JJ, et al. Parma consensus statement on metabolic disruptors. Environmental Health. 2015;14:1–7. doi: 10.1186/s12940-015-0042-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu D, Martinez A, Hornbuckle KC. Discovery of non-aroclor PCB (3,3'-dichlorobiphenyl) in Chicago air. Environmental science & technology. 2008;42:7873–7. doi: 10.1021/es801823r. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu D, Hornbuckle KC. Inadvertent polychlorinated biphenyls in commercial paint pigments. Environmental science & technology. 2010;44:2822–7. doi: 10.1021/es902413k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu D, Lehmler HJ, Martinez A, Wang K, Hornbuckle KC. Atmospheric PCB congeners across Chicago. Atmospheric environment. 2010;44:1550–1557. doi: 10.1016/j.atmosenv.2010.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu X, Adamcakova-Dodd A, Thorne PS. The fate of inhaled (14)C-labeled PCB11 and its metabolites in vivo. Environ Int. 2014;63:92–100. doi: 10.1016/j.envint.2013.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- IARC. IARC Monographs on teh evaluation of the ccarcinogenic risk of chemicals to humans. Vol. 107. IARC; Lyon: 2013. Polychlorinated biphenyls and polybrominated biphenyls. [Google Scholar]
- Jacobson JL, Jacobson SW. Intellectual Impairment in Children Exposed to Polychlorinated Biphenyls in Utero. New England Journal of Medicine. 1996;335:783–789. doi: 10.1056/NEJM199609123351104. [DOI] [PubMed] [Google Scholar]
- James MO, Kleinow KM, Zhang Y, Zheng R, Wang L, Faux LR. Increased toxicity of benzo(a)pyrene-7,8-dihydrodiol in the presence of polychlorobiphenylols. Mar Environ Res. 2004;58:343–6. doi: 10.1016/j.marenvres.2004.03.079. [DOI] [PubMed] [Google Scholar]
- James MO, Ambadapadi S. Interactions of cytosolic sulfotransferases with xenobiotics. Drug Metabolism Reviews. 2013;45:401–414. doi: 10.3109/03602532.2013.835613. [DOI] [PubMed] [Google Scholar]
- Janssen EM, Thompson JK, Luoma SN, Luthy RG. PCB-induced changes of a benthic community and expected ecosystem recovery following in situ sorbent amendment. Environmental toxicology and chemistry / SETAC. 2011;30:1819–26. doi: 10.1002/etc.574. [DOI] [PubMed] [Google Scholar]
- Kamata R, Shiraishi F, Nakajima D, Takigami H, Shiraishi H. Mono-hydroxylated polychlorinated biphenyls are potent aryl hydrocarbon receptor ligands in recombinant yeast cells. Toxicology in vitro : an international journal published in association with BIBRA. 2009;23:736–43. doi: 10.1016/j.tiv.2009.03.004. [DOI] [PubMed] [Google Scholar]
- Kamata R, Shiraishi F, Kageyama S, Nakajima D. Detection and measurement of the agonistic activities of PCBs and mono-hydroxylated PCBs to the constitutive androstane receptor using a recombinant yeast assay. Toxicology in vitro : an international journal published in association with BIBRA. 2015;29:1859–67. doi: 10.1016/j.tiv.2015.07.021. [DOI] [PubMed] [Google Scholar]
- Kania-Korwel I, Shaikh N, Hornbuckle KC, Robertson LW, Lehmler H-J. Enantioselective disposition of PCB 136 (2,2',3,3',6,6'-hexachlorobiphenyl) in C57BL/6 mice after oral and intraperitoneal administration. Chirality. 2007;19:56–66. doi: 10.1002/chir.20342. [DOI] [PubMed] [Google Scholar]
- Kania-Korwel I, Hornbuckle KC, Robertson LW, Lehmler HJ. Influence of dietary fat on the enantioselective disposition of 2,2',3,3',6,6'-hexachlorobiphenyl (PCB 136) in female mice. Food Chem. Toxicol. 2008a;46:637–644. doi: 10.1016/j.fct.2007.09.072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kania-Korwel I, Hrycay EG, Bandiera S, Lehmler H-J. 2,2',3,3',6,6'-Hexachlorobiphenyl (PCB 136) atropisomers interact enantioselectively with hepatic microsomal cytochrome P450 enzymes. Chem. Res. Toxicol. 2008b;21:1295–1303. doi: 10.1021/tx800059j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kania-Korwel I, Vyas SM, Song Y, Lehmler H-J. Gas chromatographic separation of methoxylated polychlorinated biphenyl atropisomers. J. Chromatogr. A. 2008c;1207:146–154. doi: 10.1016/j.chroma.2008.08.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kania-Korwel I, Xie W, Hornbuckle KC, Robertson LW, Lehmler H-J. Enantiomeric enrichment of 2,2’,3,3’,6,6’-hexachlorobiphenyl (PCB 136) in mice after induction of CYP enzymes. Arch. Environ. Contam. Toxicol. 2008d;55:510–517. doi: 10.1007/s00244-007-9111-4. [DOI] [PubMed] [Google Scholar]
