Abstract
Background
The current study was designed to test the hypothesis that motivational deficits in schizophrenia (SZ) are tied to a reduced ability to differentially signal gains and instances of loss-avoidance in the brain, leading to reduced ability to form adaptive representations of expected value.
Methods
We administered a reinforcement learning paradigm to 27 medicated SZ patients and 27 controls, in which participants learned three probabilistic discriminations. In regions of interest in reward networks identified a priori, we examined contrasts between trial-types with different expected values (Expected Gain – Non-monetary, e.g.), and between outcomes with the same prediction error valence but different experienced values (Gain – Loss-avoidance outcome, Miss – Loss outcome, e.g.).
Results
Both whole-brain and regions-of-interest analyses revealed that schizophrenia patients showed reduced differentiation between Gain and Loss-avoidance outcomes in dorsal anterior cingulate cortex and bilateral anterior insula. That is, SZ patients showed reduced contrasts between positive prediction errors of different objective values in these areas. Furthermore, we observed significant correlations between [Gain – Loss-avoidance outcome] contrasts in ventral striatum and ratings for Avolition/Anhedonia, and between [Expected Gain – Non-monetary] contrasts in ventral striatum and ventromedial prefrontal cortex.
Conclusions
These results provide further evidence for intact prediction error signaling in medicated schizophrenia patients, especially with regard to loss-avoidance. By contrast components of frontostriatal circuits appear to show reduced sensitivity to the absolute valence of expected and experienced outcomes, suggesting a mechanism by which motivational deficits may emerge.
Keywords: ventral striatum, anterior insula, avolition, reinforcement learning, fMRI, reward
Introduction
Considerable evidence ties the negative symptoms of schizophrenia (SZ) – especially deficits in motivation – to dysfunction in neural circuits for reward processing and reinforcement learning (RL; 1–4). Reinforcement learning is known to depend upon multiple component processes, including the signaling of the expected value of stimuli and actions, the integration of outcomes, and the signaling of mismatches between expected and experienced outcomes, called reward prediction errors (RPEs). It is thought that a variety of brain regions participate in representations of the subjective value of stimuli and rewards, as well as the use of these representations in the guidance of choice (5–7) and the updating of these value representations, following the integration of outcomes. Areas implicated in the online representation of value include: ventromedial prefrontal cortex (vmPFC; also medial orbitofrontal cortex, or mOFC) and associated ventral regions of the striatum (VS); dorsal Anterior Cingulate Cortex (dACC; closely associated with the representation of the value of actions; 8, 9, 10); and anterior insular (AI) cortex (closely associated with the representation of aversive outcomes; 11). While the signaling of RPEs has been most closely associated with the activity of dopamine neurons in the midbrain, projecting to subfields of the striatum (12–14), performance feedback has also been shown to activate or deactivate all of the PFC regions just described (vmPFC, dACC, AI; 14, 15, 16).
Frontostriatal abnormalities could contribute to motivational deficits in SZ through multiple mechanisms. For example, motivational deficits could emerge in SZ if abnormal signaling of RPEs led to maladaptive updating of value representations. Results of studies of RPE signaling in chronic, medicated SZ patients have been mixed, however, with RPE signal strength in the striatum sometimes, but not always, correlating with measures of negative symptoms like avolition and anhedonia (17, 18). By contrast, multiple studies have linked motivational deficits in SZ to abnormal signaling of expected value – as measured by neural activity associated with outcome anticipation (rather than receipt) in striatum and vmPFC (1). Furthermore, computational modeling has shown how a system with intact signaling of RPEs, could fail to adaptively represent the values of choices (19, 20). In such a regime, individuals would assign the same subjective value to all positive RPEs, regardless of whether they had been obtained from an actual gain, or merely the avoidance of an expected loss (19).
In a study using a probabilistic RL paradigm adapted from Pessiglione et al. (13), we found this pattern in a subgroup of SZ patients – those with the most severe negative symptoms (19). We observed that SZ patients with more severe negative symptoms were more likely to show equal preference for stimuli predictive of gains (i.e., positive expected value), when paired with those predictive of loss-avoidance (even when these stimuli had sometimes yielded a loss and hence had negative expected value), suggesting that SZ patients with motivational deficits exhibited a primary deficit in signaling positive expected value rather than in learning from RPEs per se (19). Furthermore, in the context of some experimental paradigms, SZ patients have exhibited greater-than-normal loss-avoidance behavior (21, 22). Thus, a possible alternative explanation for the similar valuation of monetary gains and instances of loss-avoidance in SZ patients is that individuals with SZ actually show enhanced neural activity associated with instances of loss-avoidance (relative to instances of monetary gain, monetary loss, and reward omission).
The purpose of the current study was to test the hypothesis that motivational deficits in SZ are tied to a reduced ability to differentially signal gains and instances of loss-avoidance in the brain, leading to reduced ability to form representations of expected value that map onto the objective value of choices. We predicted that more severe motivational deficits in SZ patients would be associated with reduced ability to differentially signal gains and instances of loss-avoidance in multiple frontostriatal regions (VS, vmPFC, dACC, and AI), possibly due to both attenuated signals associated with expected gains and enhanced neural activity associated with instances of loss-avoidance.
Methods and Materials
General Procedures
Twenty-nine SZ patients and 28 healthy volunteers, matched on demographic characteristics and smoking status, successfully completed an RL task in the MRI scanner (see Supplementary Methods for details of screening procedures). All patients except one were medicated with antipsychotic drugs, with all those medicated being on stable antipsychotic medication regimens (no changes for four weeks). Outside of the MRI scanner, cognitive function was assessed using three standard measures: the Wechsler Abbreviated Scale of Intelligence (WASI; 23), the Wechsler Test of Adult Reading (WTAR; 24), and the MATRICS battery (25). Standard symptom ratings were obtained for all patients using the Scale for the Assessment of Negative Symptoms (SANS; 26), the Brief Psychiatric Rating Scale (BPRS; 27), and the Calgary Depression Scale (CDS; 28).
Acquisition of behavioral and neuroimaging data
Participants performed 240 trials (average length: 6 s) of a version of the Gain vs. Loss-avoidance probabilistic RL task similar to that used in our prior work (19; Table 1). Functional MRI data were acquired simultaneous with task performance, in four EPI scanning runs, each lasting ≈7 minutes (with lead time) and involving the acquisition of 191 whole-brain functional EPI images (for measurement of T2*-weighted BOLD effects) using a 3-T Siemens Trio scanner (Erlangen, Germany; 81 2-mm axial slices; 128 × 128 matrix; FOV = 22 × 22 cm; TR = 2 s; see Supplementary Methods for additional scanning parameters). Also, in each session, we acquired a whole-brain T1-weighted structural image (MPRAGE) for anatomical reference (1-mm3 isotropic voxels). Two patients were removed from subsequent analyses due to head motion sufficient to produce phase-instability in the functional images (resulting in the loss of entire runs to artifact). One control was excluded from subsequent analyses, due to a lack of task-engagement (evidenced by random button-pressing and no attempt to learn).
