Table 1.
Current developmental status of new drugs for treatment of epilepsy.
| S. no. | Name of intervention | Sponsor | Structure | Therapeutic target | Condition | Clinical development phase |
|---|---|---|---|---|---|---|
| 1 | Muscimol | National Institute of Neurological Disorders and Stroke (NINDS) | 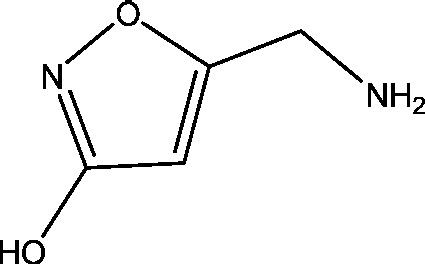 |
GABAA receptor | Epilepsy | Phase 1 |
| 2 | Bumetanide | – | 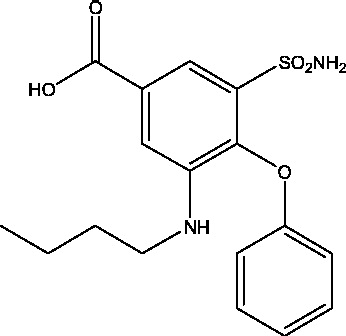 |
NKCC1 inhibition | Neonatal seizures | Phase 1 |
| 3 | BGG492 (Selurampanel) | Novartis Pharmaceuticals | 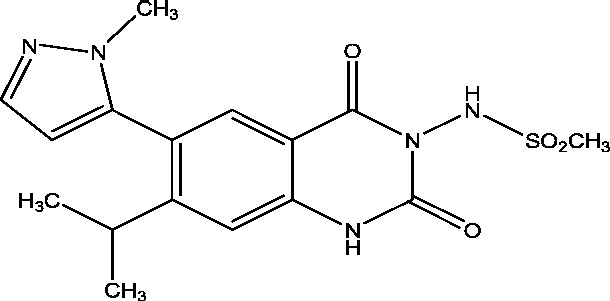 |
AMPA/kainate receptor antagonism | Refractory partial seizures | Phase 2 |
| 4 | Ganaxolone | Marinus Pharmaceuticals | 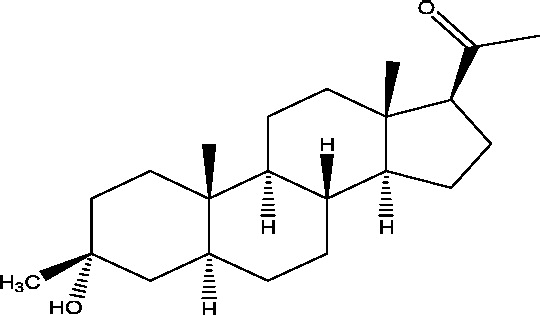 |
Positive allosteric modulator of GABAA receptors | Uncontrolled partial epilepsy; catamenial epilepsy | Phase 2 |
| 5 | Buspirone | NINDS | 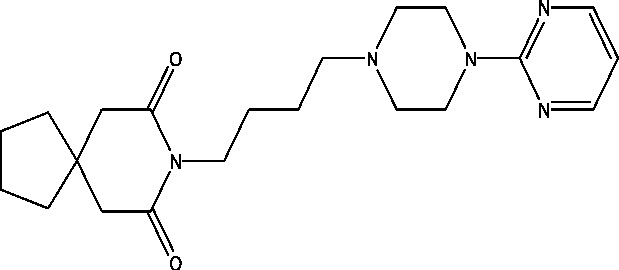 |
