Abstract
Objectives
Whether warfarin-treated patients with non-valvular atrial fibrillation (NVAF) who exhibit good control will experience deterioration in control over time is uncertain. We designed this study to examine the time in therapeutic range (TTR) in a population-based cohort of patients with NVAF recently initiated on warfarin.
Design
Retrospective cohort study using routinely collected health data from 2008 to 2015.
Setting
The Canadian province of Alberta.
Participants
All adults with NVAF who were taking warfarin for >1 month.
Main outcome measures
Frequency of international normalised ratio (INR) monitoring and the Rosendaal TTR with time zero set at 31 days after the first warfarin dispensation.
Results
Of 57 669 patients with NVAF dispensed warfarin for >1 month, 17 099 (29.7%) had <3 INRs measured in months 1–6. Of the 40 570 who went for regular INR monitoring in months 1–6 (median number of INRs 11, IQR 7–16), 16 639 (41.0%) met the definition of good control (TTR > 65%); good control continued to be exhibited by 8177 (57.1% of those who remained on warfarin) during months 7–12 and 6804 (56.8% of continuing warfarin users) in months 13–18. Good control in the first 6 months predicted good control over the subsequent year: adjusted OR (aOR) 4.0(95%CI 3.8 to 4.2), c index 0.685(95%CI 0.679 to 0.691) for months 7–12 and aOR 3.2(95%CI 3.1 to 3.3), c index 0.665(95%CI 0.659 to 0.671) for months 13–18.
Conclusions
Nearly one-third of warfarin-treated patients had insufficient INR monitoring—this could influence the initial choice of anticoagulant and identifies a target for future quality improvement efforts. Of those warfarin-treated patients who went for regular INR monitoring, 41% exhibited levels of control similar to that in randomised trials and this deteriorated by half over time. However, in patients who have already exhibited adherence with regular monitoring and good TTR, warfarin may still be a reliable anticoagulation option.
Keywords: atrial fibrillation, warfarin, quality of care
Strengths and limitations of this study.
Addresses the question of whether patients with non-valvular atrial fibrillation (NVAF) who are well controlled on warfarin could continue to be reliably anticoagulated with warfarin.
Population-based study of all adults with NVAF in an entire Canadian province, with complete capture of all interactions with the healthcare system, prescribing data and international normalised ratio (INR) results.
We assumed the target INR ranges were 2–3 for all patients, but recognised that for a small proportion of patients with NVAF, a higher (or lower) range may be targeted clinically.
We focused solely on INR control and did not examine clinical endpoints.
Although warfarin has been shown to be efficacious in preventing stroke in non-valvular atrial fibrillation (NVAF), its effectiveness is dependent on being in therapeutic range (international normalised ratio (INR) between 2 and 3).1–6 However, some practice-based studies suggest that only a minority of patients anticoagulated in the community have an average INR between 2 and 3, with wide variability from 29% to 75%.1 3 The time in therapeutic range (TTR) is a standard measure of warfarin control which incorporates both the frequency of INR measurement and the actual values to interpolate daily INR values and define the percentage of time in range for each patient.7 A TTR of at least two-thirds is often used as the cutpoint for defining ‘good INR control’ as patients randomised to warfarin in the clinical trials proving the efficacy of anticoagulation had their INRs within target range 65% of the time,2 5 a large cohort with 63% of INRs in the 2–3 range reported warfarin benefits similar to those in the randomised trials3 and a post-hoc analysis of the Atrial fibrillation Clopidogrel Trial with Irbesartan for prevention of Vascular Events (ACTIVE-W) Trial demonstrated that warfarin-treated patients with <65% of their INRs between 2 and 3 had higher rates of embolic and bleeding events than antiplatelet-treated patients.4
One of the key arguments in favour of the direct oral anticoagulants for NVAF is that the TTR for warfarin-treated patients is unpredictable and may well be markedly lower in clinical practice than in the randomised trials proving the efficacy of warfarin. While this is certainly a rationale for choosing a direct oral anticoagulant as the first agent for a patient newly diagnosed with NVAF, as clinicians we are often faced with the issue of what to do with patients who have been well controlled on warfarin—can such patients be left on warfarin or should we be switching them to direct oral anticoagulants?
A recent report from Outcomes Registry for Better Informed Treatment of Atrial Fibrillation (ORBIT-AF) raised concern that even when patients initially exhibit good INR control, this may fluctuate substantially over time: they reported that only 34% of their patients with outstanding INR control (TTR ≥80%) in the first 6 months of observation continued to exhibit that degree of control over the subsequent year.8 However, as care varies widely across regions1 9 10 and almost 80% of ORBIT-AF patients were enrolled from specialist offices, we designed this study to examine the adequacy of anticoagulation in an entire population more closely reflecting usual clinical practice where most patients with NVAF are managed by primary care physicians. We examined the TTR and stability of INR control over time in a population-based cohort of adults with NVAF in a universal access healthcare system similar to the British National Health Service (the entire Canadian province of Alberta). As a secondary goal, we evaluated whether TTR and INR stability varied by kidney function.
Methods
Design
Retrospective cohort study using routinely collected health data.