- Kania-Korwel I, El-Komy MHME, Veng-Pedersen P, Lehmler HJ. Clearance of polychlorinated biphenyl atropisomers is enantioselective in female C57Bl/6 mice. Environ. Sci. Technol. 2010;44:2828–2835. doi: 10.1021/es901781p. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kania-Korwel I, Duffel MW, Lehmler H-J. Gas chromatographic analysis with chiral cyclodextrin phases reveals the enantioselective formation of hydroxylated polychlorinated biphenyls by rat liver microsomes. Environ. Sci. Technol. 2011a;45:9590–9596. doi: 10.1021/es2014727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kania-Korwel I, Duffel MW, Lehmler HJ. Gas chromatographic analysis with chiral cyclodextrin phases reveals the enantioselective formation of hydroxylated polychlorinated biphenyls by rat liver microsomes. Environ. Sci. Technol. 2011b;45:9590–9596. doi: 10.1021/es2014727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kania-Korwel I, Barnhart CD, Stamou M, Truong KM, El-Komy MH, Lein PJ, Veng-Pedersen P, Lehmler H-J. 2,2',3,5',6-Pentachlorobiphenyl (PCB 95) and its hydroxylated metabolites are enantiomerically enriched in female mice. Environ. Sci. Technol. 2012;46:11393–401. doi: 10.1021/es302810t. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kania-Korwel I, Lehmler HJ. Assigning atropisomer elution orders using atropisomerically enriched polychlorinated biphenyl fractions generated by microsomal metabolism. J Chromatogr A. 2013;1278:133–144. doi: 10.1016/j.chroma.2012.12.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kania-Korwel I, Barnhart CD, Lein PJ, Lehmler H-J. Effect of pregnancy on the disposition of 2,2',3,5',6-pentachlorobiphenyl (PCB 95) atropisomers and their hydroxylated metabolites in female mice. Chem. Res. Toxicol. 2015;28:1774–1783. doi: 10.1021/acs.chemrestox.5b00241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kania-Korwel I, Lehmler HJ. Toxicokinetics of chiral polychlorinated biphenyls across different species--a review. Environ Sci Pollut Res Int. 2016a;23:2058–80. doi: 10.1007/s11356-015-4383-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kania-Korwel I, Lehmler HJ. Chiral polychlorinated biphenyls: absorption, metabolism and excretion--a review. Environ Sci Pollut Res Int. 2016b;23:2042–57. doi: 10.1007/s11356-015-4150-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kania-Korwel I, Lukasiewicz T, Barnhart CD, Stamou M, Chung H, Kelly KM, Bandiera S, Lein PJ, Lehmler H-J. Congener-specific disposition of chiral polychlorinated biphenyls in lactating mice and their offspring: Implications for PCB developmental neurotoxicity. Toxicol. Sci. 2017 doi: 10.1093/toxsci/kfx071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawano M, Hasegawa J, Enomoto T, Onishi H, Nishio Y, Matsuda M, Wakimoto T. Hydroxylated polychlorinated biphenyls (OH-PCBs): recent advances in wildlife contamination study. Environmental sciences : an international journal of environmental physiology and toxicology. 2005;12:315–24. [PubMed] [Google Scholar]
- Kester MH, Bulduk S, Tibboel D, Meinl W, Glatt H, Falany CN, Coughtrie MW, Bergman A, Safe SH, Kuiper GG, Schuur AG, Brouwer A, Visser TJ. Potent inhibition of estrogen sulfotransferase by hydroxylated PCB metabolites: a novel pathway explaining the estrogenic activity of PCBs. Endocrinology. 2000;141:1897–900. doi: 10.1210/endo.141.5.7530. [DOI] [PubMed] [Google Scholar]
- Koh WX, Hornbuckle KC, Thorne PS. Human Serum from Urban and Rural Adolescents and Their Mothers Shows Exposure to Polychlorinated Biphenyls Not Found in Commercial Mixtures. Environmental science & technology. 2015;49:8105–12. doi: 10.1021/acs.est.5b01854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai IK, Chai Y, Simmons D, Watson WH, Tan R, Haschek WM, Wang K, Wang B, Ludewig G, Robertson LW. Dietary selenium as a modulator of PCB 126-induced hepatotoxicity in male Sprague-Dawley rats. Toxicol Sci. 2011;124:202–14. doi: 10.1093/toxsci/kfr215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laird BD, Goncharov AB, Chan HM. Body burden of metals and persistent organic pollutants among Inuit in the Canadian Arctic. Environ Int. 2013;59:33–40. doi: 10.1016/j.envint.2013.05.010. [DOI] [PubMed] [Google Scholar]
- Lans MC, Klasson-Wehler E, Willemsen M, Meussen E, Safe S, Brouwer A. Structure-dependent, competitive interaction of hydroxy-polychlorobiphenyls, -dibenzo-p-dioxins and -dibenzofurans with human transthyretin. Chemico-biological interactions. 1993;88:7–21. doi: 10.1016/0009-2797(93)90081-9. [DOI] [PubMed] [Google Scholar]