5-HT1A receptor partial agonist | Localized epilepsy | Phase 2 |
| 6 | YKP3089 | SK Life Sciences | 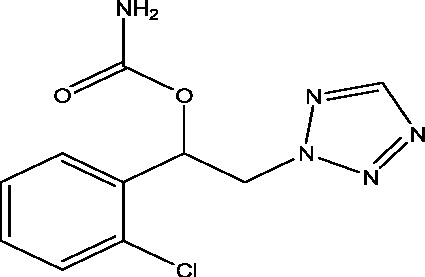 |
Sodium channel modulation, ↑ GABA release | Resistant partial onset seizures | Phase 2 |
| 7 | PRX-00023 | NINDS | 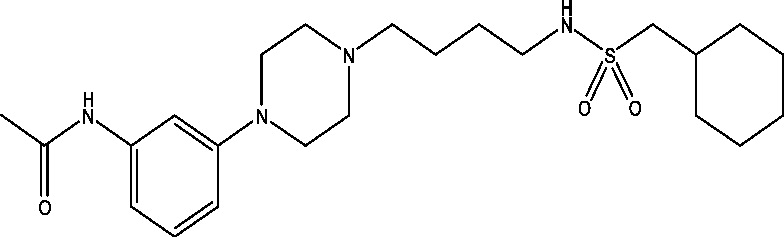 |
5-HT receptors | TLE; partial epilepsy | Phase 2 |
| 8 | GWP42003-P (Cannabidiol) | GW Research Ltd. INSYS Therapeutics Inc. |
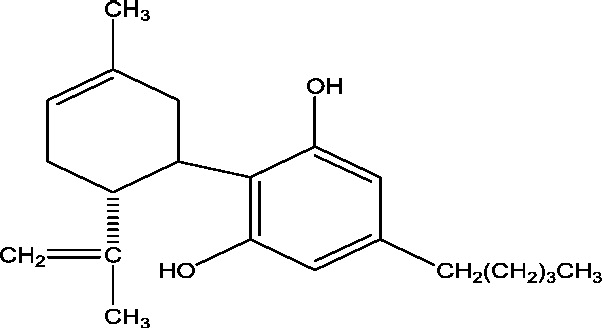 |
– | Dravet syndrome Lennox Gastaut syndrome Myoclonic epilepsy |
Phase 2 Phase 3 |
| 9 | Verapamil | Beverly S. Wical, Gillette Children's Specialty Healthcare | 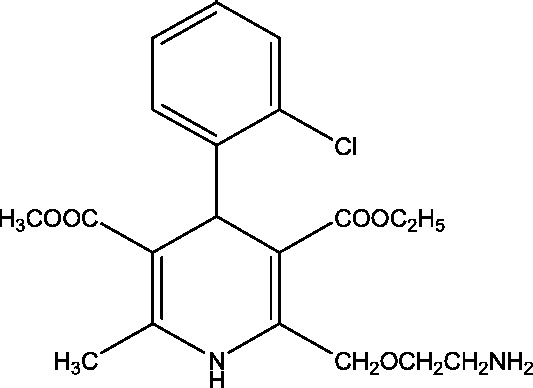 |
Calcium channel modulation | Dravet syndrome | Phase 2 |
| 10 | RWJ-333369 (carisbamate) | SK Life Science | 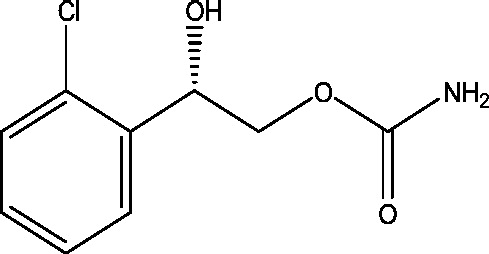 |
Not elucidated | Partial epilepsy | Phase 3 |
| 11 | Thalidomide | National Institute of Neurology and Neurosurgery, Mexico | 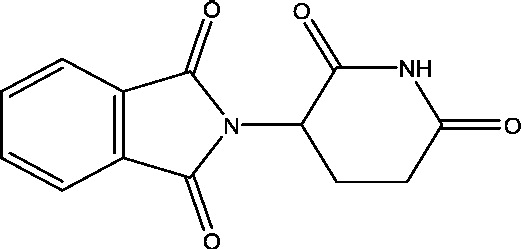 |
