Abstract
Background
While survival after malignancies is traditionally reported as actuarial survival, conditional survival (CS) may be more clinically relevant by accounting for “accrued” survival time as time progresses. We sought to compare actuarial and CS among patients with gallbladder carcinoma (GBC).
Methods
A total of 312 patients who underwent curative intent surgery for GBC between 2000 and 2014 were identified using a multi-institutional database. Overall survival (OS) was estimated using the Kaplan–Meier method. CS was calculated as the probability of surviving an additional 3 years at year “x” after surgery using the formula CS3 = S(x+3)/Sx.
Results
Among all patients, the median actuarial OS was 24.8 months (IQR 13.3–88.9). While actuarial survival decreased over time, 3-year CS (CS3) increased, with CS3 at 2 years after surgery noted to be 61.8 % compared with the 5-year actuarial OS of 31.6 %. Factors associated with reduced actuarial OS were positive margin status (HR 3.61, 95 % CI 2.47–5.26), increasing tumor size (HR = 1.02, 95 % CI 1.01–1.02), higher tumor grade (HR 2.98, 95 % CI 1.47–6.04), residual disease at repeat resection (HR = 2.78, 95 % CI 1.49–3.49, p < 0.001), and lymph node metastasis (HR = 1.95, 95 % CI 1.39–2.75, all p <0.001). The calculated CS3 exceeded the actuarial survival within each high-risk patient subgroup. For example, patients with residual disease at repeat resection had an actuarial survival 23.1 % at 5 years versus a CS3 of 56.3 % in patients alive at 2 years (Δ = 33.2 %).
Conclusions
CS provides a more accurate, dynamic estimate for survival, especially among high-risk patients. CS estimates can be used to accurately predict survival and guide clinical decision making.
With an annual incidence of 2.2 per 100,000, gall-bladder cancer (GBC) represents the most common cancer of the biliary tract and the sixth most common malignancy of the gastrointestinal tract.1–4 However, there are marked variations in incidence and risk factors for GBC by gender, ethnicity and geographical region.1–5 For example, while one of the most common causes of cancer mortality in India and Chile, GBC is less frequently encountered in the United States and Europe.1–4 The relative rarity of GBC in these regions has limited the ability to collect data on large cohorts of patients to guide management; in fact, many previous reports have been single-institutional, retrospective series.5–8 Furthermore, GBC has often been categorized with other biliary tract cancers, making the applicability of the data questionable and difficult to interpret.9
Although only a subset of patients are amenable to surgery, complete surgical resection remains the best chance for cure with 5-year survival ranging from 5 to 26 %.8,10–14 Despite the adoption of a radical surgical approach, a majority of patients who undergo a potentially curative surgical resection eventually develop recurrent metastatic disease.8,10,11 Given this, reliable prognostic tools are required to aid patients and surgeons in decisions pertaining to surgery, adjuvant therapy, surveillance and palliation. Currently three prognostic classification schemes are commonly used to predict survival among patients with GBC: the Nevin staging system, the Japanese Society of Biliary Surgery (JSBS) staging system and the American Joint Committee on Cancer (AJCC) prognostic schema.15–17 While these classification tools offer important overall prognostic information, each is limited in the ability to predict long-term survival accurately. Specifically, constructed using data collected at the time of surgery/diagnosis, these prognostic schemes are unable to account for the varied prognosis among patients who have already survived for a period of time after surgery.18 To this point, previous research from our own group as well as others has demonstrated that traditional estimates for overall survival (OS) rely too heavily on static risk factors determined at the time of surgery and are therefore disproportionately influenced by patients who die shortly after surgery.18–25 Given this, conditional survival (CS), which accounts for the time a patient has already survived after surgery/diagnosis, has been proposed as a more accurate estimate of long-term survival.21–23
While the use of CS in predicting long-term survival has been assessed among patients with colorectal, bladder and pancreatic cancer, to the best of our knowledge, no previous research has reported on the use of CS among patients with GBC.18–26 Therefore, the aim of the current study was to define conditional survival among patients with GBC using a large, multi-institutional cohort of patients. In particular, we sought to assess the impact of relevant patient and disease-specific characteristics on CS among patients undergoing surgery for GBC.