- Lans MC, Spiertz C, Brouwer A, Koeman JH. Different competition of thyroxine binding to transthyretin and thyroxine-binding globulin by hydroxy-PCBs, PCDDs and PCDFs. European journal of pharmacology. 1994;270:129–36. doi: 10.1016/0926-6917(94)90054-x. [DOI] [PubMed] [Google Scholar]
- Lauby-Secretan B, Loomis D, Baan R, El Ghissassi F, Bouvard V, Benbrahim-Tallaa L, Guha N, Grosse Y, Straif K. Use of mechanistic data in the IARC evaluations of the carcinogenicity of polychlorinated biphenyls and related compounds. Environ Sci Pollut Res Int. 2015 doi: 10.1007/s11356-015-4829-4. [DOI] [PubMed] [Google Scholar]
- Lehmann L, H LE, P AK, L WR, Ludewig G. 4-monochlorobiphenyl (PCB3) induces mutations in the livers of transgenic Fisher 344 rats. Carcinogenesis. 2007;28:471–8. doi: 10.1093/carcin/bgl157. [DOI] [PubMed] [Google Scholar]
- Lehmler H-J, Robertson LW. Atropisomers of PCBs. In: Robertson LW, Hansen L, editors. Recent Advances in teh Environmental Toxicology and Health Effects of PCBs. University Press of Kentucky; Lexington, KY: 2001. pp. 62–65. [Google Scholar]
- Lehmler HJ, Robertson LW, Garrison AW, Kodavanti PR. Effects of PCB 84 enantiomers on [3H]-phorbol ester binding in rat cerebellar granule cells and 45Ca2+-uptake in rat cerebellum. Toxicol Lett. 2005;156:391–400. doi: 10.1016/j.toxlet.2004.12.011. [DOI] [PubMed] [Google Scholar]
- Lehmler HJ, Harrad SJ, Huhnerfuss H, Kania-Korwel I, Lee CM, Lu Z, Wong CS. Chiral polychlorinated biphenyl transport, metabolism, and distribution: a review. Environmental science & technology. 2010;44:2757–66. doi: 10.1021/es902208u. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y, Apak TI, Lehmler HJ, Robertson LW, Duffel MW. Hydroxylated polychlorinated biphenyls are substrates and inhibitors of human hydroxysteroid sulfotransferase SULT2A1. Chem Res Toxicol. 2006;19:1420–5. doi: 10.1021/tx060160+. [DOI] [PubMed] [Google Scholar]
- Liu Y, Smart JT, Song Y, Lehmler HJ, Robertson LW, Duffel MW. Structure-activity relationships for hydroxylated polychlorinated biphenyls as substrates and inhibitors of rat sulfotransferases and modification of these relationships by changes in thiol status. Drug metabolism and disposition: the biological fate of chemicals. 2009;37:1065–72. doi: 10.1124/dmd.108.026021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y, Hu K, Jia H, Jin G, Glatt H, Jiang H. Potent mutagenicity of some non-planar tri- and tetrachlorinated biphenyls in mammalian cells, human CYP2E1 being a major activating enzyme. Arch Toxicol. 2016 doi: 10.1007/s00204-016-1904-7. [DOI] [PubMed] [Google Scholar]
- Londono M, Shimokawa N, Miyazaki W, Iwasaki T, Koibuchi N. Hydroxylated PCB induces Ca2+ oscillations and alterations of membrane potential in cultured cortical cells. J Appl Toxicol. 2010;30:334–42. doi: 10.1002/jat.1501. [DOI] [PubMed] [Google Scholar]
- Lu Z, Kania-Korwel I, Lehmler HJ, Wong CS. Stereoselective formation of mono- and dihydroxylated polychlorinated biphenyls by rat cytochrome P450 2B1. Environ. Sci. Technol. 2013:12184–92. doi: 10.1021/es402838f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ludewig G, Lehmann L, Esch H, Robertson LW. Metabolic Activation of PCBs to Carcinogens in Vivo - A Review. Environmental toxicology and pharmacology. 2008;25:241–6. doi: 10.1016/j.etap.2007.10.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma C, Zhai G, Wu H, Kania-Korwel I, Lehmler HJ, Schnoor JL. Identification of a novel hydroxylated metabolite of 2,2',3,5',6-pentachlorobiphenyl formed in whole poplar plants. Environmental science and pollution research international. 2016;23:2089–2098. doi: 10.1007/s11356-015-5939-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Machala M, Blaha L, Lehmler HJ, Pliskova M, Majkova Z, Kapplova P, Sovadinova I, Vondracek J, Malmberg T, Robertson LW. Toxicity of hydroxylated and quinoid PCB metabolites: inhibition of gap junctional intercellular communication and activation of aryl hydrocarbon and estrogen receptors in hepatic and mammary cells. Chem Res Toxicol. 2004;17:340–7. doi: 10.1021/tx030034v. [DOI] [PubMed] [Google Scholar]
