Abstract
Some natural compounds decrease serum levels of thyroid autoantibodies, but results are inconsistent and thyroid function has been evaluated infrequently; moreover, the effects of Aloe on thyroid autoimmunity and function have been examined in very few studies. This study stems from the observation of one co-author, who has Hashimoto’s thyroiditis (HT)-related subclinical hypothyroidism (SCH). Upon checking her biochemical thyroid panel when taking daily Aloe barbardensis Miller juice (ABMJ) for thyroid-unrelated reasons, she noticed a decrease in serum thyroperoxidase autoantibodies (TPOAb) and thyrotropin (TSH) and an increase in serum free thyroxine (FT4). Based on this observation, we enrolled 30 consecutive HT women with levothyroxine-untreated SCH and high TPOAb levels. All of them took ABMJ (50 ml daily) for nine months and were tested for serum TSH, FT4, free triiodothyronine (FT3) and TPOAb. Measurements were performed at baseline and at months 3 and 9. TSH, FT4 and TPOAb improved significantly already at month 3 and further (−61%, +23% and −56%) at month 9. However, FT3 decreased significantly at month 3 (−16%) with no further decrease at month 9, so that the FT4:FT3 ratio increased significantly (+33% and + 49%). At baseline, 100% of women had TSH > 4.0 mU/L and TPOAb > 400 U/ml, but frequencies fell to 0% and 37%, respectively, at month 9. In contrast, a control group (namely, 15 untreated SCH women of comparable age and baseline levels of TSH, FT4, FT3 and TPOAb) had no significant changes in any index. We conclude that the daily intake of 100 ml ABMJ for 9 months in women with HT-related SCH decreases the burden of thyroid autoimmune inflammation. In addition, ABMJ rescues thyrocyte function, with decreased need for conversion of the prohormone T4 into the more active T3 through ABMJ-induced inhibition of T4 deiodination.
Keywords: Aloe vera, Subclinical hypothyroidism, Thyroid autoimmunity, Thyroid function
Introduction
Aloe is a very old plant with medicinal properties that was discovered by the ancient Egyptians, who called it “the plant of immortality” [1]. The botanical name of Aloe vera is Aloe barbadensis Miller. It belongs to the Asphodelaceae (Liliaceae) family, and is a shrubby or arborescent, perennial, xerophytic, succulent, pea-green color plant. It grows mainly in the dry regions of Africa, Asia, Europe and America [1]. Records of its use were engraved in tablets thought to be from 2100 B.C. Aloe then travelled to various parts of the globe, and starting from the 17th century it had become a common medicinal plant [2]. The name Aloe vera derives from the Arabic word “Alloeh” meaning “shining bitter substance” and from the Latin word “vera” meaning “true.” Two millennia ago, Greek scientists regarded Aloe vera as the universal panacea [1]. Egyptian queens Nefertiti and Cleopatra used Aloe as part of their regular beauty regimes. Alexander the Great and Christopher Columbus used it to treat soldiers’ wounds. The first reference to Aloe vera in the English literature dates back to 1655, when John Goodyer translated the Dioscorides’ Medical treatise De Materia Medica. By the early 1800s, Aloe vera was in use as a laxative in the United States, and in the mid-1930s it was successfully employed to treat chronic and severe radiation dermatitis [1].
Aloe Barbadensis leaf juice is extracted from the leaves of the aloe plant. Aloe Barbadensis leaves contain over 200 nutritional substances, including 20 minerals (particularly iron, chromium, zinc, selenium, copper, manganese, magnesium, sodium, potassium, and calcium), 20 amino acids, a dozen vitamins (A, B1, B2, B3, B5, B6, B12, C, E, choline, and folic acid), active enzymes (alkaline phosphatase, amylase, bradykinase, carboxypeptidase, catalase, cellulase, lipase, and peroxidase), anthraquinones (mostly known for their laxative effects), sterols, lignin, saponins, salicylic acids and others [1]. Interestingly, the thyroid hormone-forming tyrosine is the rarest amino acid present in Aloe Barbadensis leaves (28 µmol/100 g), while arginine is the main one (449 µmol/100 g) [3]. The anti-oxidant and/or anti-inflammatory properties of Aloe vera are explained by its content in vitamin A, C and E, in the glycoprotein C-glucosyl chromone, in certain sterols (campesterol, β-sitosterol and lupeol), vegetal hormones (auxins and giberrelins) and bradykinase. Campesterol, β-sitosterol, lupeol and two anthraquinones (aloin and emodin) act as analgesics and antiseptics [1]. These sterols are also found in shea butter and sabal/saw palmetto (Serenoa repens) [4], [5]. Further details can be found in the Volume 1 of WHO (World Health Organization) Monographs on Selected Medicinal Plants [6]. As reviewed elsewhere [1], some uses of Aloe vera are based on scientific evidence in humans and/or animals, while other uses are based on tradition. Scientific-based therapies for Aloe vera include seborrheic dermatitis, psoriasis vulgaris, genital herpes, skin burns, wound healing, pressure ulcers, mucositis, radiation dermatitis, acne vulgaris, lichen planus, frostbite, aphthous stomatitis, type 2 diabetes mellitus, HIV infection, cancer prevention, ulcerative colitis and constipation. Traditional-based therapies for Aloe vera include alopecia, bacterial and fungal skin infections, parasitic infections, chronic leg wounds, systemic lupus erythematosus, arthritis and tic douloureux.