Table 1.
Demographic Information for Participant Groups.
|
|
|
|
|
|---|---|---|---|
| Patients (N=27) | Controls (N=27) | Sig. of Group Diff. | |
|
|
|
|
|
| Demographics | |||
| Age | 38.1 (11.9) | 38.3 (12.6) | p = 0.835 |
| Gender | 10 F, 17 M | 9 F, 18 M | p = 0.776 |
| Race | 19 W, 8 NW | 17 W, 10 NW | p = 0.564 |
| Subject Education (years) | 13.2 (2.2) | 15.0 (2.0) | p = 0.007 |
| Parental Education (years) | 14.1 (2.5) | 14.6 (2.7) | p = 0.619 |
| Neuropsychological Testing | |||
| IQ (from WASI 2-subtest) | 104.1 (14.9) | 118.8 (8.8) | p < 0.001 |
| WTAR Scaled Score | 105.9 (20.2) | 114.2 (7.8) | p = 0.052 |
| WRAT4 Scaled Score | 103.3 (18.9) | 113.2 (10.6) | p = 0.021 |
| MATRICS Composite | 37.6 (13.7) | 54.4 (6.2) | p < 0.001 |
| Symptom Ratings | |||
| Mean BPRS Item Score | 1.53 (0.35) | ||
| Mean BPRS Psychosis Item Score | 1.64 (0.79) | ||
| Mean SANS Item Score | 1.49 (0.75) | ||
| Mean SANS Avol/Anhed Item Score | 1.90 (1.00) | ||
Abbreviations: WASI, Wechsler Abbreviated Scale of Intelligence; WTAR, Wechsler Test of Adult Reading; WRAT4, Wide-ranging Achievement Test, Reading Subtest; BPRS, Brief Psychiatric Rating Scale; SANS, Scale for the Assessment of Negative Symptoms; Avol/Anhed, Avolition/Anhedonia subscales.
Analysis of Behavioral and Characterizing Data
Groups did not differ in age, gender, race, and parental education (Table 1). As expected, significant between-group differences were observed for participant educational attainment and multiple measures of overall intellectual functioning. Behavioral data were analyzed for the proportion of trials on which participants chose the optimal (more-frequently-reinforced) stimulus from a pair, as well as “win-stay” and “lose-shift” rates (see Supplementary Methods for details). Finally, we used a computational model, similar to those previously used by our group (29), in order to estimate action values and prediction errors on a trial-wise basis. Comparison of multiple models revealed that a Q-learning model with separate learning rates for positive and negative RPEs fit the behavioral data the best (see Supplementary Materials for details).
Analyses of Event-related fMRI Data
Single-subject Analyses
We performed two sets of regression analyses (general linear models, or GLMs) of single-subject voxel-wise time series, one with binary regressors corresponding to the six different trial types (three different pairs x two possible outcomes), and one using parametric regressors (see Supplementary Methods for details). Binary regressors were 3-second boxcar functions, time-locked to the onset of individual trials and convolved with a model hemodynamic response function. Parametric regressors were derived from the results of computational modeling of individual behavior, which allowed for the estimation of prediction errors on a trial-wise basis.
Whole-brain Group-level Analyses
To test for significant activations within groups, as well as for significant between-group differences in BOLD signal activations associated with parametric RPE regressors, we used whole-brain t-tests (AFNI 3dttest++ command). To test for significant within-group effects of outcome valence, we used whole-brain paired t-tests (AFNI 3dttest++ command). To test for significant between-group differences in BOLD signal contrasts with regard to experienced value and expected value, we used whole-brain multivariate models (AFNI 3dMVM command), with factors of group and value (6 levels of experienced value, 3 levels of expected value). For whole-brain analyses, we used a voxel-wise threshold of p=0.001 and a cluster-size threshold of 95 voxels, determined by Monte Carlo simulations.
Analyses of event-related neural responses in regions-of-interest (ROIs)
Based on the prior literature, we looked for effects of expected value valence, RPE valence, and outcome valence in VS (bilaterally), vmPFC/mOFC, dorsal anterior cingulate cortex (dACC), right anterior insula (R AI), and left anterior insula (L AI; see Figure 2A–C for specific coordinates of ROIs; see Supplementary Materials for justification of coordinates chosen). For the VS, we centered spheres of 5-mm radius on (±10, 8, −4) to form a single, bilateral ROI. All ROIs in PFC consisted of spheres of 10-mm radius.
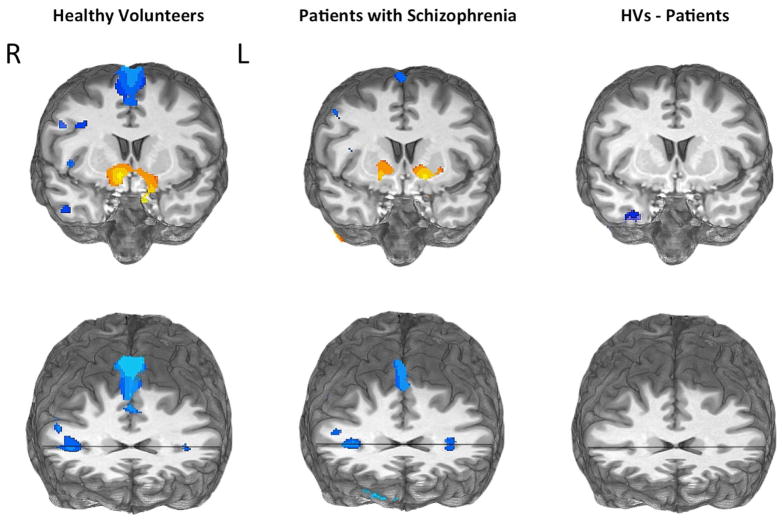
Figure 2.
Whole-brain analyses of RPE-evoked MRI activity based on trial-by-trial estimates produced by RL modeling. Brain viewed from the front. Top row cut at y=10, to display striatal activations in both healthy volunteers and patients. Bottom row cut at y=19 and z=10, to display deactivations in dorsomedial prefrontal cortex and anterior insula. Whole-brain analyses revealed no between-group differences in these regions (Table S4).