Data sources
As described in full elsewhere,11 we used de-identified but linked (using unique health number identifiers) Alberta Health administrative and laboratory databases including all residents of Alberta (population 4.3 million people). This project was approved by Alberta Health and the Health Research Ethics Boards at the University of Alberta and the University of Calgary with a waiver of individual signed patient consent (since data was de-identified).
Study sample
The cohort consisted of all adult Albertans (aged ≥18 years) with a diagnosis of AF (International Classification of Diseases - 9 (ICD-9) Canadian Modification (CM) 427.3 or ICD-10 I48) between 1 January 2008 and 31 March 2015 in any fields of either the discharge abstract database (which captures most responsible diagnosis and up to 24 secondary diagnoses for all acute care hospitalisations), the national ambulatory care reporting system (which captures all visits to emergency rooms or hospital-based specialist clinics in Alberta), or the physician billing claims databases (see online supplementary file 1 for case definitions for NVAF and all covariates listed below). Patients with a history of mitral or aortic valvular disease, valve surgery (see online supplementary appendix 1) or end-stage kidney disease (defined as documented chronic dialysis or prior kidney transplant before the onset of NVAF) were excluded (figure 1). These NVAF case definitions have been evaluated in multiple studies, with sensitivity approaching 95% and specificity 99% in those who use both inpatient and outpatient data (as we did).12 We restricted this study to patients with dispensed warfarin prescriptions of ≥30 days (we linked to the Pharmacy Information Network and Alberta Blue Cross to obtain all prescription dispensations for cohort patients of any age). In the secondary analysis by kidney function, we restricted our analysis to only those with an outpatient serum creatinine measured at least once in the 18 months after the index date.
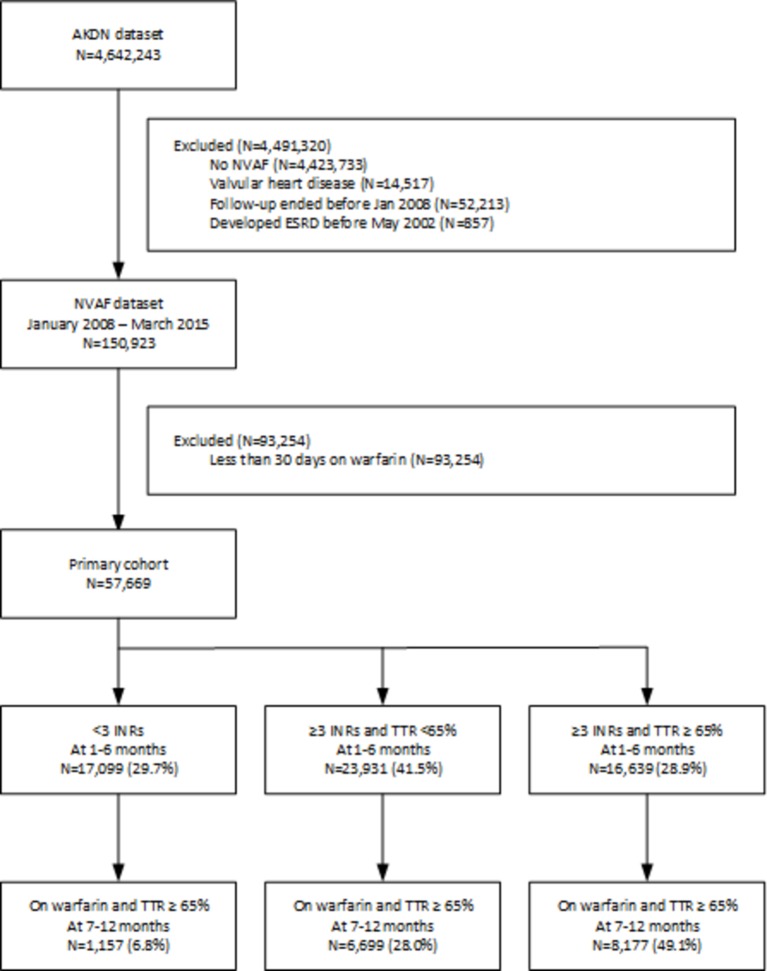
Figure 1.
Participant flow: AKDN (dataset containing administrative records for all 4.6 million Albertans) and NVAF. Note that percentages in the last row of boxes reflect the proportions among patients still prescribed warfarin in that time frame. AKDN, Alberta Kidney Disease Network; ESRD, end-stage renal disease; INR, international normalised ratio; NVAF, non-valvular atrial fibrillation; TTR, time in therapeutic range.
bmjopen-2017-016980supp001.pdf (218.3KB, pdf)
Covariates
As described fully elsewhere,11 13 14 we identified comorbidities using the ICD-9-CM or ICD-10-CA codes validated in administrative databases (with look-back beginning in April 1994) and we used estimated glomerular filtration rate (eGFR) (calculated using the Chronic Kidney Disease - Epidemiology (CKD-EPI) equation) to categorise patients by kidney function at baseline.