A number of natural compounds/nutraceuticals are being used to treat autoimmune thyroid diseases (AIT), namely Graves’ disease (GD), GD-associated ophthalmopathy, HT and postpartum thyroiditis (PPT) [7], [8], [9], [10], [11], [12], [13], [14], [15], [16], [17], [18], [19], [20], [21], [22], [23], [24], [25], [26], [27], [28], [29]. Though an exhaustive list of these substances goes beyond the scope of the present paper, examples of them include omega-3-fatty acid-rich small oily fish [7], [8], L-carnitine [9], [10], [11], selenium [12], [13], [14], [15], [16], [17], [18], [19], [20], [21], [22], [23], [24], [25], [26], [27], [28], [29], and myoinositol [27], [28], [29]. However, the observation that prompted the present study stemmed from a fortuitous observation (Table 1). One of the authors of the present paper, who has a history of HT-associated initial, mild hypothyroidism (also called subclinical hypothyroidism [SCH]), decided to take Aloe Barbadensis Miller juice (ABMJ), at the dose of 50 ml every morning on an empty stomach, as a skin soother and laxative. No other medications or over-the-counter compounds were taken. She checks thyroid function and thyroperoxidase autoantibodies [TPOAb] semiannually, since in one relative progression to overt hypothyroidism was preceded by a frank fall in FT4 and increase in both TSH and TPOAb approximately six months earlier. At the biochemical check performed three months after having started taking ABMJ, she was struck by the remarkable improvement of all indices (Table 1). The improvement was even more impressive six months later (Table 1).
Table 1.
Summary of the Aloe vera barbadensis Miller juice-induced changes in the biochemical thyroid profile of one author of the present paper and that prompted the study described herein.a
| Follow-up |
|||||
|---|---|---|---|---|---|
| Before use of Aloe juice |
During use of Aloe juice |
||||
| Range (min - max) | Last value | First value | Last value | ||
| TSH, mU/L [0.2–4.0] | 4.3–5.5 | 5.14 | 3.22 | 1.83 | |
| FT4, pmol/L [7–19] | 7.9–8.3 | 8.3 | 8.9 | 11.44 | |
| FT3, pmol/L [2.7–6.4] | 5.0–5.25 | 5.22 | 5.0 | 4.78 | |
| TPOAb, U/ml [0–35] | 1,256–1875 | 1875 | 778 | 246 | |
Follow-up prior to use of the aloe juice (50 ml twice a day) spans 14 months, respectively. The first value of each thyroid index under the juice regimen was recorded 3 months after having started taking the juice; the last value was recorded 9 months after having started taking the juice. Numbers in brackets are the reference ranges.
Based on this positive experience, we decided to test the effects of ABMJ administration in HT women with levothyroxine-untreated SCH and high levels of TPOAb.
Materials and methods
Materials
The ABMJ taken by all patients was Aloe Vera2 by ZUCCARI (Trento, Italy). Each 100 ml preparation of ABMJ, which has an energetic value of 7.40 kcal (31.20Kj), contains nonpasteurized and noncarbon filtered Aloe vera leaf juice and pulp (49.8 g), 0.2 g fats (0% saturated), 1.2 g carbohydrates, zero proteins, and 0.06 g minerals (of which, 0.02 g sodium). The juice also contains citric acid as an acidifier and sodium benzoate and potassium sorbate as typical preservatives with biocidal properties. Noteworthy, Aloe Vera2 is free of aloin, a substance primarily contained in the outer cuticles of the leaves that is irritating to the intestinal mucosa. Once opened, the one-liter bottle has to be stored in the refrigerator, as recommended by the producer.
Patients
Based on the observations summarized in Table 1, we aimed to recruit women, aged 30 to 55 years and with HT-associated SCH (TSH > 4.0 mU/L; high levels of TPOAb), who had never been treated with L-T4 and/or supplements. In addition to past and current treatment with L-T4 and/or supplements, exclusion criteria were: (i) concurrent diseases, including diabetes mellitus and other autoimmune diseases; (ii) use of any nutraceuticals/drugs that affect the hypothalamic-pituitary-thyroid axis and autoimmunity. These criteria were verified with the family physicians.
Patients were informed of the aforementioned initial observation and enrolled upon signing an informed consent form. Thirty women, aged 20 to 55 years, were enrolled and all of them completed the study. They were treated with 50 ml ABMJ (Aloe Vera2), which was taken in the morning on an empty stomach. To minimize confounding factors, all patients were directed to the same natural product health store where the said co-author bought ABMJ. The natural product health store personnel informed us that each patient had bought enough bottles to complete the study. Paralleling the initial observation, the duration of study was 9 months, with serum TSH, FT3, FT4 and TPOAb measured at baseline (time zero), and 90 ± 3 days (3 months) and 180 ± 3 days (9 months) later. Measurement of FT3 was added for sake of completeness.
For the purpose of comparison with a group of Hashimoto’s thyroiditis women who were under no thyroid hormone replacement therapy and no supplementation with nutraceuticals, we took advantage of a database on patients with Hashimoto’s thyroiditis (Interdepartmental Program of Molecular & Clinical Endocrinology and Women’s Endocrine Health, University hospital of Messina). Based on age and levels of the fundamental indices (serum TSH and TPOAb) at time zero, the 30 Aloe-treated women could be matched to 15 women with SCH of comparable age (21–57 years) and baseline levels of serum TSH and TPOAb that had been measured with the same corresponding kits.