Our primary focus was on contrasts between outcomes of different objective values for the same RPE valence (e.g., Gain vs. Loss-avoidance, Miss vs. Loss). Exploratory analyses examined contrasts between expected outcomes, corresponding to condition (e.g., Gain/Miss vs. Loss/Loss-avoidance). For each of these contrasts, we performed the following tests: 1) a repeated-measures ANOVA, within the control group, with ROI as a factor, in order to determine if the contrast differed from zero in the control group, without respect to ROI, and if the contrast differed as a function of ROI; 2) a repeated-measures ANOVA, across the entire sample, with group and ROI as factors, in order to determine if there was a between-group difference in the contrast, without respect to ROI, or a significant group x ROI interaction; 3) Spearman correlation analyses, between MRI contrasts in ROIs and RL performance measures in controls; and 4) Spearman correlation analyses, between MRI contrasts in ROIs and measures of motivational deficits in patients. In order to quantify motivational deficits in patients, we computed an avolition/anhedonia factor score by averaging item scores from the avolition/role-functioning and anhedonia/asociality subscales of the SANS. In order to determine whether measures of motivational deficits in patients were related specifically to negative symptoms, rather than measures in multiple symptom classes, as well as cognitive domains, we also performed Spearman correlation analyses, between MRI contrasts in ROIs and 1) individual psychosis scores, by averaging ratings from individual psychosis items on the BPRS (Suspiciousness, Grandiosity, Hallucinations, and Unusual Thought Content), 2) individual IQ estimates, as well as several measures of intellectual function from the MATRICS battery, and 3) standardized APD doses for SZ patients (see Supplementary Materials for conversions and results of correlation analyses).
Results
Behavioral data
As shown in Figure S1, patients with schizophrenia performed worse overall than controls on contingency acquisition (for main effect of GROUP, F1,52=7.053, p=0.010). Performance in the entire sample improved across blocks (for main effect of BLOCK, F3,156=10.486, p<0.001). Neither the main effect of PAIR (F2,312=2.639, p=0.079), nor the GROUP x PAIR interaction (F2,312=1.800, p=0.172) were significant. Individuals with schizophrenia were less likely than healthy volunteers to repeat choices that resulted in optimal outcomes (69.9% win-stay rate in SZs vs. 78.4% in HVs; t52=2.371, p=0.021), and there was a trend for patients to be less likely than healthy volunteers to switch to the alternative stimulus after choices resulting in nonoptimal outcomes (34.3% lose-shift rate in SZs vs. 27.7% in HVs; t52=1.878, p=0.066). Thus, individuals with schizophrenia showed a greater tendency to switch between response alternatives, in general.
Results of RL Modeling and Model-based MRI Analyses
Details of RL modeling results are described in Supplemental Materials. As shown in Figure S1E, we observed a significant group x valence interaction in learning rate (F1,52=7.212, p=0.010), such that controls had greater learning rates for positive RPEs than negative RPEs (alphaG > alphaN; paired t26=4.447, p<0.001), whereas patients did not (t26=−0.438, p=0.665). This observation fits with the above finding of reduced win-stay rates in SZs, as well as our previous observations of attenuated positive PE-driven/Go learning in patients with SZ (especially in patients with more severe negative symptoms; 30, 31). Actual and simulated behavioral data are shown for both groups in Figure S2.
When we performed analyses of MRI BOLD signal time-courses using parametric regressors constructed from trial-wise estimates of prediction errors, we found that the signaling of reward prediction errors in the striatum was robust, with BOLD response magnitudes not differing between patients and controls (see Figure 2, Table S4). These results were consistent with previous work (17, 32) pointing to intact striatal RPE signals in chronic, medicated SZ patients. Both groups also showed strong inverse relationships between trial-wise RPE estimates and BOLD signal time-courses in left and right AI, as well as dorsomedial PFC, indicated that these areas were activated by negative RPEs and deactivated by positive RPEs (see Figure 2, Table S4). Of note: a separate whole-brain regression analysis within the patient group, using Avolition/Anhedonia scores as a between-subjects regressor, revealed no regions where Avolition/Anhedonia scores significantly modulated RPE responses, at the appropriate cluster-size threshold. Thus, whole-brain results validated the selection of ventral striatum, anterior cingulate cortex, and anterior insula, as outcome-sensitive regions, in both patients and controls.
Experienced Value Contrasts
Whole-brain Analyses
When we examined contrasts between responses to experienced gains and instances of loss-avoidance (both positive RPEs), whole-brain t-tests revealed significant effects of Outcome Valence in striatum, dACC, and right anterior insula, in controls, but significant effects of Outcome Valence in none of these regions, in patients (Figure S3, Table S5). Significant interactions between Group and Outcome Valence were observed in caudate, anterior insula, and posterior insula. Whole-brain Analyses revealed no significant effects of outcome valence on the signaling of negative RPEs within either participant group, and no between-group differences in the [MISS – LOSS] contrast.
Analyses of Variance in a priori ROIs
A repeated-measures ANOVA with factors of group and ROI revealed a significant main effect of group on [GAIN – LOSS-AVOID] contrasts [F1,52=13.002; p=0.001], but no significant main effect of ROI [F4,49=0.817; p>0.1] or group x ROI interaction [F4,49=0.634; p>0.1; Figure 3D–E, Table 2]. In controls, [GAIN – LOSS-AVOID] contrasts differed significantly from zero in all a priori ROIs, with the magnitude of the contrast in right AI correlating significantly with both win-stay rates (ρ = 0.399; p = 0.039) and learning rates for positive RPEs (alphaG; ρ = 0.403; p = 0.037; Figure 3F; Table S7). In patients, contrasts differed significantly from zero in none of the a priori ROIs, and SZ patients showed a significant reductions, relative to controls, in the contrast in dACC (t52=2.671; p=0.010) and both right (t52=3.724; p<0.001) and left AI (t52=2.537; p=0.014). These results indicate that, while neural activity in striatum and anterior insula is modulated by RPE amplitude and valence in both healthy volunteers and patients with SZ, activity in these regions is additionally modulated by the objective value of experienced positive RPEs in HVs, but to a much lesser degree in SZ patients. A repeated-measures ANOVA with factors of group and ROI revealed no significant main effect of group on [MISS – LOSS] contrasts, suggesting that patients and controls signal negative RPEs in a similar fashion (details in Supplementary Results).
Figure 3.