Definition of INR control
To examine adequacy of anticoagulation, we examined the frequency of INR monitoring and results over subsequent time frames. We excluded INRs done during the initialisation phase for warfarin (defined as within 30 days of the first warfarin prescription) and, after setting time zero as day 31, we used the outpatient INR values in months 1–6 after the initial prescription to classify patients as having ‘good control’ (TTR > 65%) or suboptimal control (TTR <65%). We calculated TTR using the method of Rosendaal, which incorporates both the frequency of INR measurement and the actual values to interpolate daily INR values and define the percentage of time in range for each patient.7 We also examined the frequency of extreme INR values (<1.5 or >4.0 as previously defined in the literature8)—in order to not falsely attribute periods during which warfarin was deliberately held for surgical or diagnostic procedures or acute illnesses, we excluded all values drawn within 1 week before or after a hospitalisation (in Alberta all biopsy procedures or surgeries are done in publically funded hospital settings and thus captured in the discharge abstract database or the national ambulatory care reporting system).
Follow-up
We followed all study participants for 18 months from the time they met the warfarin-treated NVAF case definition (ie, had been on warfarin for at least 30 days) or until they stopped warfarin, they left the province, died, or 31 March 2015, whichever came first.
Statistical analysis
All analyses were completed in Stata/MP V.13.1 (www.stata.com). Descriptive statistics were reported as counts and percentages, or medians and IQR, as appropriate. TTR is reported at 1–6, 7–12 and 13–18 months. In order to examine the association between TTR at 1–6 months with TTR at 7–12 months and 13–18 months, we used logistic regression. Outcomes were regressed on age (categorised as 65–74, 75–84 and ≥85 years), sex, rural or urban residence, eGFR (≥60, 45–59, 30–44 and <30 mL/min×1.73 m2) and comorbidities (prior myocardial infarction, prior stroke or transient ischaemic attack, alcohol misuse, metastatic cancers, non-metastatic cancers, chronic heart failure, chronic pulmonary disease, cirrhosis, dementia, diabetes mellitus, epilepsy, hypertension, peptic ulcer disease and peripheral vascular disease). McFadden’s pseudo R2 (http://stats.idre.ucla.edu/other/mult-pkg/faq/general/faq-what-are-pseudo-r-squareds/) and the likelihood ratio test were used to compare models with and without adjustment for good control in the first 6 months. p<0.05 was considered statistically significant.
Results
Of 57 669 patients with NVAF dispensed warfarin (figure 1), 17 099 (29.7%) did not have at least three INRs measured in months 1–6. Of the 40 570 who did have at least three INRs measured in months 1–6 (median number of INRs 11, IQR 7–16), 16 639 (41.0%) demonstrated good control (TTR > 65%) and 23 931 (59.0%) had suboptimal control (at least three INRs drawn but TTR <65%) (figure 1). Longer term, of the 16 639 patients who demonstrated good control in the first 6 months, 8177 (57.1% of those who remained on warfarin) exhibited TTR > 65% in months 7–12 and 6804 (56.8% of continuing warfarin users) had TTR > 65% in months 13–18 (figure 1). Of the 17 099 patients having less than three INRs measured in months 1–6 after starting warfarin, 11 653 had refills for warfarin extending beyond 7 months—as warfarin may be ingested differently than prescribed, we cannot tell exactly when (or if) the other 5446 patients with infrequent INR monitoring actually stopped taking warfarin.
Patients were more likely to have good INR control in months 1–6 if they lived in a rural area, were older or had a lower CHADS score (with lower frequencies of heart failure, stroke and diabetes but not hypertension) (tables 1 and 2). In fact, all comorbidities (except hypertension), including worsening degrees of kidney dysfunction, were associated with less likelihood of good INR control (table 2, figure 2). Despite being less likely to have TTR > 65%, patients with CHADS scores of ≥2 had a higher frequency of INR measurements (median 11 (IQR 7–17)) during months 1–6 and median 8 (IQR 6–13) in months 7–18) than patients with CHADS scores of 1 or 0 (median 9 (IQR 6–14)) during months 1–6 and median 7 (IQR 4–11) in months 7–18).
Table 1.