METHODS
Data Sources and Patient Population
Sociodemographic and clinicopathologic data was collected for all patients undergoing surgery for GBC between January 1, 2000, and December 31, 2014 at ten academic institutions in the United States (Johns Hopkins University, Baltimore, Maryland; Emory University, Atlanta, Georgia; Stanford University, Stanford, California; University of Wisconsin, Milwaukee, Wisconsin; Ohio State University, Columbus, Ohio; Washington University, St. Louis, Missouri; Vanderbilt University, Nashville, Tennessee; New York University, New York, New York; University of Louisville, Louisville, Kentucky; Wake Forest University, Winston-Salem, North Carolina). Specifically, sociodemographic data collected included age, sex and race while clinicopathologic characteristics recorded included tumor size, AJCC-T-stage, histologic grade, presence of nodal disease, final resection margin and the presence of vascular and/or perineural invasion. Tumor size was defined using the diameter of the largest tumor within the resected specimen as per the 7th edition of the AJCC staging system. Histologic grade was defined as either well, moderate, or poorly differentiated. Using the final pathologic report, the presence of disease at the resection margin (R0: no disease at resected margin, R1: presence of disease at the resected surgical margin) and the presence of lymph node metastases was determined.
Only patients undergoing a curative intent surgery for histologically confirmed GBC were included in the final study population. To minimize potential confounding, patients who died within 30 days of their surgery were excluded from further analysis. The study was approved by the institutional review board of each participating institution.
Statistical Analysis
Categorical variables were described as whole numbers and percentages while continuous variables were reported as medians with interquartile (IQR) range. OS was calculated as the time from the date of surgery to the date of death or date of last available follow-up and estimated using the Kaplan–Meier method. The Mantel–Haenszel test was used to compare differences in OS between patient groups. Associations between OS and potential risk factors (size of primary tumor, AJCC T-stage, histologic grade, lymph node metastases, margin status, and tumor invasion and the presence of residual disease requiring repeat resection) were evaluated using Cox proportional hazards regression analyses and reported as hazard ratios (HR) with corresponding 95 % confidence intervals (95 % CI). Conditional survival was calculated as the probability of surviving an additional number of “y” years given that a patient has already survived for “x” years using the formula CS(x|y) = S(x+y)/S(x), with S(x) representing OS at x years.18 Differences in CS were compared using linear regression analyses and standardized differences (d).27, 28 All analyses were performed using SPSS 22.0 (IBM, New York). A p <0.05 was used to define statistical significance.
RESULTS
Demographic and Clinicopathologic Characteristics
A total of 312 patients were identified who met inclusion criteria. Among the entire cohort, the median age was 66 years (IQR 56–73) with two-thirds of patients being female (n = 208, 66.7 %, Supplemental Table 1). The most common race/ethnicity was white (n = 208, 71.7 %), followed by African American (n = 39, 13.4 %) and Asian (n = 20, 6.9 %). Although 93.6 % (n = 250) of patients were classified as functionally independent at the time of surgery, comorbidities were commonly noted among the study cohort. Specifically, hypertension (n = 157, 58.8 %), diabetes mellitus (n = 66, 24.7 %), and a history of heart disease (n = 32, 11.9 %) were the most commonly noted comorbidities.
The most commonly performed surgery was a radical cholecystectomy defined as the resection of segments 4b/5 of the liver with concomitant lymph node dissection (n = 240, 77.2 %); a majority of patients undergoing an open surgery (n = 285, 91.9 %). A negative microscopic margin (R0) was obtained in 85.4 % of patients (n = 264). The median tumor size was 3.0 cm (2.0–5.0) and a majority of tumors were classified as either T1 (n = 24, 8.2 %) or T2 (n = 126, 43.2 %) based on the 7th edition AJCC staging system. Similarly, most tumors were graded as either moderately (n = 141; 53.8 %) or poorly (n = 91; 34.7 %) differentiated. Lymph node metastases were noted in 122 (45.7 %) patients and lymphovascular or perineural invasion noted in 49.4 % (n = 89) and 52.8 % (n = 94) of patients, respectively.