- Marek RF, Thorne PS, Wang K, Dewall J, Hornbuckle KC. PCBs and OH-PCBs in serum from children and mothers in urban and rural U.S. communities. Environmental science & technology. 2013;47:3353–61. doi: 10.1021/es304455k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marek RF, Thorne PS, DeWall J, Hornbuckle KC. Variability in PCB and OH-PCB serum levels in children and their mothers in urban and rural U.S. communities. Environmental science & technology. 2014;48:13459–67. doi: 10.1021/es502490w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez A, Norstrom K, Wang K, Hornbuckle KC. Polychlorinated biphenyls in the surficial sediment of Indiana Harbor and Ship Canal, Lake Michigan. Environ Int. 2010a;36:849–54. doi: 10.1016/j.envint.2009.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez A, Wang K, Hornbuckle KC. Fate of PCB congeners in an industrial harbor of Lake Michigan. Environmental science & technology. 2010b;44:2803–8. doi: 10.1021/es902911a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez A, Hadnott BN, Awad AM, Herkert NJ, Tomsho K, Basra K, Scammell MK, Heiger-Bernays W, Hornbuckle KC. Release of Airborne Polychlorinated Biphenyls from New Bedford Harbor Results in Elevated Concentrations in the Surrounding Air. Environ Sci Technol Lett. 2017;4:127–131. doi: 10.1021/acs.estlett.7b00047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez JM, Stephens LC, Jones LA. Long-term effects of neonatal exposure to hydroxylated polychlorinated biphenyls in the BALB/cCrgl mouse. Environmental health perspectives. 2005;113:1022–6. doi: 10.1289/ehp.7735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLean MR, Bauer U, Amaro AR, Robertson LW. Identification of catechol and hydroquinone metabolites of 4-monochlorobiphenyl. Chem Res Toxicol. 1996;9:158–64. doi: 10.1021/tx950083a. [DOI] [PubMed] [Google Scholar]
- Meisner LF, Roloff B, Sargent L, Pitot H. Interactive cytogenetic effects on rat bone-marrow due to chronic ingestion of 2,5,2',5' and 3,4,3',4' PCBs. Mutat Res. 1992;283:179–83. doi: 10.1016/0165-7992(92)90105-q. [DOI] [PubMed] [Google Scholar]
- Milanowski B, Lulek J, Lehmler H-J, Kania-Korwel I. Assesment of disposition of chirl polychlorinated biphenyls in female mdr 1a/b knockout versus wild-type mice using multivariate analyses. Environ. Int. 2010a;36:884–892. doi: 10.1016/j.envint.2009.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milanowski B, Lulek J, Lehmler HJ, Kania-Korwel I. Assessment of the disposition of chiral polychlorinated biphenyls in female mdr 1a/b knockout versus wild-type mice using multivariate analyses. Environ Int. 2010b;36:884–92. doi: 10.1016/j.envint.2009.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niu J, Long X, Shi S. Quantitative structure-activity relationships for prediction of the toxicity of hydroxylated and quinoid PCB metabolites. J Mol Model. 2007;13:163–9. doi: 10.1007/s00894-006-0153-8. [DOI] [PubMed] [Google Scholar]
- Park JS, Bergman A, Linderholm L, Athanasiadou M, Kocan A, Petrik J, Drobna B, Trnovec T, Charles MJ, Hertz-Picciotto I. Placental transfer of polychlorinated biphenyls, their hydroxylated metabolites and pentachlorophenol in pregnant women from eastern Slovakia. Chemosphere. 2008;70:1676–84. doi: 10.1016/j.chemosphere.2007.07.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parkinson A, Ogilvie BW, Buckley DB, Kazmi F, Czerwinski M, Parkinson O. Biotransformation of Xenobiotics, Casarett and Doull's Toxicology: The Basic Science of Poisons, 8e. McGraw-Hill Education; New York, NY: 2012. [Google Scholar]
- Pavuk M, et al. Predictors of serum polychlorinated biphenyl concentrations in Anniston residents. The Science of the total environment. 2014;496:624–34. doi: 10.1016/j.scitotenv.2014.06.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pereg D, Tampal N, Espandiari P, Robertson LW. Distribution and macromolecular binding of benzo[a]pyrene and two polychlorinated biphenyl congeners in female mice. Chemico-biological interactions. 2001;137:243–58. doi: 10.1016/s0009-2797(01)00256-3. [DOI] [PubMed] [Google Scholar]
- Persoon C, Peters TM, Kumar N, Hornbuckle KC. Spatial distribution of airborne polychlorinated biphenyls in Cleveland, Ohio and Chicago, Illinois. Environmental science & technology. 2010;44:2797–802. doi: 10.1021/es901691s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pessah IN, Lehmler HJ, Robertson LW, Perez CF, Cabrales E, Bose DD, Feng W. Enantiomeric specificity of (−)-2,2',3,3',6,6'-hexachlorobiphenyl toward ryanodine receptor types 1 and 2. Chem Res Toxicol. 2009;22:201–7. doi: 10.1021/tx800328u. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pham-Tuan H, Larsson C, Hoffmann F, Bergman A, Froeba M, Huehnerfuss H. Enantioselective semipreparative HPLC separation of PCB metabolites and their absolute structure elucidation using electronic and vibrational circular dichroism. Chirality. 2005;17:266–280. doi: 10.1002/chir.20158. [DOI] [PubMed] [Google Scholar]