Refractory epilepsy | Phase 2 | |
| 12 | VX-765 | Vertex Pharmaceuticals | 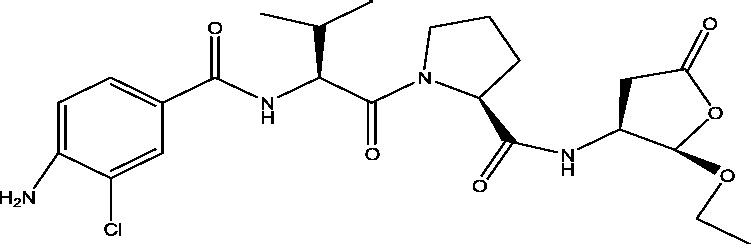 |
Caspase-1 inhibitor | Resistant partial epilepsy | Phase 2 |
| 13 | Eslicarbazepine acetate | Sunovion Pharmaceuticals Inc. Bial-Portela USA |
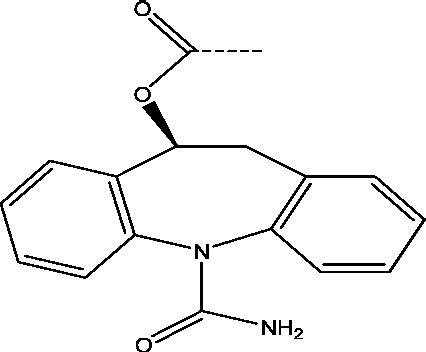 |
Sodium channel inhibition | Epilepsy with partial onset seizures Refractory partial seizures |
Phase 3 |
| 14 | Docosahexaenoic acid | Canadian College of Naturopathic Medicine (CCNM) | 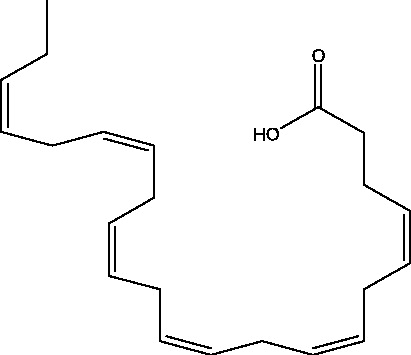 |
Refractory Seizures | Phase 3 | |
| 15 | Seletracetam (ucb 44212) | UCB, Inc. | 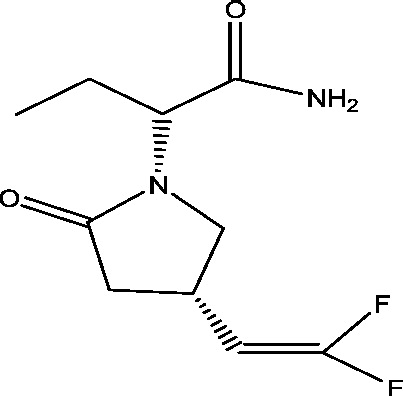 |
SV2A; presynaptic calcium channels inhibition | Partial onset seizures | Phase 3 |
| 16 | USL255 | Upsher-Smith Laboratories | 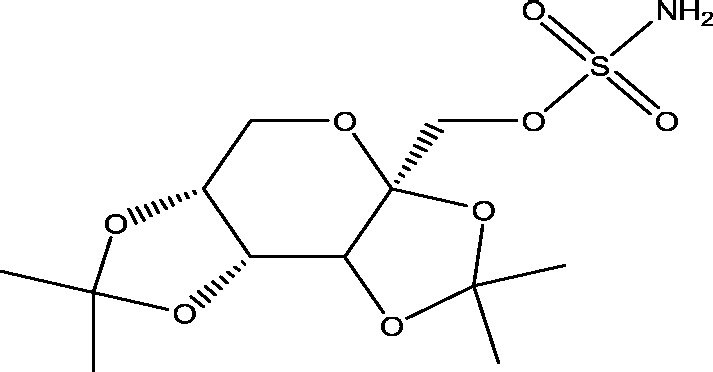 |
Sodium and calcium channels modulation | Refractory partial-onset seizures | Phase 3 |
| 17 | Brivaracetam (ucb 34714) | UCB Inc. | 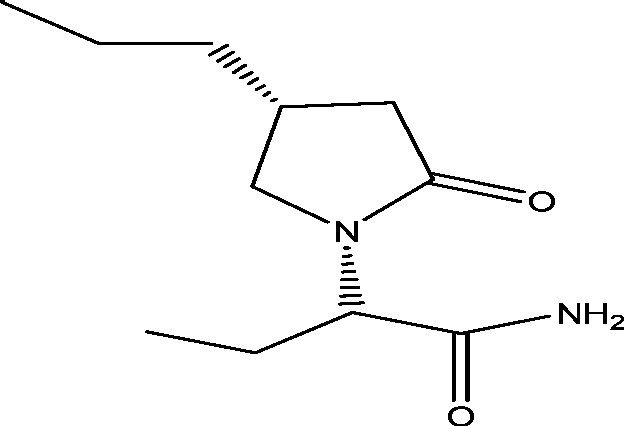 |