Baseline characteristics by time in therapeutic range in months 1–6 of warfarin use
| Overall (n=57 669) |
Time in therapeutic range <65% or infrequent INRs (n=41 030) |
Time in therapeutic range >65% (n=16 639) |
p Value | |
| Age (years) | <0.001 | |||
| 65–74 | 13 112 (22.7) | 9538 (23.2) | 3574 (21.5) | |
| 75–84 | 15 265 (26.5) | 10 663 (26) | 4602 (27.7) | |
| ≥85 | 29 292 (50.8) | 20 829 (50.8) | 8463 (50.9) | |
| Female | 25 655 (44.5) | 18 334 (44.7) | 7321 (44.0) | 0.13 |
| Rural residence | 7670 (13.3) | 5325 (13.0) | 2345 (14.1) | <0.001 |
| CHADS2 score | 2 (2,4) | 3 (2,4) | 2 (1,3) | <0.001 |
| Prior myocardial infarction | 8304 (14.4) | 6108 (14.9) | 2196 (13.2) | <0.001 |
| Prior stroke/TIA | 15 786 (27.4) | 11 350 (27.7) | 4436 (26.7) | 0.01 |
| Alcohol use disorder | 2736 (4.7) | 2268 (5.5) | 468 (2.8) | <0.001 |
| Cancer, metastatic | 1639 (2.8) | 1327 (3.2) | 312 (1.9) | <0.001 |
| Cancer, non-metastatic* | 5201 (9.0) | 3865 (9.4) | 1336 (8.0) | <0.001 |
| Chronic heart failure | 24 216 (42.0) | 18 096 (44.1) | 6120 (36.8) | <0.001 |
| Chronic pulmonary disease | 18 615 (32.3) | 13 841 (33.7) | 4774 (28.7) | <0.001 |
| Cirrhosis | 286 (0.5) | 232 (0.6) | 54 (0.3) | <0.001 |
| Dementia | 5418 (9.4) | 4228 (10.3) | 1190 (7.2) | <0.001 |
| Diabetes mellitus | 16 951 (29.4) | 12 396 (30.2) | 4555 (27.4) | <0.001 |
| eGFR (mL/min×1.73 m2) | <0.001 | |||
| ≥60 | 19 031 (55.9) | 13 390 (32.6) | 5641 (33.9) | |
| 45–59 | 7830 (23.0) | 5548 (13.5) | 2282 (13.7) | |
| 30–44 | 5035 (14.8) | 3740 (9.1) | 1295 (7.8) | |
| <30 | 2135 (6.3) | 1661 (4.0) | 474 (2.8) | |
| Epilepsy | 1455 (2.5) | 1137 (2.8) | 318 (1.9) | <0.001 |
| Hypertension | 47 534 (82.4) | 33 862 (82.5) | 13 672 (82.2) | 0.30 |
| Peptic ulcer disease | 549 (1.0) | 445 (1.1) | 104 (0.6) | <0.001 |
| Peripheral vascular disease | 3482 (6.0) | 2666 (6.5) | 816 (4.9) | <0.001 |
N (%) or median (IQR) as appropriate.
*Specifically breast, cervical, colorectal, lung and prostate.
CHADS2, congestive heart failure, hypertension, age, diabetes, stroke/transient ischaemic attack; eGFR, estimated glomerular filtration rate; TIA, transient ischaemic attack.
Table 2.
Adjusted OR associated with TTR
| 1–6 months (n=34 023) aOR (95% CI) |
7–12 months (n=42 011) aOR (95% CI) |
13–18 months (n=42 959) aOR (95% CI) |
|
| TTR ≥65%* during months 1–6 | – | 3.99 (3.81 to 4.17) | 3.19 (3.05 to 3.34) |
| Age (years) | |||
| 65–74 | 1.00 | 1.00 | 1.00 |
| 75–84 | 1.10 (1.02 to 1.18) | 1.36 (1.27 to 1.47) | 1.41 (1.31 to 1.52) |
| ≥85 | 1.10 (1.03 to 1.18) | 1.49 (1.38 to 1.59) | 1.57 (1.46 to 1.68) |
| Female | 0.91 (0.86 to 0.95) | 0.95 (0.91 to 1.00) | 0.94 (0.90 to 0.99) |
| Rural residence | 1.13 (1.05 to 1.21) | 1.09 (1.02 to 1.16) | 1.15 (1.08 to 1.23) |
| Prior myocardial infarction | 0.95 (0.88 to 1.01) | 0.93 (0.87 to 0.99) | 0.88 (0.82 to 0.94) |
| Prior stroke/TIA | 1.03 (0.97 to 1.08) | 1.03 (0.98 to 1.08) | 1.02 (0.98 to 1.08) |
| Alcohol misuse | 0.59 (0.51 to 0.67) | 0.67 (0.59 to 0.76) | 0.69 (0.61 to 0.78) |
| Cancer, metastatic | 0.61 (0.53 to 0.72) | 0.59 (0.50 to 0.69) | 0.60 (0.51 to 0.71) |
| Cancer, non-metastatic† | 0.89 (0.81 to 0.97) | 0.90 (0.82 to 0.97) | 0.88 (0.81 to 0.95) |
| Chronic heart failure | 0.81 (0.77 to 0.85) | 0.95 (0.90 to 0.99) | 0.96 (0.92 to 1.01) |
| Chronic pulmonary disease | 0.85 (0.81 to 0.90) | 0.91 (0.86 to 0.95) | 0.96 (0.91 to 1.01) |
| Cirrhosis | 0.86 (0.62 to 1.21) | 0.86 (0.61 to 1.21) | 0.60 (0.41 to 0.87) |
| Dementia | 0.72 (0.65 to 0.78) | 0.83 (0.77 to 0.90) | 0.75 (0.69 to 0.81) |
| Diabetes mellitus | 0.93 (0.89 to 0.98) | 0.95 (0.91 to 1.00) | 0.94 (0.90 to 0.99) |
| eGFR (mL/min×1.73 m2)† | |||
| ≥60 | 1.00 | 1.00 | 1.00 |
| 45–59 | 1.00 (0.94 to 1.06) | 1.01 (0.95 to 1.06) | 0.99 (0.94 to 1.05) |
| 30–44 | 0.89 (0.82 to 0.95) | 0.97 (0.90 to 1.04) | 0.92 (0.86 to 0.99) |
| <30 | 0.77 (0.69 to 0.86) | 0.78 (0.70 to 0.88) | 0.85 (0.76 to 0.95) |
| Epilepsy | 0.68 (0.57v0.80) | 0.95 (0.82 to 1.10) | 0.88 (0.76 to 1.01) |
| Hypertension | 1.06 (0.99 to 1.14) | 1.15 (1.07 to 1.23) | 1.15 (1.07 to 1.23) |
| Peptic ulcer disease | 0.69 (0.53 to 0.90) | 0.64 (0.49 to 0.84) | 0.82 (0.64 to 1.04) |
| Peripheral vascular disease | 0.82 (0.74 to 0.91) | 0.88 (0.80 to 0.96) | 0.86 (0.79 to 0.95) |
*With at least 3 INRs in months 1–6.