Assays
Serum TSH, FT4, FT3 and TPOAb were assayed using electrochemilumiscent kits (Roche, Mannheim, Germany). Reference values were 0.27–4.2 mU/L for TSH, 9.3–17.1 pg/ml (12–22 pmol/L) for FT4, 2.0–4.4 pg/ml (3.1–6.8 pmol/L) for FT3, and 0–100 U/ml for TPOAb. To avoid intra-assay variations, sera were stored at −20 °C, and all 90 sera for each analyte (30 patients × 3 time points) were assayed in one run. Intra-assay coefficients of variations for all four thyroid analytes were <3%.
Statistics
Data are given as mean ± standard deviation (SD) and median. Differences between m ± SD of continuous variables were analyzed by ANOVA or by the Wilcoxon signed rank test for the indices with nongaussian distribution (TSH and TPOAb). Differences between proportions were analyzed by the two-tailed Fisher’s exact test. The threshold for significance was set at a P value of < 0.05.
Results
Biochemical thyroid data
Data are summarized in Table 2, Table 3, and illustrated in Figs. 1 and 2.
Table 2.
Changes observed in the specified biochemical thyroid indices (all analyzed as continuous variables) during and at the end of a 9-month duration administration of Aloe vera barbadensis Miller juice in 30 women with Hashimoto’s thyroiditis-related subclinical hypothyroidism (HT-SCH), and in a control group of 15 women under no L-T4 therapy or any nutraceutical supplementation.a
| Statistics (P value) b |
||||||
|---|---|---|---|---|---|---|
| Baseline c | 3 Months c | 9 Months c | 3 Mos. vs baseline | 9 Mos. vs baseline | 9 Vs 3 months | |
| Study group | ||||||
| TSH, mU/L | 5.19 ± 0.61 [5.12] | 3.12 ± 1.01; −40% [2.99; −42%] | 2.01 ± 0.57; −61% [1.92; −62%] | 3.1 × 10−6 | 5.6 × 10−6 | 2.0 × 10−6 |
| FT4, pmol/L | 9.63 ± 1.58 [9.17] | 10.67 ± 1.31; +11% [10.53; +15%] | 11.83 ± 1.50; +23% [11.54; +26%] | 0.0073 | 1.8 × 10−6 | 0.71 |
| FT3, pmol/L | 5.29 ± 0.61 [5.22] | 4.38 ± 0.47; −17% [4.37; −16%] | 4.32 ± 0.59; −18% [4.33; −17%] | 4.2 × 10−6 | 3.0 × 10−5 | 0.62 |
| FT4:FT3 ratio | 1.83 ± 0.32 [1.80] | 2.47 ± 0.46; +35% [2.39; +33%] | 2.78 ± 0.63; +52% [2.69; +49%] | 7.1 × 10−6 | 5.5 × 10−6 | 0.035 |
| TPOAb, U/ml | 1020 ± 202 [1021] | 825 ± 183; −19% [777; −24%] | 347 ± 140; −58% [345; −56%] | 0.00034 | 1.7 × 10−6 | 1.8 × 10−6 |
| Control group | ||||||
| TSH, mU/L | 5.44 ± 0.86 [5.40] (P = 0.31) | 5.43 ± 0.78 [5.60] (P = 1.5 × 10−6) | 5.80 ± 1.18 [5.84] (P = 6.4 × 10−8) | 0.92 | 0.35 | 0.23 |
| FT4, pmol/L | 9.58 ± 1.48 [9.04] (P = 0.92) | 9.65 ± 1.46 [9.03] (P = 0.022) | 9.39 ± 1.59 [8.78] (P = 8.6 × 10−6) | 0.89 | 0.73 | 0.64 |
| FT3, pmol/L | 5.31 ± 0.75 [5.25] (P = 0.86) | 5.26 ± 0.71 [5.28] (P = 1.1 × 10−5) | 5.37 ± 0.67 [5.31] (P = 2.8 × 10−6) | 0.82 | 0.85 | 0.66 |
| FT4:FT3 ratio | 1.81 ± 0.26 [1.78] (P = 0.83) | 1.85 ± 0.30 [1.70] (P = 2.4 × 10−5) | 1.75 ± 0.32 [1.69] (P = 5.8 × 10−7) | 0.70 | 0.39 | 0.59 |
| TPOAb, U/ml | 939 ± 140 [930] (P = 0.19) | 914 ± 135 [902] (P = 0.026) | 977 ± 178 [962] (P = 5.8 × 10−7) | 0.79 | 0.58 | 0.33 |
Individual changes are illustrated in Fig. 1.
Difference between means analyzed by ANOVA or Wilcoxon signed rank test for the nongaussian distributed indices (TSH and TPOAb). Statistically significant differences are typed bold-face.
Values in brackets are medians. P values in parentheses refer to the comparison with the study group. Statistically significant differences are typed bold-face.
Table 3.
Changes observed in the specified biochemical thyroid indices (all analyzed as categorical variables) during and at the end of a 9-month duration administration of Aloe vera barbadensis Miller juice in 30 women with Hashimoto’s thyroiditis-related subclinical hypothyroidism.