Regions-of-interest (ROI) analyses of effects of obtained outcome valence on positive RPE signals. A. The ventral striatum (VS) ROI consisted of two spheres of 5-mm radius, centered on (±10, 8, −4). Cut at y=8. B. The ventromedial prefrontal cortex (vmPFC) ROI consisted of a sphere of 10-mm radius, centered on 3, 32, −7), while the dorsal anterior cingulate (dACC) ROI consisted of a sphere of 10-mm radius, centered on (5, 22, 27). Brain image cut at x=4. C. The right anterior insula (RAI) ROI consisted of a sphere of 10-mm radius, centered on (32, 18, 2), while the left anterior insula (LAI) ROI consisted of a sphere of 10-mm radius, centered on (−33, 19, 3). Brain image cut at y=19. For Panels A–C, Brains viewed from the front, with right side of brain on left side of figure. D. Healthy volunteers, as a group, showed significant contrasts between gain outcomes and loss-avoidance outcomes in all a priori ROIs. E. Schizophrenia patients, as a group, showed significant contrasts between gain outcomes and loss-avoidance outcomes in none of the a priori ROIs. F. In healthy volunteers, differences in learning rates associated with positive and negative RPEs correlated significantly with [Experienced Gain – Loss-avoidance] contrasts in the right anterior insula ROI. G. In schizophrenia patients, avolition/anhedonia scores correlate significantly (inversely) with [Experienced Gain – Loss-avoidance] contrasts in the VS ROI. Error bars represent 1 standard error in either direction.
* represents paired t-test significant at p<0.05.
Table 2.
Summary of results of [Gain - Loss-avoid] Contrasts in Regions of Interest.
| [Gain - Loss-avoid] Contrast | VS | vmPFC | dACC | R AI | L AI |
|---|---|---|---|---|---|
| Different from 0 in HVs | ✔ | ✔ | ✔ | ✔ | ✔ |
| Correlates with RL performance in HVs | ✔ | ✔ | |||
| Different from 0 in SZs | |||||
| Significant between-group difference | ✔ | ✔ | ✔ | ||
| Correlates with avolition/anhedonia in SZs | ✔ |
Abbreviations: VS, ventral striatum; vmPFC, ventromedial prefrontal cortex; dACC, dorsal anterior cingulate cortex; R AI, right anterior insula; L AI, left anterior cingulate
Expected Valence Contrasts
Consistent with the results of numerous previous studies of reward anticipation (14, 33), our whole-brain analyses revealed significant effects of Expected Valence ([Positive - Negative]) in ventral striatum in controls (Figure S5). We observed no significant between-group differences in Expected Valence signaling in ventral striatum, however (Table S6). Repeated-measures ANOVAs to assess effects of group and ROI and outcome valence on neural contrasts with regard to expected valence contrasts ([Positive – Negative], [Positive – Neutral], and [Negative – Neutral]) revealed no significant effects of condition, within either group, and no significant between-group differences in any of the contrasts.
Correlation Analyses in a priori ROIs
In the patient group, scores for Avolition/Anhedonia correlated with [Gain – Loss-avoid] contrasts in VS (ρ = −0.485; p = 0.010; Table S8). While patients, as a group, showed reduced outcome-valence contrasts in multiple PFC ROIs, we observed no significant correlations between negative symptom severity and outcome-valence contrasts in any PFC ROIs. With regard to expected valence signaling, we observed a significant correlation between Avolition/Anhedonia scores in SZ patients and the positive expected value signal ([positive – neutral expected value] contrast) in VS (ρ =−0.422; p=0.028) and vmPFC (ρ =−0.390; p=0.044; Figure 4; Table S8). Finally, we observed no significant correlation between any outcome- or expected-valence contrast and positive symptom scores from the BPRS, or any standard cognitive measure (Table S9).
Analyses of correlations between neural measures and standardized antipsychotic drug dose revealed only one significant correlation: between APD dose and the [Positive – Neutral EV] contrast in dACC (Table S9). Standardized antipsychotic drug dose was also significantly correlated with the severity of positive symptoms, as measured by the BPRS (ρ =0.396, p=0.041), suggesting that patients with more severe positive symptoms were receiving higher doses of antipsychotic drugs.
Discussion
We administered a task designed to disentangle distinct aspects of value processing and to evaluate whether schizophrenia differentially affects the neural correlates of distinct aspects of value processing. We were specifically interested in determining whether patients with SZ showed elevated loss-avoidance-related activity in frontal and striatal regions, relative to gain-related activity. In fact, we found that – unlike controls - patients, as a group, showed little differentiation between gain and loss-avoidance outcomes in frontostriatal circuits (Table 2). This observation suggests that the subjective value of loss-avoidance (perhaps reflecting relief) is similar to that of monetary gain in patients (34). Furthermore, negative symptom scores in SZ patients correlated significantly with neural activity related to individual contrasts reflecting the ability to differentiate gains from instances of loss-avoidance responses in the VS, and negative symptom scores in SZ patients correlated significantly with neural activity related to expected value-related activity in VS and vmPFC/mOFC. This finding is consistent with our previous speculation that avolitional SZ patients do not show the preference for “gain” stimuli, relative to “loss-avoidance” stimuli exhibited by controls, perhaps because of insufficient top-down input from vmPFC/mOFC to VS, regarding the expectation of rewards, causing value representation in medicated SZ patients to be disproportionately influenced by learning about potentially negative consequences, as opposed to potentially positive ones (19).
Our observations that negative symptom scores correlated significantly with expected value-related activity in VS and vmPFC/mOFC and to the relative strength of loss-avoidance responses in the VS fit with results of previous studies pointing to: 1) a link between motivational deficits in schizophrenia and dysfunction in these regions (18, 35–37), and 2) greater-than normal level of loss-avoidance by SZ patients in the context of decision making under risk (21, 22). Moreover, this and previous results argue against a general impairment in prediction error signaling in SZ, especially with regard to potential losses (1, 17, 36, 38). Furthermore, the current findings provide support for our previous conjecture, based on behavioral data (19), that motivational deficits in patients with SZ are not simply the consequence of blunted RPE signaling. Indeed, both gains and loss avoidance trials represent positive prediction errors and evoke striatal responses. However, they differ in subjective value and evoke striatal responses of different magnitudes in healthy volunteers. By contrast, instances of gain and loss avoidance evoke striatal responses of similar magnitudes in schizophrenia patients. Using a similar fMRI paradigm, Reinen et al. (39) found that unmedicated psychosis patients showed attenuated RPE responses in medial prefrontal cortex, striatum, and medial temporal lobe when learning to predict rewards, but not when learning to avoid losses. The results of this and other studies (40) suggest that psychotic individuals exhibit abnormal neural signals in PFC when they are required to integrate positive outcomes in the service of updating representations of value. Our finding, in chronic SZ patients, suggests that, even if neural responses to gains are not blunted, a lack of neural differentiation between instances of gain and loss avoidance may contribute to avolition in schizophrenia, perhaps by facilitating avoidance-learning (NoGo-learning) at the expense of Go-learning (19).