†specifically breast, cervical, colorectal, lung and prostate.
aOR, adjusted OR; eGFR, estimated glomerular filtration rate; TIA, transient ischaemic attack; TTR, time in therapeutic range.
Figure 2.
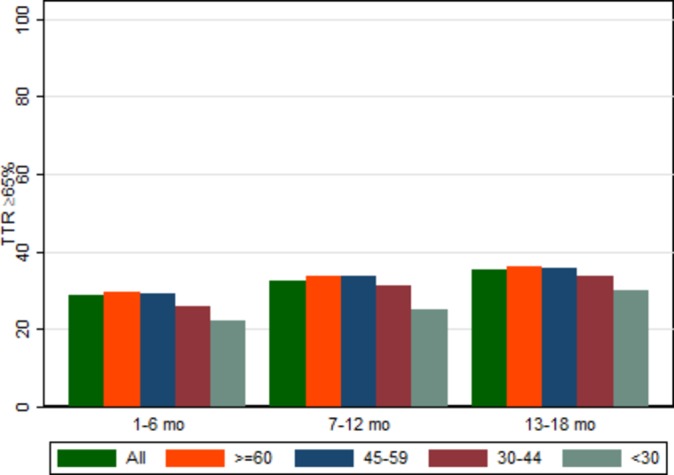
Proportion of patients with at least three INRs in months 1–6 and TTR >65% in different time frames, broken down by eGFR, INR, GFR, TTR. The height of the bars shows the percentage of participants who meet the target in each 6-month interval of follow-up. The green bars represent all participants (followed and on anticoagulants) including those without eGFR in the first 6 months. The remaining four colours represent participants with varying levels of eGFR (mL/min×1.73 m2) in the first 6 months. eGFR, estimated glomerular filtration rate; GFR, glomerular filtration rate; INR, international normalised ratio; TTR, time in therapeutic range.
Of the 16 639 patients exhibiting good INR control in the first 6 months, 8177 (57.1% of those who remained on warfarin) exhibited TTR > 65% in months 7–12 (median number of INR measurements 7, IQR 5,11) and 6804 (56.8% of continuing warfarin users) had TTR > 65% in months 13–18 (table 3). Details on the frequency of INR measurements, the proportion of out-of-range INRs and the median TTRs in each time frame are provided in table 4. We further stratify INR control by TTR 65%–80% vs >80% in figure 3;this demonstrates that the proportion of patients with excellent control (TTR >80%) actually increased over time (p<0.001 for trend). Good control in the first 6 months explained a significant amount of the variation in subsequent achievement of good control (pseudo R2 0.084, likelihood ratio test p<0.001 for months 7–12 and pseudo R2 0.064, likelihood ratio test p<0.001 for months 13–18) and exhibited reasonable discrimination for good control over the subsequent year (c index 0.685 (95% CI 0.679 to 0.691) for months 7–12 and c index 0.665 (95% CI 0.659 to 0.671) for months 13–18). Of the 14 330 patients exhibiting good INR control in 1–6 months and who continued on warfarin past 6 months, 6355 (44.3%) had at least one extreme INR value (<1.5 or>4.0) in the subsequent year. After exclusion of values drawn within 1 week of a hospitalisation, this proportion was 41.1%.
Table 3.
INR control (n=57 669)
| <3 INRs in months 1–6 (n=17 099) |
TTR <65% in months 1–6 (n=23 931) |
TTR >65% in months 1–6 (n=16 639) |
|
| 7–12 months | |||
| Still on warfarin | 11 653 (68.2) | 18 641 (77.9) | 14 330 (86.1) |
| TTR >65% | 1157 (9.9) | 6699 (35.9) | 8177 (57.1) |
| 13–18 months | |||
| Still on warfarin | 9893 (57.9) | 14 558 (60.8) | 11 987 (72.0) |
| TTR >65% | 1413 (14.3) | 5879 (40.4) | 6804 (56.8) |
INR, international normalised ratio; TTR, time in therapeutic range.
Table 4.
INR control in those with at least three INRs and TTR ≥65% in months 1–6 (n=16 639)
| Months 1–6 | Months 7–12 | Months 13–18 | |
| INR counts | 9 (3,80) | 7 (0,70) | 7 (0,84) |
| >3.0, % | 0.0 (0.0,35.0) | 5.9 (0.0,100.0) | 0.0 (0.0,100.0) |
| <2.0, % | 12.5 (0.0,35.0) | 13.0 (0.0,100.0) | 12.5 (0.0,100.0) |
| INR SD | 0.41 (0.00,3.26) | 0.46 (0.00,5.70) | 0.45 (0.00,5.66) |
| Median TTR, % (range) | 77.8 (65.0,100.0) | 66.7 (0,100.0) | 62.5 (0.0,100.0) |
INR, international normalised ratio; TTR, time in therapeutic range.