Actuarial Overall Survival and Risk Factors for Overall Survival
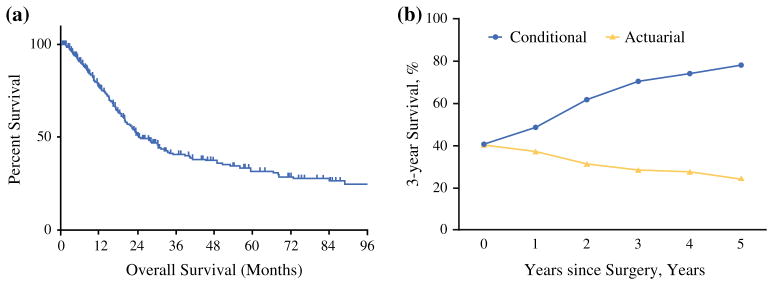
Median follow-up among all patients was 29.3 months (IQR 10.2–61.4). The median actuarial OS among all patients was 24.8 months (IQR 13.3–88.9) with 1-, 3-, and 5-year survival being 76.9, 40.7, 31.6 %, respectively. OS was noted to decrease from 76.9 % (95 % CI 71.2–81.6) 1 year after surgery to 31.6 % (95 % CI 24.9–38.6) at postoperative year 5. At 10 years after surgery, survival was only 18.4 % (95 % CI 10.9–27.4, Fig. 1a). On Cox proportional hazards analyses, a microscopically positive surgical margin (HR 3.61, 95 % CI 2.47–5.26, p < 0.001), increasing tumor size (HR 1.02; 95 % CI 1.01–1.02, p <0.001), worse tumor grade (HR 2.98, 95 % CI 1.47–6.04, p = 0.002), the presence of residual disease requiring repeat resection (HR 2.78, 95 % CI 1.49–3.49, p < 0.001), and lymph node metastases (HR 1.95, 95 % CI 1.39–2.75, p <0.001) were all associated with a worse OS.
FIG. 1.
a Actuarial overall survival for entire study cohort. b Comparison of 3-year actuarial overall survival and 3-year conditional survival for entire study cohort
Comparison of Actuarial and Conditional Survival
While the actuarial OS was noted to decrease with time, CS increased with time from the date of surgery. Specifically, the 3-year conditional survival (CS3), defined as the probability of surviving an additional 3 years, increased from 48.6 % among patients who were still alive at 1 year after surgery to 61.8 % among patients who were alive 2 years after surgery. In fact, among patients who had survived to 5 years after surgery the chance of survival to year 8 (i.e., CS3 based on 5-year survival) was 78.2 % (Fig. 1b, Table 1).
TABLE 1.
Conditional survival entire cohort
| Total survival time (y) | If the patient has survived… (%) | ||||||
|---|---|---|---|---|---|---|---|
|
| |||||||
| 1 y | 2 y | 3 y | 4 y | 5 y | 6 y | 7 y | |
| 1 | |||||||
| 2 | 66.4 | ||||||
| 3 | 52.9 | 79.6 | |||||
| 4 | 48.6 | 73.2 | 91.9 | ||||
| 5 | 41.1 | 61.8 | 77.6 | 84.5 | |||
| 6 | 37.3 | 56.2 | 70.5 | 76.7 | 90.8 | ||
| 7 | 36.0 | 54.2 | 68.1 | 74.1 | 87.7 | 96.5 | |
| 8 | 32.1 | 48.3 | 60.7 | 66.0 | 78.2 | 86.1 | 89.2 |
The effect of patient and disease-specific characteristics on CS versus OS was then assessed via subgroup analyses of patients according to prognostic factors associated with survival on Cox regression analysis Fig. 2). Factors including positive margin status, increasing tumor size, worse histologic grade, a higher tumor T stage, lymph node metastases and lymphovascular or perineural invasion, as well as the presence of residual disease at repeat resection were all associated with a worse OS. For example, 5-year actuarial survival was 38.0 % (95 % CI 10.9–27.4) among patients with a microscopically negative margin compared with 3.5 % (95 % CI 0.3–14.7) among patients with a positive surgical margin. Similarly, among patients without residual disease, 5-year actuarial survival was 46.7 % (95 % CI 32.9–59.4) compared with 23.1 % (95 % CI 12.4–35.8) among patients with residual disease (Table 2). In contrast, CS3 estimates were noted to increase with time from surgery among each patient subgroup and exceeded the OS within each strata. For example, patients with T3/T4 tumors demonstrated an actuarial survival of 9.7 % at 8 years compared with a CS3 at 5 years of 75.2 % (Δ = 65.5 %, Table 3). Similarly, patients with residual disease at repeat resection demonstrated an 8-year actuarial OS of 15.0 % compared with a CS3 at 5 years of 64.9 % (Δ = 49.9 %). Patients with lymph node metastases demonstrated an 8-year actuarial OS of 14.4 % versus a CS3 at 5 years of 65.8 % (Δ = 51.4 %). Of note, differences in CS3 and OS were less pronounced among patients within lower risk strata.