- Ptak A, Ludewig G, Lehmler HJ, Wojtowicz AK, Robertson LW, Gregoraszczuk EL. Comparison of the actions of 4-chlorobiphenyl and its hydroxylated metabolites on estradiol secretion by ovarian follicles in primary cells in culture. Reproductive toxicology (Elmsford, N.Y.) 2005;20:57–64. doi: 10.1016/j.reprotox.2004.12.003. [DOI] [PubMed] [Google Scholar]
- Püttmann M, Oesch F, Robertson L, Mannschreck A. Characteristics of polychlorinated biphenyl (PCB) atropisomers. Chemosphere. 1986;15:2061–20164. [Google Scholar]
- Püttmann M, Mannschreck A, Oesch F, Robertson L. Chiral effects in the induction of drug-metabolizing enzymes using synthetic atropisomers of polychlorinated biphenyls (PCBs) Biochemical pharmacology. 1989;38:1345–52. doi: 10.1016/0006-2952(89)90342-0. [DOI] [PubMed] [Google Scholar]
- Püttmann M, Arand M, Oesch F, Mannschreck A, Robertson L. Chirality and the induction of xenobiotic-metabolizing enzymes: Effects of the atropisomers of the polychlorinated biphenyl 2,2',3,4,4',6-hexachlorobiphenyl. In: Holmstedt B, Frank H, Testa B, editors. Chirality and Biological Activity. Alan R. Liss Inc; New York: 1990. pp. 177–184. [Google Scholar]
- Qin X, Lehmler HJ, Teesch LM, Robertson LW, Duffel MW. Chlorinated biphenyl quinones and phenyl-2,5-benzoquinone differentially modify the catalytic activity of human hydroxysteroid sulfotransferase hSULT2A1. Chem Res Toxicol. 2013;26:1474–85. doi: 10.1021/tx400207q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quinete N, Schettgen T, Bertram J, Kraus T. Occurrence and distribution of PCB metabolites in blood and their potential health effects in humans: a review. Environ Sci Pollut Res Int. 2014;21:11951–72. doi: 10.1007/s11356-014-3136-9. [DOI] [PubMed] [Google Scholar]
- Robertson LW, Ludewig G. Polychlorinated Biphenyl (PCB) carcinogenicity with special emphasis on airborne PCBs. Gefahrst Reinhalt Luft. 2011;71:25–32. [PMC free article] [PubMed] [Google Scholar]
- Rodman LE, Shedlofsky SI, Mannschreck A, Puttmann M, Swim AT, Robertson LW. Differential potency of atropisomers of polychlorinated biphenyls on cytochrome P450 induction and uroporphyrin accumulation in the chick embryo hepatocyte culture. Biochemical pharmacology. 1991;41:915–22. doi: 10.1016/0006-2952(91)90196-c. [DOI] [PubMed] [Google Scholar]
- Rodriguez EA, Li X, Lehmler HJ, Robertson LW, Duffel MW. Sulfation of Lower Chlorinated Polychlorinated Biphenyls Increases Their Affinity for the Major Drug-Binding Sites of Human Serum Albumin. Environmental science & technology. 2016;50:5320–7. doi: 10.1021/acs.est.6b00484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruiz P, Faroon O, Moudgal CJ, Hansen H, De Rosa CT, Mumtaz M. Prediction of the health effects of polychlorinated biphenyls (PCBs) and their metabolites using quantitative structure–activity relationship (QSAR) Toxicology Letters. 2008;181:53–65. doi: 10.1016/j.toxlet.2008.06.870. [DOI] [PubMed] [Google Scholar]
- Ruiz P, Myshkin E, Quigley P, Faroon O, Wheeler JS, Mumtaz MM, Brennan RJ. Assessment of hydroxylated metabolites of polychlorinated biphenyls as potential xenoestrogens: a QSAR comparative analysis *. SAR QSAR Environ Res. 2013;24:393–416. doi: 10.1080/1062936X.2013.781537. [DOI] [PubMed] [Google Scholar]
- Ruiz P, Ingale K, Wheeler JS, Mumtaz M. 3D QSAR studies of hydroxylated polychlorinated biphenyls as potential xenoestrogens. Chemosphere. 2016;144:2238–46. doi: 10.1016/j.chemosphere.2015.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sacco JC, James MO. Sulfonation of environmental chemicals and their metabolites in the polar bear (Ursus maritimus) Drug metabolism and disposition: the biological fate of chemicals. 2005;33:1341–8. doi: 10.1124/dmd.105.004648. [DOI] [PubMed] [Google Scholar]
- Sacco JC, Lehmler HJ, Robertson LW, Li W, James MO. Glucuronidation of polychlorinated biphenylols and UDP-glucuronic acid concentrations in channel catfish liver and intestine. Drug metabolism and disposition: the biological fate of chemicals. 2008;36:623–30. doi: 10.1124/dmd.107.019596. [DOI] [PubMed] [Google Scholar]
- Sargent L, Roloff B, Meisner L. In vitro chromosome damage due to PCB interactions. Mutat Res. 1989;224:79–88. doi: 10.1016/0165-1218(89)90006-2. [DOI] [PubMed] [Google Scholar]
