Figure 4. Selective Chemical Inhibition of GSK3β, Not GSK3α, Promotes ESC Self-Renewal.
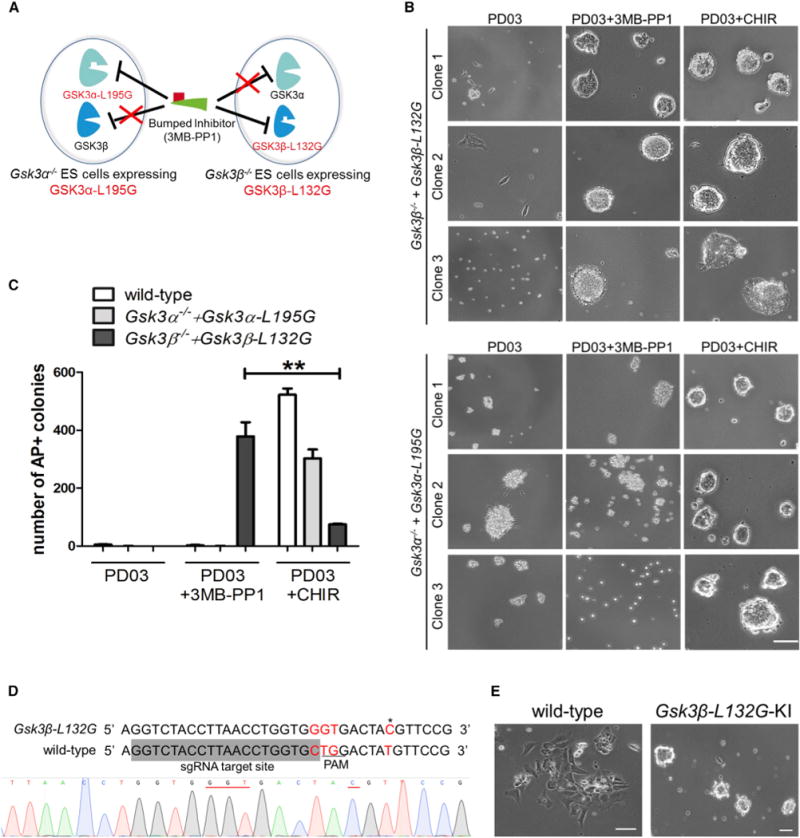
(A) Diagram showing the chemical-genetic strategy to selectively inhibit GSK3α or GSK3β.
(B) Phase-contrast images of Gsk3β−/− + Gsk3β-L132G and Gsk3α−/− + Gsk3α-L195G ESCs plated at a clonal density and cultured in N2B27 medium supplemented with the indicated inhibitors for 7 days. Representative images show results from thee independent clones of mutant ESC lines with GSK3 transgene levels similar to the physiological levels. Scale bar, 100 μm.
(C) Quantification of AP+ colonies of different ESC lines. ESCs were plated onto six-well plates at a density of 1,000 cells/well and cultured for 7 days in N2B27 medium supplemented with the indicated inhibitors. Data represent means ± SD of three biological replicates. **p < 0.01.
(D) Strategy and sequencing result for introducing the L132G point mutation at the Gsk3β locus in mouse ESCs by CRISPR/Cas9. The single guide RNA (sgRNA) target site is shaded in gray. A targeting vector containing 5′ homologous arm-loxP-CAG-PuroR-loxP-3′ homologous arm was used as the template for homologous recombination. The 5′-NGG PAM site (TGG) is underlined in the wild-type locus. The mutated code (CTG→GGT) is highlighted in red. The restriction fragment length polymorphism (RFLP) site is labeled in red and indicated by an asterisk. This RFLP site (AˆCGT) can be digested by HpyCH4IV. Sequencing result showing the knockin of the CTG→GGT (underlined in red) mutation at the Gsk3β-L132G locus in one of the ESC clones.
(E) Representative images of wild-type and Gsk3β-L132G knockin (Gsk3β-L132G-KI) ESCs cultured in N2B27 medium supplemented with PD03 and 3MB-PP1 for 5 days. Gsk3β-L132G-KI ESCs self-renewed continuously in PD03 + 3MB-PP1, whereas wild-type ESCs differentiated rapidly under the same conditions. Scale bars, 100 μm.
