Abstract
Serum albumin and conventional BMI (cBMI) are commonly used indices of malnutrition status. Because cBMI does not reflect fluid accumulation, modified body mass index (mBMI, serum albumin × cBMI) is a more accurate measure of malnutrition status. This study investigated the association between preoperative mBMI and postoperative 1-year mortality, in comparison with serum albumin and cBMI. Medical records of 80,969 adult patients who underwent surgical procedures in a tertiary care hospital between 1 January, 2011 and 31 December, 2015 were retrospectively reviewed. Postoperative 1-year mortality increased with reduction in cBMI, mBMI, and albumin separately (P < 0.001). When considering interaction between cBMI and albumin, albumin was the only significant factor of postoperative 1-year mortality [odds ratio: 0.377, 95% confidence interval (0.245–0.579), P < 0.001], while cBMI or interaction (cBMI * albumin) was not significant (P > 0.05). Adjusted area under the curve (AUC) was highest (0.885) in the overall model (cBMI + albumin + cBMI * albumin); adjusted AUC between mBMI and albumin did not differ significantly (P = 0.558). Low albumin is the strongest independent predictor of postoperative 1-year mortality among the three variables (albumin, cBMI, mBMI). Adding cBMI to albumin does not increase the validity of the AUC of albumin.
Introduction
Malnutrition, a common complication of chronic or severe disease, is known to exacerbate disease prognoses1. Patients’ malnutrition status may have adverse effects on their postoperative mortality or prognosis2. Therefore, preoperative malnutrition is an important clinical problem; conventional body mass index (cBMI) and serum albumin level are commonly used as indicators of malnutrition status3,4. Recent studies have reported that low preoperative BMI5,6 and low serum albumin level7,8 exacerbate postoperative mortality and prognosis. This indicates the importance of preoperative nutritional status given the recent trend of increasing number of surgeries among elderly patients9.
However, it is still not clear how accurately cBMI measures the nutritional status of patients10,11, because it has limitations in reflecting fluid balance, such as fluid accumulation or dehydration. In fact, such limitations of cBMI were suggested to affect patients undergoing liver transplantation, in which ascites is a common problem. A new approach suggested to overcome these limitations, is the use of the modified BMI (mBMI)12,13. The mBMI considers serum albumin level and cBMI in combination, and thus is a relevant tool for patients who undergo liver transplantation10–13. However, there are no data on the effects of mBMI on the postoperative prognosis in the general population.
Therefore, we aimed to investigate the value of preoperative mBMI as a predictor of postoperative mortality, as compared to previously used predictors (cBMI and serum and albumin).
Results
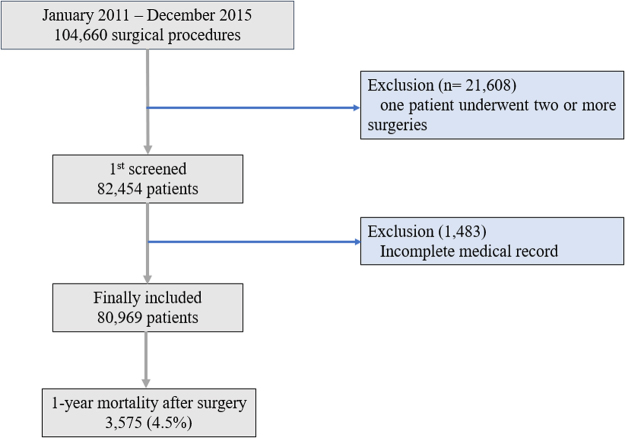
A total of 82,452 adult patients underwent 104,660 cases of operations or surgical procedures at Seoul National University Bundang Hospital (SNUBH) between January 2011 and December 2015. Of these, 1,483 patients were excluded due to inaccurate or incomplete medical records; the remaining 80,969 patients were included in the final analysis. Among the 80,969 patients, 3,575 (4.4%) died within 1 year of the operation (Fig. 1). The demographic and clinical characteristics of the final analysis are presented in Table 1.
Figure 1.
Flow chart for patient selection.
Table 1.
Baseline characteristics of all patients.
| Total (n = 80,969) | Number (%) | Mean | SD |
|---|---|---|---|
| Operation | |||
| Cardiovascular surgery | 1,134 (1.4%) | ||
| Non-cardiovascular surgery | 79,835 (98.6%) | ||
| Male (%) | 35,302 (43.6%) | ||
| Age (year) | 54.5 | 16.2 | |
| cBMI (kg/m2) | 24.1 | 3.6 | |
| Preoperative Laboratory Test Result | |||
| Hemoglobin (g/dL) | 13.5 | 1.9 | |
| WBC (x1000/uL) | 6.7 | 2.4 | |
| Platelet (x1000/uL) | 246.3 | 71.0 | |
| Prothrombin time (INR) | 1.1 | 0.1 | |
| Aspartate aminotransferase (IU/L) | 24.6 | 104.0 | |
| Alanine aminotransferase (IU/L) | 24.4 | 41.6 | |
| Albumin (g/dL) | 4.3 | 0.5 | |
| Glucose (mg/dL) | 109.0 | 35.9 | |
| aPTT (sec) | 36.7 | 5.9 | |
| Blood urea nitrogen (mg/dL) | 14.7 | 7.4 | |
| Serum creatinine (mg/dL) | 0.9 | 0.8 | |
| Serum sodium (mmol/L) | 140.4 | 2.8 | |
| Serum potassium (mmol/L) | 4.2 | 0.4 | |
| Cormack grade | |||
| I,II | 67,771 (83.7%) | ||
| III, IV | 13,198 (16.3%) | ||
| Preoperative Comorbidity | |||
| Diabetes mellitus | 9,635 (11.9%) | ||
| Hypertension | 20,404 (25.2%) | ||
| Ischemic heart disease | 4,534 (5.6%) | ||
| Neurologic disease | 3,400 (4.2%) | ||
| ASA classification | |||
| I | 36,921 (45.6%) | ||
| II | 39,350 (48.6%) | ||
| III | 4,534 (5.6%) | ||
| IV, V, VI | 162 (0.2%) | ||
| Type of anesthesia | |||
| History of general anesthesia | 31,578 (39.0%) | ||
| General anesthesia | 54, 897 (67.8%) | ||
| Regional anesthesia | 10,607 (13.1%) | ||
| Monitored anesthesia care | 14,103 (17.4%) | ||
| Local anesthesia | 1,281 (1.6%) | ||
| Postoperative ICU admission | 3.7% | ||
SD, standard deviation; cBMI, conventional body mass index; WBC, white blood cell; ASA, American Society of Anesthesiologists; aPTT, activated partial thromboplastin time; ICU, intensive care unit.