- Sargent L, Dragan YP, Erickson C, Laufer CJ, Pitot HC. Study of the separate and combined effects of the non-planar 2,5,2',5'- and the planar 3,4,3',4'-tetrachlorobiphenyl in liver and lymphocytes in vivo. Carcinogenesis. 1991;12:793–800. doi: 10.1093/carcin/12.5.793. [DOI] [PubMed] [Google Scholar]
- Schnellmann R, Putnam C, Sipes I. Metabolism of 2,2',3,3',6,6'-hexachlorobiphenyl and 2,2',4,4',5,5'-hexachlorobiphenyl by human hepatic microsomes. Biochem. Pharmacol. 1983;32:3233–3239. doi: 10.1016/0006-2952(83)90209-5. [DOI] [PubMed] [Google Scholar]
- Schuur AG, Legger FF, van Meeteren ME, Moonen MJ, van Leeuwen-Bol I, Bergman A, Visser TJ, Brouwer A. In vitro inhibition of thyroid hormone sulfation by hydroxylated metabolites of halogenated aromatic hydrocarbons. Chem Res Toxicol. 1998;11:1075–81. doi: 10.1021/tx9800046. [DOI] [PubMed] [Google Scholar]
- Schuur AG, Bergman A, Brouwer A, Visser TJ. Effects of pentachlorophenol and hydroxylated polychlorinated biphenyls on thyroid hormone conjugation in a rat and a human hepatoma cell line. Toxicology in vitro : an international journal published in association with BIBRA. 1999;13:417–25. doi: 10.1016/s0887-2333(99)00005-3. [DOI] [PubMed] [Google Scholar]
- Sethi S, Keil KP, Chen H, Hayakawa K, Li X, Lin Y, Lehmler HJ, Puschner B, Lein PJ. Detection of 3,3'-Dichlorobiphenyl in Human Maternal Plasma and Its Effects on Axonal and Dendritic Growth in Primary Rat Neurons. Toxicol Sci. 2017 doi: 10.1093/toxsci/kfx100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shanahan CE, Spak SN, Martinez A, Hornbuckle KC. Inventory of PCBs in Chicago and Opportunities for Reduction in Airborne Emissions and Human Exposure. Environmental science & technology. 2015;49:13878–88. doi: 10.1021/acs.est.5b00906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimada T, Kakimoto K, Takenaka S, Koga N, Uehara S, Murayama N, Yamazaki H, Kim D, Guengerich FP, Komori M. Roles of human CYP2A6 and monkey CYP2A24 and 2A26 cytochrome P450 enzymes in the oxidation of 2,5,2',5'-tetrachlorobiphenyl. Drug Metab. Dispos. 2016;44:1899–1909. doi: 10.1124/dmd.116.072991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimokawa N, Miyazaki W, Iwasaki T, Koibuchi N. Low dose hydroxylated PCB induces c-Jun expression in PC12 cells. Neurotoxicology. 2006;27:176–83. doi: 10.1016/j.neuro.2005.09.005. [DOI] [PubMed] [Google Scholar]
- Silberhorn EM, Glauert HP, Robertson LW. Carcinogenicity of polyhalogenated biphenyls: PCBs and PBBs. Critical reviews in toxicology. 1990;20:440–96. doi: 10.3109/10408449009029331. [DOI] [PubMed] [Google Scholar]
- Stamou M, Wu X, Kania-Korwel I, Lehmler HJ, Lein PJ. Cytochrome p450 mRNA expression in the rodent brain: species-, sex-, and region-dependent differences. Drug metabolism and disposition: the biological fate of chemicals. 2014;42:239–44. doi: 10.1124/dmd.113.054239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stenberg M, Hamers T, Machala M, Fonnum F, Stenius U, Lauy AA, van Duursen MB, Westerink RH, Fernandes EC, Andersson PL. Multivariate toxicity profiles and QSAR modeling of non-dioxin-like PCBs--an investigation of in vitro screening data from ultra-pure congeners. Chemosphere. 2011;85:1423–9. doi: 10.1016/j.chemosphere.2011.08.019. [DOI] [PubMed] [Google Scholar]
- Stowell CL, Barvian KK, Young Peter CM, Bigsby RM, Verdugo Dawn E, Bertozzi CR, Widlanski TS. A Role for Sulfation-Desulfation in the Uptake of Bisphenol A into Breast Tumor Cells. Chemistry & Biology. 2006;13:891–897. doi: 10.1016/j.chembiol.2006.06.016. [DOI] [PubMed] [Google Scholar]
- Tampal N, Lehmler HJ, Espandiari P, Malmberg T, Robertson LW. Glucuronidation of hydroxylated polychlorinated biphenyls (PCBs) Chem Res Toxicol. 2002;15:1259–66. doi: 10.1021/tx0200212. [DOI] [PubMed] [Google Scholar]
- Thayer KA, Heindel JJ, Bucher JR, Gallo MA. Role of environmental chemicals in diabetes and obesity: a National Toxicology Program workshop review. Environmental health perspectives. 2012;120:779–89. doi: 10.1289/ehp.1104597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thelen K, Dressman JB. Cytochrome P450-mediated metabolism in the human gut wall. The Journal of pharmacy and pharmacology. 2009;61:541–58. doi: 10.1211/jpp/61.05.0002. [DOI] [PubMed] [Google Scholar]
- Toda M, Matsumura C, Tsurukawa M, Okuno T, Nakano T, Inoue Y, Mori T. Absolute configuration of atropisomeric polychlorinated biphenyl 183 enantiomerically enriched in human samples. J Phys Chem A. 2012;116:9340–6. doi: 10.1021/jp306363n. [DOI] [PubMed] [Google Scholar]