| Statistics (P value) a |
||||||
|---|---|---|---|---|---|---|
| Baseline | 3 Months | 9 Months | 3 Mos. vs baseline | 9 Mos. vs baseline | 9 Vs 3 months | |
| TSH > 4.0 mU/L | 30 (100%) | 5 (16.7%) | 0 |
<0.0001 OR = 0.003 |
<0.0001 OR = 0.0003 |
0.05 OR = 0.08 |
| TSH ≤ 4.0 mU/L | 0 | 25 (83.3%) | 30 (100%) | |||
| TSH > 2.5 mU/L | 30 (100%) | 20 (66.7%) | 5 (16.7%) |
0.0008 OR = 0.032 |
<0.0001 OR = 0.0035 |
0.0002 OR = 0.10 |
| TSH ≤ 2.5 mU/L | 0 | 10 (33.3%) | 25 (83.3%) | |||
| FT4 < 10 pmol/L | 18 (60%) | 8 (26.7%) | 1 (3.4%) |
0.018 OR = 0.24 |
<0.0001 OR = 0.023 |
0.026 OR = 0.09 |
| FT4 ≥ 10 pmol/L | 12 (40%) | 22 (73.3%) | 29 (96.6%) | |||
| TPOAb > 1000 U/ml | 18 (60%) | 5 (16.7%) | 0 |
0.0012 OR = 0.13 |
<0.0001 OR = 0.011 |
0.05 OR = 0.08 |
| TPOAb ≤ 1000 U/ml | 12 (40%) | 25 (83.3%) | 30 (100%) | |||
| TPOAb > 400 U/ml | 30 (100%) | 30 (100%) | 11 (37%) | 1.0 |
<0.0001 OR = 0.0097 |
<0.0001 OR = 0.0097 |
| TPOAb ≤ 400 U/ml | 0 | 0 | 19 (63%) | |||
| TSH > 4 mU/L + FT4 < 10 pmol/L + TPOAb > 1000 U/ml | 12 (40%) | 0 | 0 |
<0.0001 OR = 0.023 |
<0.0001 OR = 0.023 |
1.0 |
| TSH > 4 mU/L + FT4 < 10 pmol/L + TPOAb > 400 U/ml | 18 (60%) | 8 (26.7%) | 1 (3.4%) |
0.018 OR = 0.24 |
<0.0001 OR = 0.023 |
0.026 OR = 0.09 |
Differences between proportions analyzed by the Fisher’s exact test. Statistically significant differences are typed bold-face. Borderline significant differences (P values between 0.10 and 0.05) are typed bold-face italics. OR = Odds ratio.
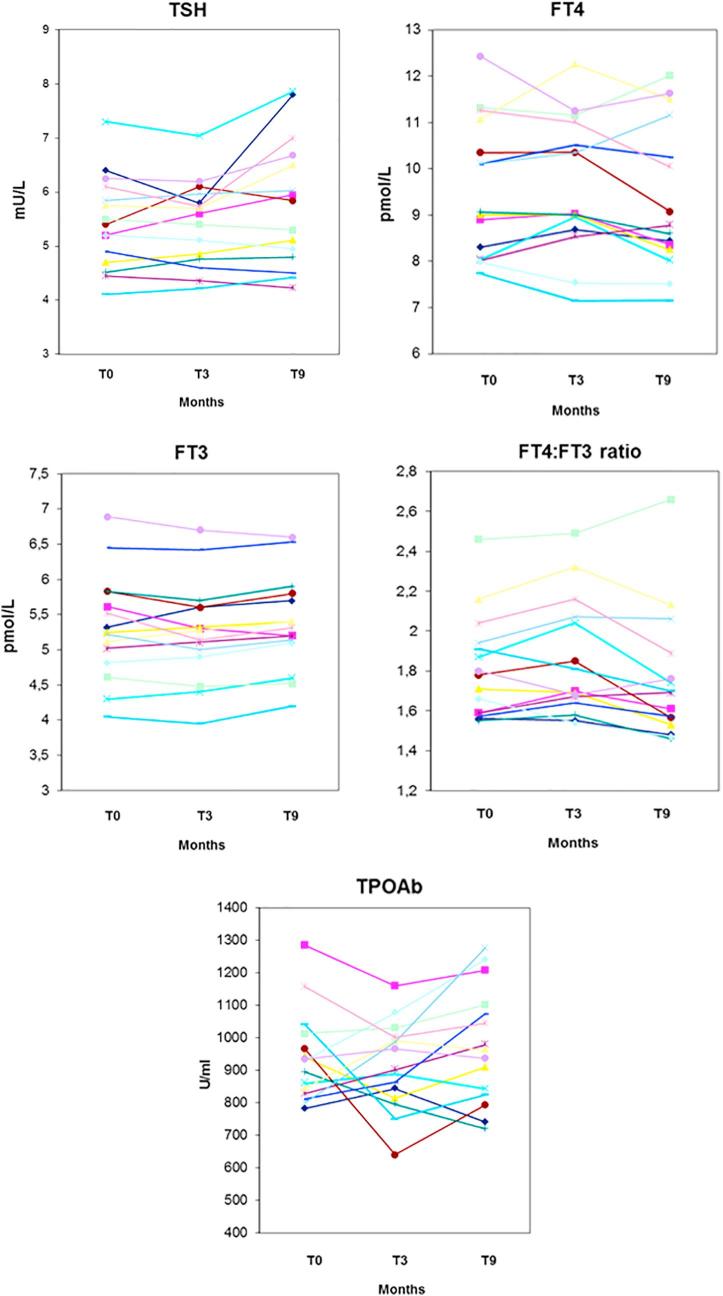
Fig. 1.
Individual changes of serum TSH, FT4, FT3, FT4 to FT3 ratio and TPOAb in 30 women with Hashimoto’s thyroiditis-associated subclinical hypothyroidism who took Aloe Barbadensis Miller juice for 9 months.
Fig. 2.
Individual changes of serum TSH, FT4, FT3, FT4 to FT3 ratio and TPOAb in 15 controls (women with Hashimoto’s thyroiditis-associated subclinical hypothyroidism who did not take Aloe Barbadensis Miller juice).