Figure 3.
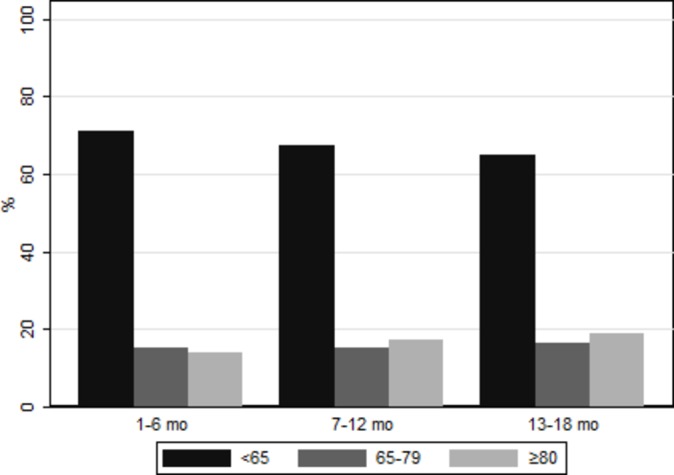
Proportion of patients with TTR in various strata over time. The height of the bars shows the percentage of all participants (followed and on anticoagulants) that fall into each ‘TTR’ interval by each 6-month interval of follow-up. The black bars show the percentage of patients that met target <65% of the time, the medium grey bars show the percentage of patients that met target between 65% and 80% of the time and the light grey bars show the percentage of patients that met target at least 80% of the time. TTR, time in therapeutic range.
Of the 41 030 patients who either had insufficient INR monitoring or exhibited suboptimal INR control during the first 6 months, 7856 (25.9% of those who remained on warfarin) met the definition of good INR control for months 7–12 and 7292 (29.8% of those who remained on warfarin during that time frame) for months 13–18 (table 3).
Discussion
We found that 30% of patients with NVAF dispensed warfarin had less than three INRs measured in months 1–6 (with approximately one-third of these patients having apparently stopped warfarin at some point in that first 6 months), and 41% of those who had regular INR measurements exhibited TTR of 65% or better. Just over half of those patients with good control in their first 6 months of warfarin therapy continued to exhibit good control over the subsequent 6 and 12 months. It is concerning that patients with higher CHADS scores or comorbidities were less likely to have INRs measured in the first 6 months and less likely to be in target range, suggesting a risk-treatment paradox in atrial fibrillation management in that higher risk patients appear to receive less optimal care.15 A similar pattern was seen in the ORBIT-AF.16 This may not necessarily reflect physician intent and may result from the fact that comorbidities such as heart failure, cirrhosis, cancer, alcohol misuse, kidney dysfunction and dementia can negatively impact TTR either through poorer medication adherence or biological variation in clotting factors (as are other factors such as malnutrition, fluctuating liver function and so on which are not captured by administrative databases).
Although there is a published prediction score for identifying patients who are more likely to have poor INR control,17 this includes factors (such as ethnicity or tobacco use) which cannot be derived from administrative data and thus it has limited utility for comparative effectiveness research. Practitioner and healthcare system factors are also predictors of suboptimal TTR ratios in the literature.9 10
Our TTR and INR stability results are similar to those reported from ORBIT-AF in the USA,8 but much lower than the levels of control reported in the Veterans Health Administration10 and a recent Swedish nationwide study.18 Although we suspect that the higher degree of INR control and better maintenance of that control over time in the Swedish and Veterans Administration (VA) studies reflects better integration and continuity of primary care in those settings, this cannot be definitively answered in observational studies such as these. However, results from a recent audit of 474 primary care physicians in Canada would support this contention as the median TTR for warfarin-treated patients with atrial fibrillation who had regular primary care physician follow-up was 75%.19
As we were able to link inpatient and outpatient administrative data, prescribing data and outpatient laboratory data to examine INR control for patients with NVAF in an entire Canadian province whether they were treated by primary care physicians or specialists, our results are generalisable to the broader population of patients with NVAF treated in a single-payer universal access healthcare system such as Alberta. Indeed, our study design avoids the potential selection biases that most AF registries are prone to. However, there are some limitations to our analysis. First, as we focused on patients newly initiated on warfarin, some may argue that this would artificially inflate their INR variability; however, we excluded inpatient and outpatient INRs drawn within the first 30 days of warfarin dispensation to limit this impact. Second, it could be argued that excluding patients who were prescribed warfarin for <30 days may have introduced a selection bias but our interest was on patients chronically using warfarin. We have no information on why some patients stopped warfarin after <30 days of therapy (in later years some may have in fact been switched to a novel direct oral anticoagulant which in the years studied was not covered publically in Alberta except with special authorisation). Third, we relied on pharmacy dispensation records to determine which patients were taking warfarin and acknowledge that warfarin may be ingested differently than prescribed or patients may be non-adherent even if filling prescriptions. Fourth, we assumed that the target INR ranges were 2–3 for all patients, but recognised that for a small proportion of patients with NVAF a higher (or lower) range may be targeted clinically if patients have had thromboembolic (or bleeding) events when INR was between 2 and 3. Fifth, we focused solely on INR control and did not examine clinical endpoints and any association with out-of-range values, although other studies have demonstrated a clear relationship between out-of-range INRs and bleeding or thromboembolic events.4–6 18 It is important to acknowledge that some patients may still have events even if well anticoagulated, which may merely reflect the expected rate of non-cardioembolic strokes in patients of the same age, sex and comorbidity profile without NVAF rather than failure of anticoagulant treatment.20