FIG. 2.
Comparison of actuarial overall survival (a, c, e, g) and 3-year conditional survival (b, d, f, h) stratified by a, b AJCC 7th edition T stage, c, d presence of lymph node metastasis, e, f final margin status, and g, h presence of residual disease at repeat resection
TABLE 2.
Overall survival stratified by risk factor
| Variable | Patient survival, %
|
p | ||
|---|---|---|---|---|
| 2 y | 5 y | 8 y | ||
| All patients | 51.1 | 31.6 | 24.7 | |
| Age | ||||
| <65 y | 53.6 | 37.5 | 27.0 | |
| ≥65 y | 49.0 | 26.4 | 22.7 | 0.427 |
| Sex | ||||
| Female | 49.2 | 30.5 | 21.9 | |
| Male | 54.8 | 34.0 | 31.4 | 0.642 |
| Type of resection | ||||
| Minor | 55.1 | 34.2 | 26.4 | |
| Major | 22.6 | 0.0 | 0.0 | 0.002 |
| Margin status | ||||
| R0 | 57.9 | 38.0 | 29.6 | |
| R1 | 14.0 | 3.5 | – | <0.001 |
| Tumor size | ||||
| ≤2.5 cm | 59.1 | 41.8 | 38.0 | |
| >2.5 cm | 38.4 | 21.0 | 21.0 | 0.007 |
| Grade | ||||
| Well differentiated | 83.3 | 53.2 | 53.2 | |
| Moderately differentiated | 49.0 | 31.0 | 21.3 | |
| Poorly differentiated | 34.4 | 22.4 | 17.9 | 0.002 |
| AJCC T stage | ||||
| T1–2 | 73.0 | 50.2 | 40.6 | |
| T3–4 | 26.8 | 12.9 | 9.7 | <0.001 |
| Lymph node meastases | ||||
| No | 64.0 | 41.6 | 34.6 | |
| Yes | 37.8 | 21.9 | 14.4 | <0.001 |
| Lymphovascular invasion | ||||
| No | 67.6 | 34.0 | 30.2 | |
| Yes | 29.5 | 15.9 | 12.7 | <0.001 |
| Perineural invasion | ||||
| No | 68.2 | 41.4 | 37.6 | |
| Yes | 35.8 | 15.3 | 11.5 | <0.001 |
| Incidentally discovered | ||||
| No | 31.0 | 23.5 | 20.9 | |
| Yes | 61.3 | 35.3 | 26.2 | 0.001 |
| Residual disease repeat resection | ||||
| No | 75.3 | 46.7 | 38.5 | |
| Yes | 41.0 | 23.1 | 15.0 | <0.001 |
TABLE 3.
Three-year conditional survival stratified by risk factor
| Variable | Time elapsed since operative resection | |||||
|---|---|---|---|---|---|---|
|
| ||||||
| 0 y | 1 y | 2 y | 3 y | 4 y | 5 y | |
| All patients | 40.7 | 48.6 | 61.8 | 70.5 | 74.1 | 78.2 |
| Type of resection | ||||||
| Minor | 43.9 | 52.4 | 62.1 | 70.6 | 73.0 | 77.2 |
| Major | 18.1 | 18.9 | 59.7 | 74.6 | – | – |
| D | 0.58 | 0.75 | 0.05 | −0.09 | – | – |
| Margin status | ||||||
| R0 | 45.4 | 51.2 | 65.6 | 75.8 | 78.6 | 77.9 |
| R1 | 14.0 | 22.1 | 25.0 | 25.0 | 33.3 | – |
| D | 0.73 | 0.63 | 0.89 | 1.18 | 1.03 | – |
| Tumor size | ||||||
| ≤2.5 cm | 51.1 | 58.7 | 70.7 | 74.4 | 78.0 | 90.9 |
| > 2.5 cm | 32.1 | 37.7 | 54.7 | 65.4 | 79.8 | – |
| D | 0.39 | 0.43 | 0.34 | 0.20 | −0.04 | – |
| Grade | ||||||
| Well differentiated | 62.0 | 70.3 | 63.9 | 85.8 | – | – |
| Moderately/poorly differentiated | 35.8 | 43.3 | 63.9 | 65.6 | 68.2 | 73.4 |
| D | 0.54 | 0.57 | 0.00 | 0.48 | – | – |
| AJCC T stage | ||||||
| T1–2 | 58.5 | 62.0 | 68.8 | 78.6 | 78.9 | 80.9 |
| T3–4 | 19.1 | 26.8 | 48.1 | 67.5 | 77.7 | 75.2 |
| D | 0.88 | 0.76 | 0.43 | 0.25 | 0.03 | 0.14 |
| Lymph node meastases | ||||||
| No | 54.5 | 60.5 | 65.0 | 76.3 | 74.9 | 83.2 |
| Yes | 26.9 | 37.2 | 57.9 | 61.0 | 56.5 | 65.8 |
| D | 0.59 | 0.48 | 0.15 | 0.34 | 0.39 | 0.41 |
| Lymphovascular invasion | ||||||
| No | 52.0 | 47.1 | 50.3 | 58.1 | 73.8 | 88.8 |
| Yes | 22.7 | 37.9 | 53.9 | 55.9 | 55.9 | 79.9 |
| D | 0.64 | 0.19 | −0.07 | 0.04 | 0.38 | 0.25 |
| Perineural invasion | ||||||
| No | 57.3 | 61.6 | 60.3 | 65.6 | 72.9 | 90.8 |