- Tue NM, Takahashi S, Subramanian A, Sakai S, Tanabe S. Environmental contamination and human exposure to dioxin-related compounds in e-waste recycling sites of developing countries. Environmental science. Processes & impacts. 2013;15:1326–31. doi: 10.1039/c3em00086a. [DOI] [PubMed] [Google Scholar]
- Uwimana E, Li X, Lehmler H-J. 2,2′,3,5′,6-Pentachlorobiphenyl (PCB 95) is atropselectively metabolized to para-hydroxylated metabolites by human liver microsomes. Chem. Res. Toxicol. 2016;29:2108–2110. doi: 10.1021/acs.chemrestox.6b00371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uwimana E, Maiers A, Li X, Lehmler HJ. Microsomal metabolism of prochiral polychlorinated biphenyls results in the enantioselective formation of chiral metabolites. Environ. Sci. Technol. 2017;51:1820–1829. doi: 10.1021/acs.est.6b05387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van den Hurk P, Kubiczak GA, Lehmler HJ, James MO. Hydroxylated polychlorinated biphenyls as inhibitors of the sulfation and glucuronidation of 3-hydroxy-benzo[a]pyrene. Environmental health perspectives. 2002;110:343–8. doi: 10.1289/ehp.02110343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vickers AEM, Sipes G, Brendel K. Metabolism-related spectral characterization and subcellular distribution of polychlorinated biphenyl congeners in isolated rat hepatocytes. Biochem. Pharmacol. 1986;35:297–306. doi: 10.1016/0006-2952(86)90529-0. [DOI] [PubMed] [Google Scholar]
- Vondracek J, Machala M, Bryja V, Chramostova K, Krcmar P, Dietrich C, Hampl A, Kozubik A. Aryl hydrocarbon receptor-activating polychlorinated biphenyls and their hydroxylated metabolites induce cell proliferation in contact-inhibited rat liver epithelial cells. Toxicol Sci. 2005;83:53–63. doi: 10.1093/toxsci/kfi009. [DOI] [PubMed] [Google Scholar]
- Waller SC, He YA, Harlow GR, He YQ, Mash EA, Halpert JR. 2,2',3,3',6,6'-hexachlorobiphenyl hydroxylation by active site mutants of cytochrome P450 2B1 and 2B11. Chem. Res. Toxicol. 1999a;12:690–699. doi: 10.1021/tx990030j. [DOI] [PubMed] [Google Scholar]
- Waller SC, He YA, Harlow GR, He YQ, Mash EA, Halpert JR. 2,2',3,3',6,6'-Hexachlorobiphenyl hydroxylation by active site mutants of cytochrome P450 2B1 and 2B11. Chem. Res. Toxicol. 1999b;12:690–699. doi: 10.1021/tx990030j. [DOI] [PubMed] [Google Scholar]
- Wang H, Wei L, Wu Y, Jia H, Jiang H, Liu Y. Induction of micronuclei and cell cycle arrest by some tri- and tetrachlorobiphenyls in mammalian cells deficient in xenobiotic-metabolizing enzymes. Environ Mol Mutagen. 2017;58:199–208. doi: 10.1002/em.22090. [DOI] [PubMed] [Google Scholar]
- Wang LQ, Lehmler HJ, Robertson LW, James MO. Polychlorobiphenylols are selective inhibitors of human phenol sulfotransferase 1A1 with 4-nitrophenol as a substrate. Chemico-biological interactions. 2006;159:235–46. doi: 10.1016/j.cbi.2005.12.004. [DOI] [PubMed] [Google Scholar]
- Wangpradit O, Teesch LM, Mariappan SV, Duffel MW, Norstrom K, Robertson LW, Luthe G. Oxidation of 4-chlorobiphenyl metabolites to electrophilic species by prostaglandin H synthase. Chem Res Toxicol. 2009;22:64–71. doi: 10.1021/tx800300t. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warner NA, Martin JW, Wong CS. Chiral polychlorinated biphenyls are biotransformed enantioselectively by mammalian cytochrome P-450 isozymes to form hydroxylated metabolites. Environ. Sci. Technol. 2009;43:114–121. doi: 10.1021/es802237u. [DOI] [PubMed] [Google Scholar]
- Wu X, Pramanik A, Duffel MW, Hrycay EG, Bandiera SM, Lehmler H-J, Kania-Korwel I. 2,2',3,3',6,6'-Hexachlorobiphenyl (PCB 136) is enantioselectively oxidized to hydroxylated metabolites by rat liver microsomes. Chem. Res. Toxicol. 2011a;24:2249–2257. doi: 10.1021/tx200360m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu X, Pramanik A, Duffel MW, Hrycay EG, Bandiera SM, Lehmler HJ, Kania-Korwel I. 2,2',3,3',6,6'-Hexachlorobiphenyl (PCB 136) is enantioselectively oxidized to hydroxylated metabolites by rat liver microsomes. Chem. Res. Toxicol. 2011b;24:2249–2257. doi: 10.1021/tx200360m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu X, Duffel M, Lehmler H-J. Oxidation of polychlorinated biphenyls by liver tissue slices from phenobarbital-preatreated mice is congener-specific and atropselective. Chem. Res. Toxicol. 