While many of the early quality improvement studies in NVAF focused on warfarin dosing algorithms,21 22 more recent studies23 have highlighted the contribution of gaps in laboratory monitoring to suboptimal warfarin management—indeed we also found that a large subset of patients did not have sufficient INR values measured to calculate TTR. Our findings support the emphasis in current quality improvement efforts24 to not only increase the use of dosing algorithms but to also encourage regular monitoring of INRs. Our findings also support those who argue for choosing a direct oral anticoagulant as the first agent for patients with NVAF given that future adherence with monitoring cannot accurately be predicted. However, our findings challenge the assumption that patients who have been well controlled on warfarin in clinical practice will invariably exhibit deteriorating control over time—one of the key arguments advanced in favour of switching from warfarin to the direct oral anticoagulants in chronically treated patients. We would agree with the authors of a recent nationwide audit from Sweden that ‘well-managed warfarin therapy…is still a valid alternative for prophylaxis of AF-associated stroke’.18
Supplementary Material
Acknowledgments
The authors of this report aregrateful to Sophanny Tiv for database development. This study is based in partby data provided by Alberta Health and Alberta Health Services. Theinterpretation and conclusions are those of the researchers and do notrepresent the views of the Government of Alberta or Alberta Health Services. Neither the Government of Alberta nor, Alberta Health or Alberta HealthServices express any opinion in relation to this study.
Footnotes
Contributors: FM: conceived the study concept and design; and wrote the first and subsequent drafts of the manuscript. NW: did the analyses. NW and BRH: involved in acquisition of the data. FM and NW: have access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. All authors: reviewed and revised the manuscript for intellectual content.
Funding: This work was supported by a peer-reviewed operating grant from the Heart and Stroke Foundation of Canada (GIA-15-0008887). FM is supported by a career salary award from Alberta Innovates-Health Solutions and holds the University of Alberta Chair in Cardiovascular Outcomes Research. The funders had no role in the design or analysis of this study, nor in the drafting or approval of this manuscript.
Disclaimer: FM affirms that the manuscript is an honest, accurate and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned (and, if relevant, registered) have been explained.
Competing interests: All authors have completed the Unified Competing Interest form (available on request from the corresponding author) and declare no relevant conflicts of interest; no financial relationships with any organisations that might have an interest in the submitted work in the previous 3 years and no other relationships or activities that could appear to have influenced the submitted work.
Patient consent: Detail has been removed from this case description/these case descriptions to ensure anonymity. The editors and reviewers have seen the detailed information available and are satisfied that the information backs up the case the authors are making.
Ethics approval: University of Alberta Health Research Ethics Board.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data sharing statement: No additional data are available as we are bound by the Health Information Act in the province of Alberta, Canada which does not permit sharing of public data beyond approved Alberta institutions.
References
- 1.Wan Y, Heneghan C, Perera R, et al. Anticoagulation control and prediction of adverse events in patients with atrial fibrillation: a systematic review. Circ Cardiovasc Qual Outcomes 2008;1:84–91. 10.1161/CIRCOUTCOMES.108.796185 [DOI] [PubMed] [Google Scholar]
- 2.Evans A, Kalra L. Are the results of randomized controlled trials on anticoagulation in patients with atrial fibrillation generalizable to clinical practice? Arch Intern Med 2001;161:1443–7. 10.1001/archinte.161.11.1443 [DOI] [PubMed] [Google Scholar]
- 3.Go AS, Hylek EM, Chang Y, et al. Anticoagulation therapy for stroke prevention in atrial fibrillation: how well do randomized trials translate into clinical practice? JAMA 2003;290:2685–92. 10.1001/jama.290.20.2685 [DOI] [PubMed] [Google Scholar]
- 4.Connolly SJ, Pogue J, Eikelboom J, et al. Benefit of oral anticoagulant over antiplatelet therapy in atrial fibrillation depends on the quality of International Normalized Ratio control achieved by centers and countries as measured by time in therapeutic range. Circulation 2008;118:2029–37. 10.1161/CIRCULATIONAHA.107.750000 [DOI] [PubMed] [Google Scholar]