| Yes | 26.2 | 31.9 | 42.7 | 43.9 | 54.8 | 75.2 |
| D | 0.66 | 0.62 | 0.36 | 0.45 | 0.38 | 0.43 |
| Incidentally discovere | ||||||
| No | 29.6 | 42.4 | 75.8 | 70.6 | 75.5 | 88.9 |
| Yes | 46.0 | 50.5 | 57.6 | 70.0 | 73.0 | 74.2 |
| D | −0.34 | −0.16 | 0.39 | 0.01 | 0.06 | 0.39 |
| Residual disease repeat resection | ||||||
| No | 59.0 | 61.4 | 62.0 | 79.2 | 84.9 | 82.4 |
| Yes | 30.6 | 38.2 | 56.3 | 58.8 | 53.0 | 64.9 |
| D | 0.60 | 0.48 | 0.12 | 0.45 | 0.73 | 0.41 |
DISCUSSION
Complete surgical resection remains the only option for cure for patients with GBC; however, long-term survival can be poor.1–4 Given the relative rarity of GBC in the United States and Europe, there are relatively few data reporting on long-term survival after surgery for GBC. In addition, most reports on GBC utilize data from single-institutional, retrospective series that often categorize GBC with other biliary tract cancers, thereby limiting the applicability of the data.5–7 The current study is important in that it reports on long-term survival among a large cohort of patients undergoing surgery for GBC at 10 academic centers across the United States. Using Cox proportional hazards regression analysis we were able to identify risk factors associated with a worse OS including advanced T-stage, the presence of nodal disease and a microscopically positive surgical margin. Perhaps of greater importance, we assessed the use of conditional survival to estimate long-term survival among a cohort of 312 patients who underwent resection and noted that survival increased among the subset of patients who survived varied periods of time from surgery. Furthermore, marked differences in conditional and actuarial survival were also observed when patients were categorized based on observed high risk factors. Interestingly, while conditional survival was consistently higher than actuarial survival among all patient subgroups, the effect was most pronounced among patients in those subgroups characterized by the initial highest risk of long-term death.
For patients undergoing curative intent resection of GBC, the median actuarial survival was only 24.8 months. In addition, several patient and disease characteristics were strongly associated with worse long-term prognosis. Specifically, microscopically positive margins, increasing tumor size, a worse tumor grade, the presence of residual disease, as well as the presence of lymph node metastases were all associated with a worse OS. Similar to data in the current study, Duffy and colleagues reported a median actuarial survival of 30.3 months in a single-institutional review of patients with GBC.7 Factors associated with a decreased survival also included microscopically positive margins on final histopathology, as well as residual disease at the time of repeat resection.7 Similarly, de Aretxabala et al. also noted a worse actuarial survival among patients undergoing surgical resection for tumors classified as AJCC 7th edition T3/T4 disease.5, 29 Collectively, results from the current study and previous reports highlight the ability of using certain risk factors to classify patients into different prognostic subgroups thereby aiding in patient and provider decision making.