In the control group (Table 2), all biochemical indices were statistically similar to the corresponding ones of the study group. In addition, they did not change significantly over time.
In sharp contrast, in the study group all indices changed significantly at the first evaluation (end of the third month) (Table 2). At this first post-treatment evaluation, serum TPOAb decreased by one-fourth, while FT4 increased by one-sixth and FT3 decreased by the same magnitude. As expected from the well-known linear-log relationship in the thyroid hormone-driven feedback regulation of TSH secretion, serum TSH changed in the expected direction (decrease) and to a greater extent (3 times more) than the 15% increase in serum FT4. The 3-fold greater change held six months later, in response to the further increase in serum FT4 (approximately 60% compared to 24% increase in serum FT4); TPOAb levels continued to fall. Changes of serum TPOAb, TSH and FT4 augmented over time, whereas FT3 did not decrease further at month 9 compared with month 3. As a result, the FT4:FT3 ratio continued to increase (Table 2). Fig. 1, Fig. 2 show that, both in the patients and in the control group, changes for each given index were uniform, with virtually no outliers.
In brief, all the changes observed are consistent with an amelioration of the intrathyroid autoimmunity process, with rescue of T4 synthesis by the thyrocyte (and subsequent decline in serum TSH) and simultaneous impairment of the deiodinase-mediated peripheral T3 formation.
Table 3 shows that euthyroidism (TSH ≤ 4.0 mU/L) was achieved in 83% of patients already at the end of the third month of treatment. This proportion becomes exactly one-third (33%) using the very stringent criterion of TSH ≤ 2.5 mU/L. At the end of the ninth month, 100% or 83% of women were euthyroid, depending on TSH threshold. In contrast, all control women retained their SCH status, with a TSH > 4.0 mU/L (data not shown).
The bottom part of Table 3 shows that the worst biochemical scenario based on the combination of three indices (TSH, FT4 and TPOAb) occurred at baseline in 40 or 60% of patients (depending on the TPOAb threshold used). However, already by the third month of treatment the scenario improved at the best possible level (disappearance) or improved significantly.
None of the 30 women complained of any side-effects during the nine months of study.
Discussion
As checked with a PubMed search on September 30, 2017 using the string “Aloe and thyroid”, except for a letter to the Editor reporting on a single patient [30] and one paper on male mice [31], there are no other studies describing the effects of Aloe on thyroid function. A 56-yr-old woman with lichen planus, who refused treatment with corticosteroids, took Aloe vera juice (10 ml/day) for 11 months [30]. No improvement of lichen occurred. Serum FT3 was barely low, FT4 at the lower reference limit and TSH normal. After discontinuation of Aloe, thyroid hormone levels were reported to be normal. Taking into account the few data provided and not having mentioned the presence of lymphocytes in the thyroid smear, the hormone picture is consistent with central hypothyroidism or with the nonthyroid illness syndrome, rather than primary hypothyroidism.
In the mice study by Kar et al. [31], groups of seven male mice were treated, for 15 days, with extracts of Aloe vera (125 mg/kg) or other plants by gastric intubation. Compared with administration of vehicle (control), a significant increase in serum T4 concentration was observed in mice treated with Aegle marmelos or Bacopa monnieri (+31% or +41%, P < 0.01 for both), while T3 concentration decreased in the former group only (−62% [P < 0.001] or +5.7%). In Aloe vera-treated animals, both T4 and T3 levels decreased significantly (−25% [P < 0.01] and −13% [P < 0.05]). No mechanistic explanation was provided [31].
Here we have shown that the fortuitous observation of decreased serum TSH and TPOAb in association with an increased concentration of serum FT4 after a 9-month administration of Aloe vera juice was confirmed in a cohort of women with the same thyroid disorder: HT-related SCH. We also measured serum FT3 and found a decline after 3 months only, with no further decline after another six months. In sharp contrast, untreated SCH women of comparable age and baseline levels of TSH, FT4, FT3 and TPOAb had no significant changes in any index.
As summarized in Table 4, several studies tested the effects of selenium in patients with autoimmune thyroiditis, due to its role in endocrine and immune functions, and modulation of the inflammatory response. Supplementation with selenium for one to 12 months in HT patients has given variable results on the endpoint measured (serum TPOAb levels), with more consistent and overall greater decline upon using the organic and more bioavailable form (selenomethionine) [14], [15], [16], [17], [18], [19], [20], [21], [22], [23], [24], [25], [26]. With a single exception (−56%) in a Greek study [19], the decrease from baseline in serum TPOAb is not >44%, but in 5/13 studies is nil (Table 4). Recent Italian studies [27], [28], [29] have evaluated the effects of the 6-month administration of selenomethionine associated with an insulin-sensitizer (myo-inositol). In addition to TPOAb, serum TSH and, sometimes, serum thyroid hormones were evaluated (Table 4). With a single exception (−14%) [28], the fall in serum TPOAb was just below 50% (Table 4). Serum TSH decreased by approximately 30%, while the only study that evaluated serum FT4 [28] reported an increase of 14% from baseline. Therefore, our findings of changes in TPOAb, TSH and FT4 at 3 months (−24%, −42% and +15%) and 9 months (−56%, −62% and +26%) are comparable, if not superior, to those reported in the literature with selenium alone or combined with myo-inositol (Table 4).
Table 4.