Finally, our observations provide further support for overlapping, but not identical, roles for components of frontostriatal circuits in both the online representation of value (5, 18, 35, 36, 41), as well as the signaling of performance feedback (14–16). While ventral striatum has been implicated in RPE-signaling, in particular (14), it has also been implicated in the signaling of both expected (14, 33) and obtained value (14). While the vmPFC has been linked to the representation of both expected (5) and obtained value (42), it has also been described as a component of the default mode network (43). While anterior cingulate has been implicated in the representation of the expected value of actions, it has also been tied to the representation of the expected cost of actions (44, 45), as well as the resolution of response conflict (46) and the integration of negative feedback (15). Finally, the anterior insula has been linked not only to the signaling of aversive outcomes (47), but also the anticipation of aversive outcomes (11), as well as the signaling of salient outcomes, without respect to valence (48). Understanding the probable role played by nodes in frontostriatal circuits at a given time will likely depend on 1) the identification of distinct functional subregions of these larger brain areas, and 2) the characterization of network activity, as a whole, during the performing of specific aspects of reinforcement learning and decision making.
Limitations
One potential confound for the interpretation of the results of this study is the fact that all schizophrenia patients in the study, except one, were treated with antipsychotic medications. When we computed correlations between measures of brain activity and doses of antipsychotic drugs, however, we observed no significant correlations between haloperidol-equivalent antipsychotic drug dose and any of the neural signals showing systematic relationships with negative symptom ratings. Therefore, there is little indication that antipsychotic medication status drove the observed relationships between negative symptom severity and value-related neural signals. While it is likely that effective pharmacological treatment restricted the range of positive symptoms in our patients, there is little evidence that antipsychotics, as currently used (including second-generation antipsychotics), have an effect on negative symptom severity (in terms of either improvement or exacerbation; 45–47). Based on the fact that we observed a significant correlation between APD dose and residual positive symptom severity, we believe that the observed correlations between antipsychotic dose and RL-related brain responses are likely secondary to the fact that the most symptomatic patients in our study were taking the highest doses of antipsychotic medication. Thus, we are confident that our results represent an effect of illness on reward circuit functioning, and not an effect of psychotropic medication.
A second limitation of the results presented above is the fact that we tested our predictions using a large number of t-tests and correlation analyses. Many of the results identified as significant would not survive correction for multiple comparisons. However, based on our prior work, we had reason to hypothesize that motivational negative symptoms would track value-related signals in the five ROIs we selected.
A third limitation of the study is that the signals we attribute to the expectation of value might be confounded by several factors. Because our design did not include jitter (on the order of seconds) between choices and outcomes, it was not optimized to dissociate MRI activity associated with expected value from MRI activity associated with outcome processing. While our findings, in the current study, of significant correlations between avolition/anhedonia scores and expected value contrasts fit with results obtained from our previous work (36), they should be regarded with caution.
General Conclusions
In sum, we observed elevated neural activity associated with loss-avoidance feedback, relative to gain feedback, in multiple regions implicated in reward- and punishment-sensitivity, in schizophrenia patients. Furthermore, we found systematic relationships, in SZ patients, between negative symptom scores and value-related signals. We interpret these findings as evidence that motivational deficits in psychotic illness may, in part, result from reduced ability to differentially signal gains and instances of loss-avoidance in the brain, leading to compromised ability to form adaptive representations of the expected value of choices. Given the growing interest in trans-diagnostic mechanisms of apathy and avolition (52), future studies should seek to determine whether the relationships observed in this study are unique to schizophrenia, or if measures of apathy and avolition relate systematically to value-related neural signals in conditions such as mood disorders and degenerative disorders. By doing so, we may be able to uncover general neural processes underlying motivational deficits, as well as cross-diagnostic treatment targets.
Supplementary Material
Figure S1. Analyses of behavioral data from the GLA task. While both groups showed improvement across blocks, schizophrenia patients (SZs) showed worse overall performance, relative to controls (NCs). Comparisons of patient and control performance on A. Gain/Miss, B. Non-monetary, and C. Loss/Avoid pairs. D. Results of computational modeling analyses with a Gain-Loss Q-learning model revealed that patients and controls did not differ in their Beta (inverse temperature) parameters. E–F. Controls showed greater learning rates for positive prediction errors than for negative prediction errors, whereas patients did not. We observed a significant group x prediction error interaction. Error bars represent 1 standard error in either direction. * represents group x valence interaction significant at p<0.05.
Figure S2. Side-by-side comparison of behavioral and computational modeling results.
Figure S3. Whole-brain analyses of effects of obtained outcome valence on positive RPE signals, illustrating significant within-group contrasts and between-group differences in the contrasts. Whole-brain t-tests revealed significant [Experienced Gain – Loss-avoidance] contrasts in striatum, dACC, and right anterior insula, in controls, but in none of these regions, in patients (left two columns; Table S5). Significant Group and Outcome Valence interactions were observed in caudate, anterior insula, and posterior insula (right-most column). Abbreviations: R, right; L, left; A, anterior; P, posterior. Coordinates in Talairach space. Cuts as shown.
Figure S4. Regions-of-interest analyses of effects of obtained outcome valence on negative RPE signals. A repeated-measures ANOVA with factors of group and ROI revealed no main effect of group on [MISS – LOSS] contrasts, suggesting that patients and controls signal negative RPEs in a similar fashion. Coordinates of a priori ROIs as described above. Abbreviations: VS, ventral striatum; vmPFC, ventromedial prefrontal cortex; dACC, dorsal anterior cingulate cortex; RAI, right anterior insula; LAI, left anterior insula.
Figure S5. Whole-brain analyses of effects of expected outcome valence. Brains viewed from the front, cut at y=8, to display striatal activations in healthy volunteers. (Table S4).
Figure S6. Regions-of-interest analyses of MRI activity associated with manipulations of expected value in A. healthy volunteers (HVs) and B. patients with schizophrenia (SZs). Repeated-measures ANOVAs revealed no significant expected valence contrasts ([Positive – Negative], [Positive – Neutral], and [Negative – Neutral]), within either group, and no significant between-group differences in any of the contrasts. Coordinates of a priori ROIs as described above. Abbreviations: VS, ventral striatum; vmPFC, ventromedial prefrontal cortex; dACC, dorsal anterior cingulate cortex; RAI, right anterior insula; LAI, left anterior insula.
Table S1. Antipsychotic doses for individual patients, with standardized doses expressed in mg of oral haloperidol.
Table S2. Individual fitting parameters from best-fitting reinforcement learning model.
Table S3. Mean fitting parameters, by group, from three reinforcement learning models.