Currently, traditional risk factors have been incorporated into three existing prognostic schemes to risk-stratify patients undergoing surgery for GBC: the Nevin staging system, the Japanese Society of Biliary Surgery prognostic scheme and the AJCC staging system.15–17 These staging systems, however, may be limited because each one relies exclusively on data collected at the time of surgery and therefore cannot account for survival time accrued after surgery. In contrast, estimates for CS define survival probability given the precondition of having already survived a certain length of time. To this point, in the current study, we sought to investigate the use of CS in predicting long-term survival after surgery for GBC. In contrast to actuarial survival, which was noted to decrease with time, estimates for CS improved as the lifetime accrued from the date of surgery increased. In fact, upon stratified analyses, the difference between predicted actuarial survival and CS was greater among that strata of patients who had tumors characterized by traditionally poor risk factors. For example, among patients with T3/T4 tumors, 8-year actuarial survival was only 9.7 % compared with a CS3 at 5 years of 75.2 %. Similarly, patients with nodal metastasis demonstrated an 8-year actuarial survival of 14.4 % versus a CS3 at 5 years of 65.8 %. In effect, these data suggest that among patients who had T3/T4 or nodal disease who survived to 5 years, that these patients had a markedly better chance of living an additional 3 years than what would have been predicted at the time of surgery (Δ65.5 and Δ51.4 %, respectively). These results are consistent with previous reports from our own group, as well as others, highlighting the potential for inaccurately estimating prognosis among patients who have survived a period of time after surgery.21–26 In fact, the greatest increase in CS—and therefore difference from actuarial survival—was among patients who had the highest initial risk of death suggesting that as the time from surgery increases, the prognostic impact/importance of certain risk factors collected at the time of surgery decreases.19 Under these circumstances, the use of CS, which accounts for life years accrued, can serve as a more valuable tool in predicting long-term survival.
Results of the current study should be interpreted with the following limitations. First, while the use of a large multi-institutional database allowed for a large sample size and therefore an adequately powered analysis, we were unable to control for the minimal differences in clinical and operative practices between the 10 centers. Despite the rigorous validation of data between and within each center, it is possible that these difference in clinical practices may have resulted in some residual confounding that remained unaccounted for. Second, given the retrospective nature of the analysis, we were unable to exclude any selection bias that may have occurred regarding the treatment of patients. However, to minimize this, we included only patients undergoing curative intent surgery as well as those who did not die within the immediate postoperative time period (30 days). Furthermore, multiple subanalyses were performed by prognostic factors to minimize the potential effect of differences in patient characteristics. Lastly, as the current study reported only on overall survival, we were unable to comment of the effect of recurrence and the potential differences in conditional and actuarial survival among patients who developed a recurrence.
In conclusion, estimates for overall survival were observed to be dynamic and increased with time from surgery. Patients presenting with factors associated with a poor prognosis demonstrated the most appreciable increase in conditional versus actuarial survival. Conditional survival can therefore be used to provide a more accurate estimate for long-term prognosis among patients who have already survived over time and may serve as an important tool to inform patients and providers regarding adjuvant therapy and surveillance as well as be important in discussions around long-term survivorship.
Supplementary Material
Footnotes
Presented in part as a poster presentation at the Annual Cancer Symposium, Society of Surgical Oncology, March 2–5, 2016, Boston, MA.
DISCLOSURE The authors declare no conflict of interest.
Electronic supplementary material The online version of this article (doi:10.1245/s10434-016-5470-2) contains supplementary material, which is available to authorized users.