Summary of changes in serum thyroperoxidase (TPOAb) after the specified supplementation in patients with thyroid autoimmune thyroiditis, as reported in the literature.a
| Author (Ref.) | No. of patients | Supplementation | Changes in TPOAb |
|---|---|---|---|
| Metro et al. [this study] | 30 (all F) | Aloe vera barbadensis juice (50 ml/d × 9 months) | −34% (3 months), −56% (9 months) |
| Eskes [12] | 61 (all F) | Sodium selenite (80 µg/d × 6 months) | None |
| Nacamulli [13] | 76 (65F, 11M) | Sodium selenite (80 µg/d × 12 months) | −30% |
| Karanikas [14] | 36 (all F) | Sodium selenite (200 µg/d × 3 months) | None |
| Gartner [15] | 70 (all F) | Sodium selenite (200 µg/d × 3 months) | −36% |
| Gartner [16] | 70 (all F) | Sodium selenite (200 µg/d × 6 months) | −44% |
| Bonfig [17] | 49 (33F, 16M) | Sodium selenite (200 µg/d × 12 months) | None |
| Turker [18] | 88 (all F) | Selenomethionine (100 µg/d × 3 months) | None |
| Turker [18] | 88 (all F) | Selenomethionine (200 µg/d × 3 months) | −26% |
| Duntas [19] | 65 (56F, 9M) | Selenomethionine (200 µg/d × 6 months) | −56% |
| Mazopakis [20] | 80 (all F) | Selenomethionine (200 µg/d × 6 months) | None |
| Zhu [21] | 77 (?) | Selenomethionine (200 µg/d × 6 months) | −20% |
| Balazs [22] | 70 (all F) | Selenomethionine (200 µg/d × 6 months) | % reduction not specified |
| Pirola [23] | 96 (60F, 36M) | Selenomethionine (83 µg/d × 4 months) | −7% in the responders; −10% in the nonresponders |
| Negro [24] | 77 (all F) | Selenomethionine (200 µg/d from 1st trimester of gestation to 12 months postpartum) | −48% (12 months postpartum) |
| Nordio [25] | 22 (all F) | Selenomethionine (83 µg/d × 6 months) | −42% |
| Nordio [25] | 24 (all F) | Selemethionine + Myoinositol (83 µg/d + 600 mg/d × 6 months) | −43% |
| Nordio [26] | 86 (78F, 8M) | Selenomethionine + Myoinositol (83 µg/d + 600 mg/d × 6 months) | −14% |
| Ferrari [27] | 21 (16F, 5M) | Selemethionine + Myoinositol (83 µg/d + 600 mg/d × 6 months) | −45% |
The following studies have also evaluated TSH and FT4. Metro et al. [this study] found a reduction in serum TSH of −42% (3 months) and −62% (9 months), with an increase in serum FT4 of 15% (3 months) and 26% (9 months). Nordio et al. [26] reported a decrease in serum TSH of 28% and an increase in serum FT4 of 14% (6 months). This study [26] also measured FT3, which increased by 4.5%. Ferrari et al. [27] reported a decrease in serum TSH of 33% (6 months). Pirola et al. [23] studied 192 patients with HT-associated subclinical hypothyroidism (HT-SCH) and serum TSH > 4.5 mU/L who were supplemented (study group) or not supplemented (control group) with selenomethionine for 4 months. After 4 months, responders (that is, patients with normalized TSH) were 30/96 (31%) compared to 3/96 (3.1%) in the untreated HT-SCH group. In the treated or untreated group, TSH declined by 45% or 41%, while FT4 increased by 3% or 2%.
Similarly to Nordio and Basciani [28], we complemented measurement of serum FT4 with that of serum FT3 in our subclinically hypothyroid HT patients. Unlike the 4.5% increase in serum FT3 following a 6-month supplementation with selenomethionine plus myo-inositol [28], we found a 16% decrease after 3 months of supplementation with Aloe vera, and this value did not change in either direction after another 6 months of supplementation (Table 2). Consequently, the FT4:FT3 ratio (pmol/L:pmol/L) increased from 1.83 at baseline to 2.47 (+35%) at month 3 and 2.78 (+52%) at month 9 after Aloe vera supplementation, as did, but to a lower extent (2.95 to 3.21 [+9%]), after 6 months of supplementation with selenomethionine plus myo-inositol [28]. Upon studying in vitro other HT patients’ circulating lymphocytes that were cultured in the presence of selenomethionine plus myo-inositol, our group has found a dose-dependent decline in the extracellular concentration of the hydrogen peroxide-stimulated lymphocyte-secreted chemokines (CCL2, CXCL9 and CXCL10) [32]. Because (i) circulating lymphocytes can be a proxy of the lymphocytes that infiltrate the thyroid in HT, (ii) the said chemokines are markers of functional damage of the thyrocyte [33], [34], and (iii) increased peripheral deiodination of T4 into the more potent T3 is a compensatory mechanism for the diminished secretion of T4 from the thyroid gland, we propose the following interpretation for the amelioration of the thyroid autoantibody profile and hormone profile induced by either selenemethionine plus myo-inositol or Aloe vera juice. Upon supplementation, the decreased intrathyroid inflammation and decreased chemokine-mediated damage of the thyrocyte result in greater T4 secretion and decreased need to generate T3 in the periphery compared to the pre-supplementation period.