Table S4. Results of whole-brain analyses using parametric regressors based on trial-wise estimates of RPE.
Table S5. Results of whole-brain analyses of experienced value contrasts.
Table S6. Results of whole-brain analyses of expected value contrasts.
Table S7. Analyses of correlations between RL performance measures and MRI contrasts in ROIs in Healthy Volunteers.
Table S8. Analyses of correlations between clinical measures and MRI contrasts in ROIs in Patients with SZ. Table S9. Analyses of correlations between cognitive measures and MRI contrasts in ROIs in Patients with SZ.
Figure 1.
Trial structure and task outcome contingencies. A. On each trial, participants were presented with one of three pairs of landscapes, and had 2 s to choose either the left or right image, after which, their choice was indicated by a red border around the image. Stimuli were shown on the screen for a total of 3 seconds, with the choice highlighted, and the feedback presented centrally, for the remainder of the trial, following the response (for 3 seconds - the response time). A running tally of their total earned points was shown at the end of each trial, for the duration of the inter-trial interval. B. Trials belonged to one of three conditions: potential gain (“Gain-Miss”, or “GM”), non-monetary (“Correct-Incorrect”, or “CI”), and potential loss (“Loss-Avoid”, or “LA”). In a “Gain-Miss” pair, possible outcomes were a 25-cent gain or a neutral outcome, and thus, the expected values of those choices were positive. In a “Loss-Avoid” pair, outcomes were either neutral or a 25-cent loss, and thus, the expected values of those choices were negative. In the non-monetary pair, subjects received only verbal feedback (“Correct” or “Incorrect”), and thus, the expected values of those choices were neutral. In all three conditions, the better item was reinforced 70% of the time. For example, 70% of the time the better item was chosen on potential gain trials, the word “Win!” along with an image of a quarter was presented, while “Not a winner” was displayed 70% of the time the worse item was selected. On potential loss trials, the better choice prompted the feedback “Keep your money” 70% of the time, while choices of the worse item resulted in the image of a crossed out quarter 70% of the time.
Acknowledgments
Supported by National Institutes of Health (NIH) Grant # 5R01MH094460 (PI: Waltz). Clinical staff at the Maryland Psychiatric Research Center (MPRC) and NIDA-IRP performed screening and evaluation of research participants. Sharon August and Leeka Hubzin assisted with neuropsychological testing of research participants.
Footnotes
Financial Disclosures
Dr. Waltz has consulted for Hoffman-La Roche and NeuroCog Trials. Dr. Frank has consulted for Hoffman-La Roche. Dr. Gold has consulted for Hoffman-La Roche. The financial interests held by the authors have had no impact on the work reported. All other authors report no biomedical financial interests or potential conflicts of interest.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Waltz JA, Gold JM. Motivational Deficits in Schizophrenia and the Representation of Expected Value. Curr Top Behav Neurosci. 2016;27:375–410. doi: 10.1007/7854_2015_385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Deserno L, Boehme R, Heinz A, Schlagenhauf F. Reinforcement learning and dopamine in schizophrenia: dimensions of symptoms or specific features of a disease group? Frontiers in psychiatry. 2013;4:172. doi: 10.3389/fpsyt.2013.00172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ziauddeen H, Murray GK. The relevance of reward pathways for schizophrenia. Current opinion in psychiatry. 2010;23:91–96. doi: 10.1097/YCO.0b013e328336661b. [DOI] [PubMed] [Google Scholar]
- 4.Morris RW, Vercammen A, Lenroot R, Moore L, Langton JM, Short B, et al. Disambiguating ventral striatum fMRI-related BOLD signal during reward prediction in schizophrenia. Mol Psychiatry. 2012;17:235, 280–239. doi: 10.1038/mp.2011.75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kahnt T, Heinzle J, Park SQ, Haynes JD. The neural code of reward anticipation in human orbitofrontal cortex. Proc Natl Acad Sci U S A. 2010;107:6010–6015. doi: 10.1073/pnas.0912838107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Schoenbaum G, Roesch M. Orbitofrontal cortex, associative learning, and expectancies. Neuron. 2005;47:633–636. doi: 10.1016/j.neuron.2005.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Metereau E, Dreher JC. The medial orbitofrontal cortex encodes a general unsigned value signal during anticipation of both appetitive and aversive events. Cortex. 2015;63:42–54. doi: 10.1016/j.cortex.2014.08.012. [DOI] [PubMed] [Google Scholar]
- 8.Jocham G, Neumann J, Klein TA, Danielmeier C, Ullsperger M. Adaptive coding of action values in the human rostral cingulate zone. J Neurosci. 2009;29:7489–7496. doi: 10.1523/JNEUROSCI.0349-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rushworth MF, Behrens TE. Choice, uncertainty and value in prefrontal and cingulate cortex. Nat Neurosci. 2008;11:389–397. doi: 10.1038/nn2066. [DOI] [PubMed] [Google Scholar]
- 10.Noonan MP, Mars RB, Rushworth MF. Distinct roles of three frontal cortical areas in reward-guided behavior. J Neurosci. 2011;31:14399–14412. doi: 10.1523/JNEUROSCI.6456-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Palminteri S, Justo D, Jauffret C, Pavlicek B, Dauta A, Delmaire C, et al. Critical roles for anterior insula and dorsal striatum in punishment-based avoidance learning. Neuron. 2012;76:998–1009. doi: 10.1016/j.neuron.2012.10.017. [DOI] [PubMed] [Google Scholar]
- 12.Seymour B, O’Doherty JP, Dayan P, Koltzenburg M, Jones AK, Dolan RJ, et al. Temporal difference models describe higher-order learning in humans. Nature. 2004;429:664–667. doi: 10.1038/nature02581. [DOI] [PubMed] [Google Scholar]
- 13.Pessiglione M, Seymour B, Flandin G, Dolan RJ, Frith CD. Dopamine-dependent prediction errors underpin reward-seeking behaviour in humans. Nature. 2006;442:1042–1045. doi: 10.1038/nature05051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dillon DG, Holmes AJ, Jahn AL, Bogdan R, Wald LL, Pizzagalli DA. Dissociation of neural regions associated with anticipatory versus consummatory phases of incentive processing. Psychophysiology. 2008;45:36–49. doi: 10.1111/j.1469-8986.2007.00594.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Holroyd CB, Nieuwenhuis S, Yeung N, Nystrom L, Mars RB, Coles MG, et al. Dorsal anterior cingulate cortex shows fMRI response to internal and external error signals. Nat Neurosci. 2004;7:497–498. doi: 10.1038/nn1238. [DOI] [PubMed] [Google Scholar]