References
- 1.Lazcano-Ponce EC, Miquel JF, Muñoz N, Herrero R, Ferrecio C, Wistuba II, et al. Epidemiology and molecular pathology of gallbladder cancer. CA Cancer J Clin. 2001;51:349–64. doi: 10.3322/canjclin.51.6.349. [DOI] [PubMed] [Google Scholar]
- 2.Kayahara M, Nagakawa T. Recent trends of gallbladder cancer in Japan: an analysis of 4,770 patients. Cancer. 2007;110:572–80. doi: 10.1002/cncr.22815. [DOI] [PubMed] [Google Scholar]
- 3.Donohue JH, Stewart AK, Menck HR. The National Cancer Data Base report on carcinoma of the gallbladder, 1989–1995. Cancer. 1998;83:2618–28. doi: 10.1002/(sici)1097-0142(19981215)83:12<2618::aid-cncr29>3.0.co;2-h. [DOI] [PubMed] [Google Scholar]
- 4.Müller BG, De Aretxabala X, González Domingo M. A review of recent data in the treatment of gallbladder cancer: what we know, what we do, and what should be done. Am Soc Clin Oncol Educ Book. 2014:e165–70. doi: 10.14694/EdBook_AM.2014.34.e165. [DOI] [PubMed] [Google Scholar]
- 5.De Aretxabala X, Roa I, Burgos L, Losada H, Roa JC, Mora J, et al. Gallbladder cancer: an analysis of a series of 139 patients with invasion restricted to the subserosal layer. J Gastrointest Surg. 2006;10:186–92. doi: 10.1016/j.gassur.2005.11.003. [DOI] [PubMed] [Google Scholar]
- 6.Foster JM, Hoshi H, Gibbs JF, Iyer R, Javle M, Chu Q, et al. Gallbladder cancer: defining the indications for primary radical resection and radical re-resection. Ann Surg Oncol. 2007;14:833–40. doi: 10.1245/s10434-006-9097-6. [DOI] [PubMed] [Google Scholar]
- 7.Duffy A, Capanu M, Abou-Alfa GK, Huitzil D, Jarnagin W, Fong Y, et al. Gallbladder cancer (GBC): 10-year experience at Memorial Sloan-Kettering Cancer Center (MSKCC) J Surg Oncol. 2008;98:485–9. doi: 10.1002/jso.21141. [DOI] [PubMed] [Google Scholar]
- 8.Wakabayashi H, Ishimura K, Hashimoto N, Otani T, Kondo A, Maeta H. Analysis of prognostic factors after surgery for stage III and IV gallbladder cancer. Eur J Surg Oncol. 2004;30:842–6. doi: 10.1016/j.ejso.2004.06.001. [DOI] [PubMed] [Google Scholar]
- 9.Lepage C, Capocaccia R, Hackl M, Lemmens V, Molina E, Pierannunzio D, et al. Survival in patients with primary liver cancer, gallbladder and extrahepatic biliary tract cancer and pancreatic cancer in Europe, 1999–2007: results of EUROCARE-5. Eur J Cancer. doi: 10.1016/j.ejca.2015.07.034. In press. [DOI] [PubMed] [Google Scholar]
- 10.Ishihara S, Miyakawa S, Takada T, Takasaki K, Nimura Y, Tanaka M, et al. Status of surgical treatment of biliary tract cancer. Dig Surg. 2007;24:131–6. doi: 10.1159/000101901. [DOI] [PubMed] [Google Scholar]
- 11.Shimizu H, Kimura F, Yoshidome H, Ohtsuka M, Kato A, Yoshitomi H, et al. Aggressive surgical approach for stage IV gallbladder carcinoma based on Japanese Society of Biliary Surgery classification. J Hepatobiliary Pancreat Surg. 2007;14:358–65. doi: 10.1007/s00534-006-1188-z. [DOI] [PubMed] [Google Scholar]
- 12.Kai M, Chijiiwa K, Ohuchida J, Nagano M, Hiyoshi M, Kondo K. A curative resection improves the postoperative survival rate even in patients with advanced gallbladder carcinoma. J Gastrointest Surg. 2007;11:1025–32. doi: 10.1007/s11605-007-0181-4. [DOI] [PubMed] [Google Scholar]
- 13.Choi SB, Han HJ, Kim CY, Kim WB, Song T-J, Suh SO, et al. Surgical outcomes and prognostic factors for T2 gallbladder cancer following surgical resection. J Gastrointest Surg. 2010;14:668–78. doi: 10.1007/s11605-009-1132-z. [DOI] [PubMed] [Google Scholar]
- 14.Chijiiwa K, Nakano K, Ueda J, Noshiro H, Nagai E, Yamaguchi K, et al. Surgical treatment of patients with T2 gallbladder carcinoma invading the subserosal layer. J Am Coll Surg. 2001;192:600–7. doi: 10.1016/s1072-7515(01)00814-6. [DOI] [PubMed] [Google Scholar]
- 15.Ishihara S, Horiguchi A, Miyakawa S, Endo I, Miyazaki M, Takada T. Biliary tract cancer registry in Japan from 2008 to 2013. J Hepatobiliary Pancreat Sci. doi: 10.1002/jhbp.314. In press. [DOI] [PubMed] [Google Scholar]