Same as of levetiracetam | Epilepsy | Phase 3 |
| 18 | Pregabalin | Pfizer | 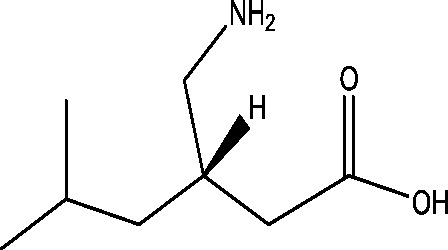 |
Calcium channel modulation | Partial onset seizures; primary GTC seizures | Phase 3 |
| 19 | RAD001 (Everolimus) | Novartis Pharmaceuticals Baylor College of Medicine New York University School of Medicine |
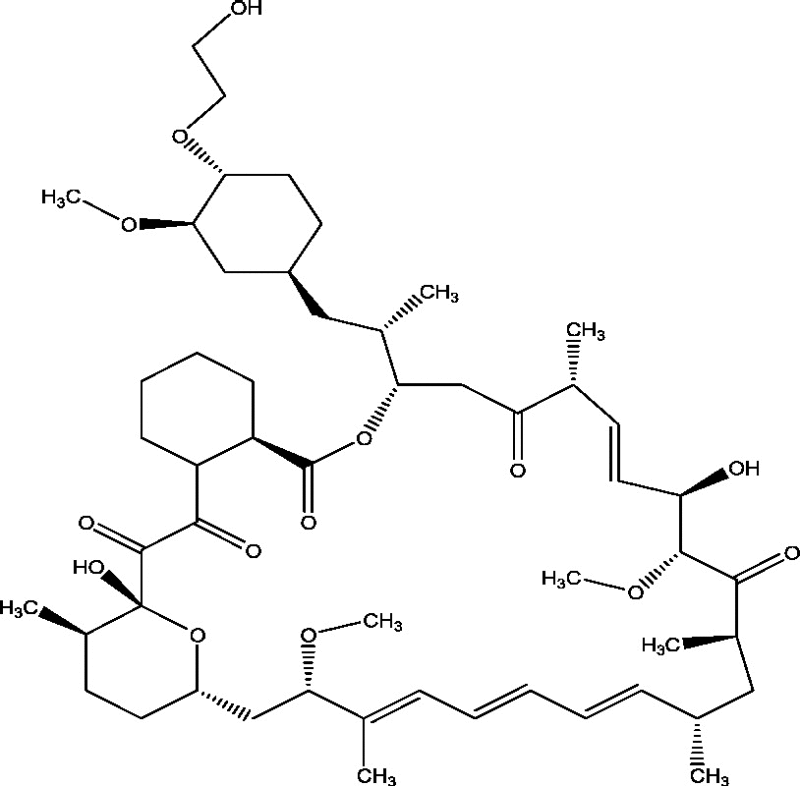 |
mTOR inhibition | Tuberous sclerosis complex-associated refractory seizures | Phase 3 |
| 20 | E2007 (Perampanel) | Eisai Inc. | 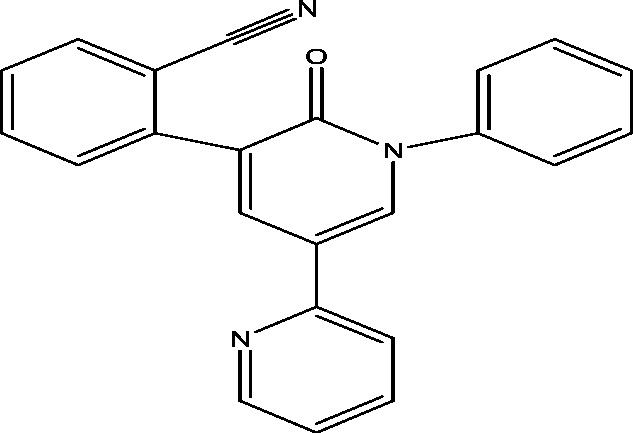 |
Non-competitive AMPA antagonism | Refractory partial seizures; primary generalized tonic clonic seizures | Phase 3 |
| 21 | TRI476 | Novartis Pharmaceuticals | 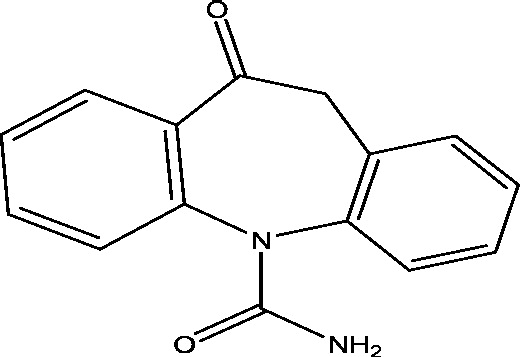 |
Sodium channel modulation | Partial onset seizures | Phase 3 |
| 22 | Retigabine/ezogabine | GlaxoSmithKline | 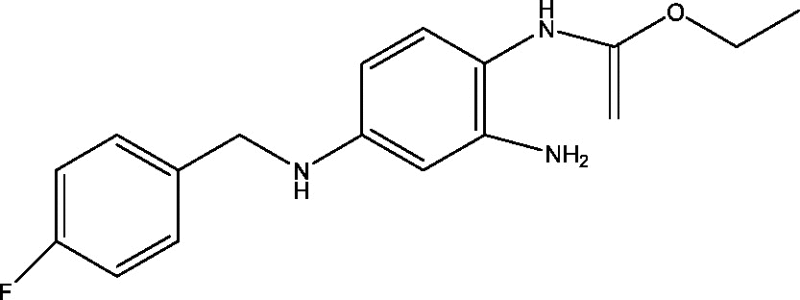 |
Potassium channel modulation | Partial onset refractory seizures | Phases 3, 4 |