- 16.O’Doherty J, Critchley H, Deichmann R, Dolan RJ. Dissociating valence of outcome from behavioral control in human orbital and ventral prefrontal cortices. J Neurosci. 2003;23:7931–7939. doi: 10.1523/JNEUROSCI.23-21-07931.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Waltz JA, Kasanova Z, Ross TJ, Salmeron BJ, McMahon RP, Gold JM, et al. The roles of reward, default, and executive control networks in set-shifting impairments in schizophrenia. PLoS One. 2013;8:e57257. doi: 10.1371/journal.pone.0057257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dowd EC, Barch DM. Pavlovian reward prediction and receipt in schizophrenia: relationship to anhedonia. PLoS One. 2012;7:e35622. doi: 10.1371/journal.pone.0035622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gold JM, Waltz JA, Matveeva TM, Kasanova Z, Strauss GP, Herbener ES, et al. Negative symptoms and the failure to represent the expected reward value of actions: behavioral and computational modeling evidence. Arch Gen Psychiatry. 2012;69:129–138. doi: 10.1001/archgenpsychiatry.2011.1269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Collins AG, Brown JK, Gold JM, Waltz JA, Frank MJ. Working memory contributions to reinforcement learning impairments in schizophrenia. J Neurosci. 2014;34:13747–13756. doi: 10.1523/JNEUROSCI.0989-14.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cheng GL, Tang JC, Li FW, Lau EY, Lee TM. Schizophrenia and risk-taking: impaired reward but preserved punishment processing. Schizophrenia Research. 2012;136:122–127. doi: 10.1016/j.schres.2012.01.002. [DOI] [PubMed] [Google Scholar]
- 22.Brown EC, Hack SM, Gold JM, Carpenter WT, Fischer BA, Prentice KP, et al. Integrating frequency and magnitude information in decision-making in schizophrenia: An account of patient performance on the Iowa Gambling Task. J Psychiatr Res. 2015;66–67:16–23. doi: 10.1016/j.jpsychires.2015.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wechsler D. Wechsler Abbreviated Scale of Intelligence (WASI) San Antonio TX: The Psychological Corporation; 1999. [Google Scholar]
- 24.Wechsler D. Wechsler Test of Adult Reading (WTAR) San Antonio, TX: The Psychological Corporation; 2001. [Google Scholar]
- 25.Green MF, Nuechterlein KH, Gold JM, Barch DM, Cohen J, Essock S, et al. Approaching a consensus cognitive battery for clinical trials in schizophrenia: the NIMH-MATRICS conference to select cognitive domains and test criteria. Biol Psychiatry. 2004;56:301–307. doi: 10.1016/j.biopsych.2004.06.023. [DOI] [PubMed] [Google Scholar]
- 26.Andreasen NC. The Scale for the Assessment of Negative Symptoms (SANS) Iowa City, IA: University of Iowa; 1984. [Google Scholar]
- 27.Overall JE, Gorman DR. The Brief Psychiatric Rating Scale. Psychological Reports. 1962;10:799–812. [Google Scholar]
- 28.Addington D, Addington J, Maticka-Tyndale E, Joyce J. Reliability and validity of a depression rating scale for schizophrenics. Schizophr Res. 1992;6:201–208. doi: 10.1016/0920-9964(92)90003-n. [DOI] [PubMed] [Google Scholar]
- 29.Waltz JA, Demro C, Schiffman J, Thompson E, Kline E, Reeves G, et al. Reinforcement Learning Performance and Risk for Psychosis in Youth. J Nerv Ment Dis. 2015;203:919–926. doi: 10.1097/NMD.0000000000000420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Waltz JA, Frank MJ, Robinson BM, Gold JM. Selective reinforcement learning deficits in schizophrenia support predictions from computational models of striatal-cortical dysfunction. Biol Psychiatry. 2007;62:756–764. doi: 10.1016/j.biopsych.2006.09.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Waltz JA, Frank MJ, Wiecki TV, Gold JM. Altered probabilistic learning and response biases in schizophrenia: behavioral evidence and neurocomputational modeling. Neuropsychology. 2011;25:86–97. doi: 10.1037/a0020882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Culbreth AJ, Westbrook A, Xu Z, Barch DM, Waltz JA. Intact ventral striatal prediction error signaling in medicated schizophrenia patients. Biological Psychiatry: Cognitive Neuroscience and Neuroimaging. 2016;1:474–483. doi: 10.1016/j.bpsc.2016.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Knutson B, Taylor J, Kaufman M, Peterson R, Glover G. Distributed neural representation of expected value. J Neurosci. 2005;25:4806–4812. doi: 10.1523/JNEUROSCI.0642-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lefebvre G, Lebreton M, Meyniel F, Bourgeois-Gironde S, Palminteri S. Behavioural and neural characterization of optimistic reinforcement learning. Nature Human Behaviour. 2017;1:0067. [Google Scholar]
- 35.Simon JJ, Biller A, Walther S, Roesch-Ely D, Stippich C, Weisbrod M, et al. Neural correlates of reward processing in schizophrenia - Relationship to apathy and depression. Schizophr Res. 2010;118:154–161. doi: 10.1016/j.schres.2009.11.007. [DOI] [PubMed] [Google Scholar]
- 36.Waltz JA, Schweitzer JB, Ross TJ, Kurup PK, Salmeron BJ, Rose EJ, et al. Abnormal responses to monetary outcomes in cortex, but not in the basal ganglia, in schizophrenia. Neuropsychopharmacology. 2010;35:2427–2439. doi: 10.1038/npp.2010.126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Ehrlich S, Yendiki A, Greve DN, Manoch DS, Ho BC, White TL, et al. Striatal function in relation to negative symptoms in schizophrenia. Psychological Medicine. 2012;42:267–282. doi: 10.1017/S003329171100119X. [DOI] [PubMed] [Google Scholar]
- 38.Waltz JA, Schweitzer JB, Gold JM, Kurup PK, Ross TJ, Salmeron BJ, et al. Patients with schizophrenia have a reduced neural response to both unpredictable and predictable primary reinforcers. Neuropsychopharmacology. 2009;34:1567–1577. doi: 10.1038/npp.2008.214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Reinen JM, Van Snellenberg JX, Horga G, Abi-Dargham A, Daw ND, Shohamy D. Motivational Context Modulates Prediction Error Response in Schizophrenia. Schizophr Bull. 2016;42:1467–1475. doi: 10.1093/schbul/sbw045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kirschner M, Hager OM, Bischof M, Hartmann-Riemer MN, Kluge A, Seifritz E, et al. Deficits in context-dependent adaptive coding of reward in schizophrenia. NPJ Schizophr. 2016;2:16020. doi: 10.1038/npjschz.2016.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Padoa-Schioppa C. Range-adapting representation of economic value in the orbitofrontal cortex. J Neurosci. 2009;29:14004–14014. doi: 10.1523/JNEUROSCI.3751-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Knutson B, Fong GW, Adams CM, Varner JL, Hommer D. Dissociation of reward anticipation and outcome with event-related fMRI. Neuroreport. 2001;12:3683–3687. doi: 10.1097/00001756-200112040-00016. [DOI] [PubMed] [Google Scholar]