- 16.Nevin JE, Moran TJ, Kay S, King R. Carcinoma of the gall-bladder: staging, treatment, and prognosis. Cancer. 1976;37:141–8. doi: 10.1002/1097-0142(197601)37:1<141::aid-cncr2820370121>3.0.co;2-y. [DOI] [PubMed] [Google Scholar]
- 17.Oh TG, Chung MJ, Bang S, Park SW, Chung JB, Song SY, et al. Comparison of the sixth and seventh editions of the AJCC TNM classification for gallbladder cancer. J Gastrointest Surg. 2013;17:925–30. doi: 10.1007/s11605-012-2134-9. [DOI] [PubMed] [Google Scholar]
- 18.Zabor EC, Gonen M, Chapman PB, Panageas KS. Dynamic prognostication using conditional survival estimates. Cancer. 2013;119:3589–92. doi: 10.1002/cncr.28273. [DOI] [PubMed] [Google Scholar]
- 19.Spolverato G, Kim Y, Ejaz A, Alexandrescu S, Marques H, Aldrighetti L, et al. Conditional probability of long-term survival after liver resection for intrahepatic cholangiocarcinoma. JAMA Surg. doi: 10.1001/jamasurg.2015.0219. In press. [DOI] [PubMed] [Google Scholar]
- 20.Cucchetti A, Piscaglia F, Cescon M, Ercolani G, Terzi E, Bolondi L, et al. Conditional survival after hepatic resection for hepatocellular carcinoma in cirrhotic patients. Clin Cancer Res. 2012;18:4397–405. doi: 10.1158/1078-0432.CCR-11-2663. [DOI] [PubMed] [Google Scholar]
- 21.Kim Y, Margonis GA, Prescott JD, Tran TB, Postlewait LM, Maithel SK, et al. Curative surgical resection of adrenocortical carcinoma: determining long-term outcome based on conditional disease-free probability. Ann Surg. doi: 10.1097/SLA.0000000000001527. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nathan H, de Jong MC, Pulitano C, Ribero D, Strub J, Mentha G, et al. Conditional survival after surgical resection of colorectal liver metastasis: an international multi-institutional analysis of 949 patients. J Am Coll Surg. 2010;210:755–64. 764–6. doi: 10.1016/j.jamcollsurg.2009.12.041. [DOI] [PubMed] [Google Scholar]
- 23.Mayo SC, Nathan H, Cameron JL, Olino K, Edil BH, Herman JM, et al. Conditional survival in patients with pancreatic ductal adenocarcinoma resected with curative intent. Cancer. 2012;118:2674–81. doi: 10.1002/cncr.26553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kim Y, Ejaz A, Spolverato G, Squires MH, Poultsides G, Fields RC, et al. Conditional survival after surgical resection of gastric cancer: a multi-institutional analysis of the us gastric cancer collaborative. Ann Surg Oncol. 2015;22:557–64. doi: 10.1245/s10434-014-4116-5. [DOI] [PubMed] [Google Scholar]
- 25.Bischof DA, Kim Y, Dodson R, Jimenez MC, Behman R, Cocieru A, et al. Conditional disease-free survival after surgical resection of gastrointestinal stromal tumors: a multi-institutional analysis of 502 patients. JAMA Surg. 2015;150:299–306. doi: 10.1001/jamasurg.2014.2881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Skuladottir H, Olsen JH. Conditional survival of patients with the four major histologic subgroups of lung cancer in Denmark. J Clin Oncol. 2003;21:3035–40. doi: 10.1200/JCO.2003.04.521. [DOI] [PubMed] [Google Scholar]
- 27.Crespi CM, Smith SK, Petersen L, Zimmerman S, Ganz PA. Measuring the impact of cancer: a comparison of non-Hodgkin lymphoma and breast cancer survivors. J Cancer Surviv. 2010;4:45–58. doi: 10.1007/s11764-009-0106-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Burnand B, Kernan WN, Feinstein AR. Indexes and boundaries for “quantitative significance” in statistical decisions. J Clin Epidemiol. 1990;43:1273–84. doi: 10.1016/0895-4356(90)90093-5. [DOI] [PubMed] [Google Scholar]
- 29.De Aretxabala X, Roa I, Hepp J, Maluenda F, Mordojovich G, Leon J, et al. Early gallbladder cancer: is further treatment necessary? J Surg Oncol. 2009;100:589–93. doi: 10.1002/jso.21389. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.