2013a;26:1642–1651. doi: 10.1021/tx400229e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu X, Kania-Korwel I, Chen H, Stamou M, Dammanahalli KJ, Duffel M, Lein PJ, Lehmler H-J. Metabolism of 2,2',3,3',6,6'-hexachlorobiphenyl (PCB 136) atropisomers in tissue slices from phenobarbital or dexamethasone-induced rats is sex-dependent. Xenobiotica; the fate of foreign compounds in biological systems. 2013b;43:933–947. doi: 10.3109/00498254.2013.785626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu X, Kania-Korwel I, Chen H, Stamou M, Dammanahalli KJ, Duffel M, Lein PJ, Lehmler HJ. Metabolism of 2,2',3,3',6,6'-hexachlorobiphenyl (PCB 136) atropisomers in tissue slices from phenobarbital or dexamethasone-induced rats is sex-dependent. Xenobiotica; the fate of foreign compounds in biological systems. 2013c;43:933–947. doi: 10.3109/00498254.2013.785626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu X, Kammerer A, Lehmler H-J. Microsomal oxidation of 2,2',3,3',6,6'-hexachlorobiphenyl (PCB 136) results in species-dependent chiral signatures of the hydroxylated metabolites. Environ. Sci. Technol. 2014a;48:2436–2444. doi: 10.1021/es405433t. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu X, Kammerer A, Lehmler HJ. Microsomal oxidation of 2,2',3,3',6,6'-hexachlorobiphenyl (PCB 136) results in species-dependent chiral signatures of the hydroxylated metabolites. Environ. Sci. Technol. 2014b;48:2436–44. doi: 10.1021/es405433t. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu X, Barnhart C, Lein PJ, Lehmler HJ. Hepatic metabolism affects the atropselective disposition of 2,2',3,3',6,6'-hexachlorobiphenyl (PCB 136) in mice. Environ. Sci. Technol. 2015;49:616–625. doi: 10.1021/es504766p. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wyndham C, Safe S. In vitro metabolism of 4-chlorobiphenyl by control and induced rat liver microsomes. Biochemistry. 1978;17:208–15. doi: 10.1021/bi00595a002. [DOI] [PubMed] [Google Scholar]
- Xie W, Wang K, Robertson LW, Ludewig G. Investigation of mechanism(s) of DNA damage induced by 4-monochlorobiphenyl (PCB3) metabolites. Environ Int. 2010;36:950–61. doi: 10.1016/j.envint.2009.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang D, Kania-Korwel I, Ghogha A, Chen H, Stamou M, Bose DD, Pessah IN, Lehmler HJ, Lein PJ. PCB 136 atropselectively alters morphometric and functional parameters of neuronal connectivity in cultured rat hippocampal neurons via ryanodine receptor-dependent mechanisms. Toxicol Sci. 2014;138:379–92. doi: 10.1093/toxsci/kft334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zettner MA, Flor S, Ludewig G, Wagner J, Robertson LW, Lehmann L. Quinoid metabolites of 4-monochlorobiphenyl induce gene mutations in cultured Chinese hamster v79 cells. Toxicol Sci. 2007;100:88–98. doi: 10.1093/toxsci/kfm204. [DOI] [PubMed] [Google Scholar]
- Zhai G, Lehmler HJ, Schnoor JL. Hydroxylated metabolites of 4-monochlorobiphenyl and its metabolic pathway in whole poplar plants. Environmental science & technology. 2010a;44:3901–7. doi: 10.1021/es100230m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhai G, Lehmler HJ, Schnoor JL. Identification of hydroxylated metabolites of 3,3',4,4'-tetrachlorobiphenyl and metabolic pathway in whole poplar plants. Chemosphere. 2010b;81:523–8. doi: 10.1016/j.chemosphere.2010.07.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhai G, Hu D, Lehmler H-J, Schnoor JL. Enantioselective biotransformation of chiral PCBs in whole poplar plants. Environ. Sci. Technol. 2011a;45:2308–2316. doi: 10.1021/es1033662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhai G, Lehmler HJ, Schnoor JL. New hydroxylated metabolites of 4-monochlorobiphenyl in whole poplar plants. Chem Cent J. 2011b;5:87. doi: 10.1186/1752-153X-5-87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhai G, Lehmler HJ, Schnoor JL. Inhibition of cytochromes P450 and the hydroxylation of 4-monochlorobiphenyl in whole poplar. Environmental science & technology. 2013a;47:6829–35. doi: 10.1021/es304298m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhai G, Wu X, Lehmler HJ, Schnoor JL. Atropisomeric determination of chiral hydroxylated metabolites of polychlorinated biphenyls using HPLC-MS. Chemistry Central journal. 2013b;7:183. doi: 10.1186/1752-153X-7-183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang C, Lai Y, Jin G, Glatt H, Wei Q, Liu Y. Human CYP2E1-dependent mutagenicity of mono- and dichlorobiphenyls in Chinese hamster (V79)-derived cells. Chemosphere. 2016;144:1908–15. doi: 10.1016/j.chemosphere.2015.10.083. [DOI] [PubMed] [Google Scholar]