- 5.White HD, Gruber M, Feyzi J, et al. Comparison of outcomes among patients randomized to warfarin therapy according to anticoagulant control: results from SPORTIF III and V. Arch Intern Med 2007;167:239–45. 10.1001/archinte.167.3.239 [DOI] [PubMed] [Google Scholar]
- 6.Veeger NJ, Piersma-Wichers M, Tijssen JG, et al. Individual time within target range in patients treated with vitamin K antagonists: main determinant of quality of anticoagulation and predictor of clinical outcome. A retrospective study of 2300 consecutive patients with venous thromboembolism. Br J Haematol 2005;128:513–9. 10.1111/j.1365-2141.2004.05348.x [DOI] [PubMed] [Google Scholar]
- 7.Rosendaal FR, Cannegieter SC, van der Meer FJM, et al. A method to determine the optimal intensity of oral anticoagulant therapy. Thromb Haemost 1993;29:236–9. [PubMed] [Google Scholar]
- 8.Pokorney SD, Simon DN, Thomas L, et al. Stability of International Normalized Ratios in Patients taking Long-term Warfarin therapy. JAMA 2016;316:661–3. 10.1001/jama.2016.9356 [DOI] [PubMed] [Google Scholar]
- 9.Singer DE, Hellkamp AS, Piccini JP, et al. Impact of global geographic region on time in therapeutic range on warfarin anticoagulant therapy: data from the ROCKET AF clinical trial. J Am Heart Assoc 2013;2:e000067 10.1161/JAHA.112.000067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rose AJ, Hylek EM, Ozonoff A, et al. Risk-adjusted percent time in therapeutic range as a quality Indicator for outpatient oral anticoagulation: results of the Veterans Affairs Study to improve anticoagulation (VARIA). Circ Cardiovasc Qual Outcomes 2011;4:22–9. 10.1161/CIRCOUTCOMES.110.957738 [DOI] [PubMed] [Google Scholar]
- 11.McAlister FA, Wiebe N, Jun M, et al. Are existing risk scores for Nonvalvular Atrial Fibrillation Useful for prediction or risk Adjustment in Patients with chronic kidney disease? Can J Cardiol 2017;33:243–52. 10.1016/j.cjca.2016.08.018 [DOI] [PubMed] [Google Scholar]
- 12.Jensen PN, Johnson K, Floyd J, et al. A systematic review of validated methods for identifying atrial fibrillation using administrative data. Pharmacoepidemiol Drug Saf 2012;21(S1):141–7. 10.1002/pds.2317 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Quan H, Sundararajan V, Halfon P, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care 2005;43:1130–9. 10.1097/01.mlr.0000182534.19832.83 [DOI] [PubMed] [Google Scholar]
- 14.Tonelli M, Wiebe N, Fortin M, et al. For the Alberta kidney disease Network. methods for identifying 30 chronic conditions: application to administrative data. BMC Med Inform Decis Mak 2015;15:31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sandhu RK, Bakal JA, Ezekowitz JA, et al. Risk stratification schemes, anticoagulation use and outcomes: the risk--treatment paradox in patients with newly diagnosed non-valvular atrial fibrillation. Heart 2011;97:2046–50. 10.1136/heartjnl-2011-300901 [DOI] [PubMed] [Google Scholar]
- 16.Pokorney SD, Simon DN, Thomas L, et al. Patients' time in therapeutic range on warfarin among US patients with atrial fibrillation: results from ORBIT-AF registry. Am Heart J 2015;170:141–8. 10.1016/j.ahj.2015.03.017 [DOI] [PubMed] [Google Scholar]
- 17.Apostolakis S, Sullivan RM, Olshansky B, et al. Factors affecting quality of anticoagulation control among patients with atrial fibrillation on warfarin: the SAMe-TT₂R₂ score. Chest 2013;144:1555–63. 10.1378/chest.13-0054 [DOI] [PubMed] [Google Scholar]
- 18.Björck F, Renlund H, Lip GY, et al. Outcomes in a Warfarin-treated population with Atrial fibrillation. JAMA Cardiol 2016;1:172–80. 10.1001/jamacardio.2016.0199 [DOI] [PubMed] [Google Scholar]
- 19.Angaran P, Dorian P, Tan MK, et al. The risk stratification and stroke prevention therapy care gap in Canadian atrial fibrillation patients. Can J Cardiol 2016;32:336–43. 10.1016/j.cjca.2015.07.012 [DOI] [PubMed] [Google Scholar]
- 20.Freedman B, Martinez C, Katholing A, et al. Residual risk of stroke and death in anticoagulant-treated patients with atrial fibrillation. JAMA Cardiol 2016;1:366–8. 10.1001/jamacardio.2016.0393 [DOI] [PubMed] [Google Scholar]
- 21.Sandhu RK, McAlister FA. Stroke prevention for patients with atrial fibrillation: improving but not perfect yet. Heart 2013;99:1141–2. 10.1136/heartjnl-2013-303656 [DOI] [PubMed] [Google Scholar]
- 22.Van Spall HGC, Wallentin L, Yusuf S, et al. Variation in Warfarin dose adjustment practice is responsible for differences in the quality of anticoagulation control between centers and countries. Circulation 2012;126:2309–16. [DOI] [PubMed] [Google Scholar]
- 23.Rose AJ, Miller DR, Ozonoff A, et al. Gaps in monitoring during oral anticoagulation: insights into care transitions, monitoring barriers, and medication nonadherence. Chest 2013;143:751–7. 10.1378/chest.12-1119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rose AJ, Park A, Gillespie C, et al. Results of a regional effort to improve Warfarin management. Ann Pharmacother 2017;51:373–9. 10.1177/1060028016681030 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjopen-2017-016980supp001.pdf (218.3KB, pdf)