- 43.Sridharan D, Levitin DJ, Menon V. A critical role for the right fronto-insular cortex in switching between central-executive and default-mode networks. Proc Natl Acad Sci U S A. 2008;105:12569–12574. doi: 10.1073/pnas.0800005105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Prevost C, Pessiglione M, Metereau E, Clery-Melin ML, Dreher JC. Separate valuation subsystems for delay and effort decision costs. J Neurosci. 2010;30:14080–14090. doi: 10.1523/JNEUROSCI.2752-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kurniawan IT, Guitart-Masip M, Dayan P, Dolan RJ. Effort and valuation in the brain: the effects of anticipation and execution. J Neurosci. 2013;33:6160–6169. doi: 10.1523/JNEUROSCI.4777-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Minzenberg MJ, Laird AR, Thelen S, Carter CS, Glahn DC. Meta-analysis of 41 functional neuroimaging studies of executive function in schizophrenia. Arch Gen Psychiatry. 2009;66:811–822. doi: 10.1001/archgenpsychiatry.2009.91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Metereau E, Dreher JC. Cerebral correlates of salient prediction error for different rewards and punishments. Cereb Cortex. 2013;23:477–487. doi: 10.1093/cercor/bhs037. [DOI] [PubMed] [Google Scholar]
- 48.Rutledge RB, Dean M, Caplin A, Glimcher PW. Testing the reward prediction error hypothesis with an axiomatic model. J Neurosci. 2010;30:13525–13536. doi: 10.1523/JNEUROSCI.1747-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Harvey RC, James AC, Shields GE. A Systematic Review and Network Meta-Analysis to Assess the Relative Efficacy of Antipsychotics for the Treatment of Positive and Negative Symptoms in Early-Onset Schizophrenia. CNS Drugs. 2016;30:27–39. doi: 10.1007/s40263-015-0308-1. [DOI] [PubMed] [Google Scholar]
- 50.Remington G, Foussias G, Fervaha G, Agid O, Takeuchi H, Lee J, et al. Treating Negative Symptoms in Schizophrenia: an Update. Curr Treat Options Psychiatry. 2016;3:133–150. doi: 10.1007/s40501-016-0075-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fusar-Poli P, Papanastasiou E, Stahl D, Rocchetti M, Carpenter W, Shergill S, et al. Treatments of Negative Symptoms in Schizophrenia: Meta-Analysis of 168 Randomized Placebo-Controlled Trials. Schizophr Bull. 2015;41:892–899. doi: 10.1093/schbul/sbu170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Morris SE, Cuthbert BN. Research Domain Criteria: cognitive systems, neural circuits, and dimensions of behavior. Dialogues in clinical neuroscience. 2012;14:29–37. doi: 10.31887/DCNS.2012.14.1/smorris. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1. Analyses of behavioral data from the GLA task. While both groups showed improvement across blocks, schizophrenia patients (SZs) showed worse overall performance, relative to controls (NCs). Comparisons of patient and control performance on A. Gain/Miss, B. Non-monetary, and C. Loss/Avoid pairs. D. Results of computational modeling analyses with a Gain-Loss Q-learning model revealed that patients and controls did not differ in their Beta (inverse temperature) parameters. E–F. Controls showed greater learning rates for positive prediction errors than for negative prediction errors, whereas patients did not. We observed a significant group x prediction error interaction. Error bars represent 1 standard error in either direction. * represents group x valence interaction significant at p<0.05.
Figure S2. Side-by-side comparison of behavioral and computational modeling results.
Figure S3. Whole-brain analyses of effects of obtained outcome valence on positive RPE signals, illustrating significant within-group contrasts and between-group differences in the contrasts. Whole-brain t-tests revealed significant [Experienced Gain – Loss-avoidance] contrasts in striatum, dACC, and right anterior insula, in controls, but in none of these regions, in patients (left two columns; Table S5). Significant Group and Outcome Valence interactions were observed in caudate, anterior insula, and posterior insula (right-most column). Abbreviations: R, right; L, left; A, anterior; P, posterior. Coordinates in Talairach space. Cuts as shown.
Figure S4. Regions-of-interest analyses of effects of obtained outcome valence on negative RPE signals. A repeated-measures ANOVA with factors of group and ROI revealed no main effect of group on [MISS – LOSS] contrasts, suggesting that patients and controls signal negative RPEs in a similar fashion. Coordinates of a priori ROIs as described above. Abbreviations: VS, ventral striatum; vmPFC, ventromedial prefrontal cortex; dACC, dorsal anterior cingulate cortex; RAI, right anterior insula; LAI, left anterior insula.
Figure S5. Whole-brain analyses of effects of expected outcome valence. Brains viewed from the front, cut at y=8, to display striatal activations in healthy volunteers. (Table S4).
Figure S6. Regions-of-interest analyses of MRI activity associated with manipulations of expected value in A. healthy volunteers (HVs) and B. patients with schizophrenia (SZs). Repeated-measures ANOVAs revealed no significant expected valence contrasts ([Positive – Negative], [Positive – Neutral], and [Negative – Neutral]), within either group, and no significant between-group differences in any of the contrasts. Coordinates of a priori ROIs as described above. Abbreviations: VS, ventral striatum; vmPFC, ventromedial prefrontal cortex; dACC, dorsal anterior cingulate cortex; RAI, right anterior insula; LAI, left anterior insula.
Table S1. Antipsychotic doses for individual patients, with standardized doses expressed in mg of oral haloperidol.
Table S2. Individual fitting parameters from best-fitting reinforcement learning model.
Table S3. Mean fitting parameters, by group, from three reinforcement learning models.
Table S4. Results of whole-brain analyses using parametric regressors based on trial-wise estimates of RPE.
Table S5. Results of whole-brain analyses of experienced value contrasts.
Table S6. Results of whole-brain analyses of expected value contrasts.
Table S7. Analyses of correlations between RL performance measures and MRI contrasts in ROIs in Healthy Volunteers.
Table S8. Analyses of correlations between clinical measures and MRI contrasts in ROIs in Patients with SZ. Table S9. Analyses of correlations between cognitive measures and MRI contrasts in ROIs in Patients with SZ.