The benefit provided by the Aloe vera juice can be measured on a clinically relevant scale, namely return to the euthyroid status. Before supplementation, 100% of our patients were subclinically hypothyroid (TSH > 4.0 mU/L), but nine months after supplementation none was so. If the TSH threshold is lowered to 2.5 mU/L (which is relevant in the setting of the first trimester of gestation), after 3 and 9 months of Aloe vera supplementation two-thirds and one-sixth of women would have the undesirable level of TSH > 2.5 mU/L. Remaining in the obstetrical setting, the decline in TPOAb from the first week of gestation through 12 months postpartum upon supplementing Italian women with selemethionine for all this time was beneficial [26], in that the decline of TPOAb (a well-known risk factor for PPT development) was followed by a 2-fold lower rate of PPT and hypothyroidism compared to the untreated group. We speculate that the same outcome would very likely occur should women take Aloe vera juice for the entire duration of gestation and the first 12 months after delivery.
In conclusion, a 9-month treatment with 50 ml/day of Aloe vera juice restores euthyroidism in 100% of HT-related initial/mild hypothyroidism (also known as subclinical hypothyroidism), with appreciable results already evident after 3 months, and markedly decreases their highly elevated serum TPOAb levels (again already evident after 3 months). These beneficial results are comparable to, if not greater than, those reported in the literature for selenium alone or selenium associated with myo-inositol. In addition to an ameliorative effect on thyroid inflammation and subsequent rescue of the hormone biosynthesis by the thyrocytes, Aloe vera juice is likely to act on the peripheral deiodination of T4 into T3. Randomized, double-blind placebo-controlled trials are warranted.
Declarations of interest
None. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
- 1.Surjushe A., Vasani R., Saple D.G. Aloe vera: a short review. Indian J Dermatol. 2008;53:163–166. doi: 10.4103/0019-5154.44785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.http://www.livestrong.com/article/255743-what-is-an-aloe-barbadensis-leaf (accessed May 20, 2017).
- 3.Waller G.R., Mangiafico S., Ritchey C.R. A chemical investigation of Aloe Barbadensis Miller. Proc Okla Acad Sci. 1978;58:69–76. [Google Scholar]
- 4.http: //www.nacomi.it/materie-prime/burro-karite.html (accessed January 8, 2018).
- 5.https://www.sciencedirect.com/topics/medicine-and-dentistry/serenoa (accessed January 8, 2018).
- 6.WHO Monographs on Selected Medicinal Plants - Volume 1. http://apps.who.int/medicinedocs/en/d/Js2200e/5.html (accessed May 20, 2017).
- 7.Breese McCoy S.J. Coincidence of remission of postpartum Graves’ disease and use of omega-3 fatty acid supplements. Thyroid Res. 2011;4:16. doi: 10.1186/1756-6614-4-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Benvenga S., Vigo M.T., Metro D., Granese R., Vita R., Le Donne M. Type of fish consumed and thyroid autoimmunity in pregnancy and postpartum. Endocrine. 2016;52:120–129. doi: 10.1007/s12020-015-0698-3. [DOI] [PubMed] [Google Scholar]
- 9.Benvenga S., Amato A., Calvani M., Trimarchi F. Effects of carnitine on thyroid hormone action. Ann N Y Acad Sci. 2004;1033:158–167. doi: 10.1196/annals.1320.015. [DOI] [PubMed] [Google Scholar]
- 10.An J.H., Kim Y.J., Kim K.J., Kim S.H., Kim N.H., Kim H.Y. L-carnitine supplementation for the management of fatigue in patients with hypothyroidism on levothyroxine treatment: a randomized, double-blind, placebo-controlled trial. Endocr J. 2016;63:885–895. doi: 10.1507/endocrj.EJ16-0109. [DOI] [PubMed] [Google Scholar]
- 11.Benvenga S., Sindoni A. L-carnitine supplementation for the management of fatigue in patients with hypothyroidism on levothyroxine treatment. Endocr J. 2016;63:937–938. doi: 10.1507/endocrj.EJ16-0374. [DOI] [PubMed] [Google Scholar]
- 12.Marcocci C., Kahaly G.J., Krassas G.E., Bartalena L., Prummel M., Stahl M. Selenium and the course of mild Graves’ orbitopathy. N Engl J Med. 2011;364:1920–1931. doi: 10.1056/NEJMoa1012985. [DOI] [PubMed] [Google Scholar]
- 13.Duntas L.H., Benvenga S. Selenium: an element for life. Endocrine. 2015;48:756–775. doi: 10.1007/s12020-014-0477-6. [DOI] [PubMed] [Google Scholar]
- 14.Eskes S.A., Endert E., Fliers E., Birnie E., Hollenbach B., Schomburg L. Selenite supplementation in euthyroids subjects with thyroid peroxidase antibodies. Clin Endocrinol (Oxf) 2014;80:444–451. doi: 10.1111/cen.12284. [DOI] [PubMed] [Google Scholar]
- 15.Nacamulli D., Mian C., Petricca D., Lazzarotto F., Barollo S., Pozza D. Influence of physiological dietary selenium supplementation on the natural course of autoimmune thyroiditis. Clin Endocrinol (Oxf) 2010;73:535–539. doi: 10.1111/j.1365-2265.2009.03758.x. [DOI] [PubMed] [Google Scholar]
- 16.Karanikas G., Schuetz M., Kontur S., Duan H., Kommata S., Schoen R. No immunological benefit of selenium in consecutive patients with autoimmune thyroiditis. Thyroid. 2008;18:7–12. doi: 10.1089/thy.2007.0127. [DOI] [PubMed] [Google Scholar]
- 17.Gartner R., Gasnier B.C., Dietrich J.W., Krebs B., Angstwurm M.W. Selenium supplementation in patients with autoimmune thyroiditis decreases thyroid peroxidase antibodies concentrations. J Clin Endocrinol Metab. 2002;87:1687–1691. doi: 10.1210/jcem.87.4.8421. [DOI] [PubMed] [Google Scholar]
- 18.Gartner R., Gasnier B.C. Selenium in the treatment of autoimmune thyroiditis. BioFactors. 2003;19:165–170. doi: 10.1002/biof.5520190309. [DOI] [PubMed] [Google Scholar]
- 19.Bonfig W., Gartner R., Schmidt H. Selenium supplementation does not decrease thyroid peroxidase antibody concentration in children and adolescents with autoimmune thyroiditis. Sci World J. 2010;10:990–996. doi: 10.1100/tsw.2010.91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Turker O., Kumanlioglu K., Karapolat I., Dogan I. The Selenium treatment in autoimmune thyroiditis: 9-month follow-up with variable doses. J Endocrinol. 2006;190:151–156. doi: 10.1677/joe.1.06661. [DOI] [PubMed] [Google Scholar]
- 21.Duntas L.H., Mantzou E., Koutras D.A. Effects of a six month treatment with selenomethionine in patients with autoimmune thyroiditis. Eur J Endocrinol. 2003;148:389–393. doi: 10.1530/eje.0.1480389. [DOI] [PubMed] [Google Scholar]
- 22.Mazokopakis E.E., Papadakis J.A., Papadomanolaki M.G., Batistakis A.G., Giannakopoulos T.G., Protopapadakis E.E. Effects of 12 months treatment with L-selenomethionine on serum anti-TPO levels in patients with Hashimoto’s thyroiditis. Thyroid. 2007;17:609–612. doi: 10.1089/thy.2007.0040. [DOI] [PubMed] [Google Scholar]
- 23.Zhu L., Bai X., Teng W.P., Shan Z.Y., Wang W.W., Fan C.L. Effects of selenium supplementation on antibodies of autoimmune thyroiditis. Zhonghua Yi Xue Za Zhi. 2012;92:2256–2260. [PubMed] [Google Scholar]
- 24.Balázs C. The effect of selenium therapy on autoimmune thyroiditis. Orv Hetil. 2008;149:1227–1232. doi: 10.1556/OH.2008.28408. [DOI] [PubMed] [Google Scholar]
- 25.Pirola I., Gandossi E., Agosti B., Delbarba A., Cappelli C. Selenium supplementation could restore euthyroidism in subclinical hypothyroid patients with autoimmune thyroiditis. Endokrynol Pol. 2016;67:567–571. doi: 10.5603/EP.2016.0064. [DOI] [PubMed] [Google Scholar]
- 26.Negro R., Greco G., Mangieri T., Pezzarossa A., Dazzi D., Hassan H. The influence of selenium supplementation on postpartum thyroid status in pregnant women with thyroid peroxidase autoantibodies. J Clin Endocrinol Metab. 2007;92:1263–1268. doi: 10.1210/jc.2006-1821. [DOI] [PubMed] [Google Scholar]
- 27.Nordio M., Pajalich R. Combined treatment with Myo-inositol and selenium ensures euthyroidism in subclinical hypothyroidism patients with autoimmune thyroiditis. J Thyroid Res. 2013;2013:424163. doi: 10.1155/2013/424163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nordio M., Basciani S. Treatment with myo-inositol and selenium ensures euthyroidism in patients with autoimmune thyroiditis. Int J Endocrinol. 2017;2017:2549491. doi: 10.1155/2017/2549491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ferrari S.M., Fallahi P., Di Bari F., Vita R., Benvenga S., Antonelli A. Myo-inositol and selenium reduce the risk of developing overt hypothyroidism in patients with autoimmune thyroiditis. Eur Rev Med Pharmacol Sci. 2017;21(2 Suppl):36–42. [PubMed] [Google Scholar]
- 30.Pigatto P.G., Guzzi G. Aloe linked to thyroid dysfunction. Arch Med Res. 2005;36:608. doi: 10.1016/j.arcmed.2005.03.022. [DOI] [PubMed] [Google Scholar]
- 31.Kar A., Panda S., Bharti S. Relative efficacy of three medicinal plant extracts in the alteration of thyroid hormone concentrations in male mice. J Ethnopharmacol. 2002;81:281–285. doi: 10.1016/s0378-8741(02)00048-x. [DOI] [PubMed] [Google Scholar]
- 32.Benvenga S., Vicchio T., Di Bari F., Vita R., Fallahi P., Ferrari S.M. Favorable effects of Myo-inositol, selenomethionine or their combination on the hydrogen peroxide induced oxidative stress of peripheral mononuclear cells from patients with Hashimoto’s thyroiditis: preliminary in vitro studies. Eur Rev Med Pharmacol Sci. 2017;21(2 Suppl):89–101. [PubMed] [Google Scholar]
- 33.Antonelli A., Ferrari S.M., Giuggioli D., Ferrannini E., Ferri C., Fallahi P. Chemokine (C-X-C motif) ligand (CXCL)10 in autoimmune diseases. Autoimmun Rev. 2014;13:272–280. doi: 10.1016/j.autrev.2013.10.010. [DOI] [PubMed] [Google Scholar]
- 34.Antonelli A., Ferrari S.M., Frascerra S., Galetta F., Franzoni F., Corrado A. Circulating chemokine (CXCmotif) ligand (CXCL)9 is increased in aggressive chronic autoimmune thyroiditis, in association with CXCL10. Cytokine. 2011;55:288–293. doi: 10.1016/j.cyto.2011.04.022. [DOI] [PubMed] [Google Scholar]