One-year mortality with respect to preoperative cBMI, mBMI, and albumin
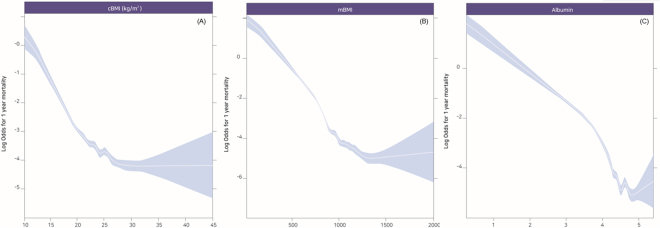
The (log) ORs of postoperative 1-year mortality were plotted against changes in preoperative cBMI, mBMI, and albumin (Fig. 2A–C). Table 2 shows the univariate logistic regression analysis for postoperative 1-year mortality in all patients. Supplemental Table 1 shows the results of individual multivariate logistic regression model without considering the interaction between cBMI and albumin. Lower cBMI (0.847, 95% confidence interval [CI]: 0.836–0.858), mBMI (0.995, 95% CI: 0.995–0.995), and albumin (0.230, 95% CI: 0.214–0.247) were associated with increased postoperative 1-year mortality separately. However, in the multivariate model as shown in Table 3, when interaction among the three variables were considered, only albumin was significantly associated with postoperative 1-year mortality [OR: 0.377, 95% CI (0.245–0.579), P < 0.001], whereas interaction (cBMI * albumin) and cBMI showed no significant association with postoperative 1-year mortality (P > 0.05).
Figure 2.
Log odds for postoperative 1-year mortality plotted against the changes of preoperative cBMI (A), mBMI (B), and albumin (C).
Table 2.
Univariate logistic regression analysis for 1-year mortality in all patients.
| Variable | 1-year death (n = 3,575) | P-value | Odds ratio (95% CI) | P-value |
|---|---|---|---|---|
| Operation | <0.001 | |||
| Non-cardiovascular surgery | 3,428 (95.9%) | Ref | ||
| Cardiovascular surgery | 147 (4.1%) | 3.14 (2.56–3.86) | <0.001 | |
| Gender | <0.001 | |||
| Male | 2,252 (63.0%) | Ref | ||
| Female | 1,323 (37.0%) | 0.44 (0.41–0.48) | <0.001 | |
| Age (yr), mean (SD) | 67.5 (13.9) | <0.001 | 1.06 (1.06–1.07) | <0.001 |
| cBMI (kg/m2), mean (SD) | 22.09 (3.70) | <0.001 | 0.83 (0.82–0.84) | <0.001 |
| <18.5 | 575 (16.1%) | Ref | ||
| 18.5–24.9 | 2,266 (63.4%) | 0.25 (0.23–0.28) | <0.001 | |
| 25–29.9 | 661 (18.5%) | 0.14 (0.12–0.16) | <0.001 | |
| ≥30 | 73 (2.0%) | 0.09 (0.07–0.12) | <0.001 | |
| mBMI, mean (SD) | 805.9 (223.7) | <0.001 | 0.99 (0.99–0.99) | <0.001 |
| Albumin (g/dL), mean (SD) | 3.6 (0.7) | <0.001 | ||
| <2.5 | 182 (5.1%) | Ref | ||
| 2.5–3.5 | 1,312 (36.7%) | 0.31 (0.25–0.39) | <0.001 | |
| >3.5 | 2,081 (58.2%) | 0.03 (0.03–0.04) | <0.001 | |
| Diabetes mellitus | 21.5% | <0.001 | 2.09 (1.90–2.30) | <0.001 |
| Hypertension | 33.3% | <0.001 | 1.51 (1.39–1.64) | <0.001 |
| Ischemic heart disease | 14.3% | <0.001 | 2.96 (2.65–3.31) | <0.001 |
| Neurologic disease | 10.0% | <0.001 | 2.65 (2.33–3.02) | <0.001 |
| ASA classification | ||||
| I | 382 (10.7%) | Ref | ||
| II | 2,066 (57.8%) | 5.23 (4.61–5.94) | <0.001 | |
| III | 1,047 (29.3%) | 26.23 (23.43–30.94) | <0.001 | |
| IV | 58 (1.9%) | 72.76 (50.77–104.28) | <0.001 | |
| V | 12 (0.3%) | 31.83 (8.94–113.40) | <0.001 | |
| VI | 10 (0.3%) | 79.58 (25.88–244.72) | <0.001 | |
| History of general anesthesia | 2,045 (57.2%) | <0.001 | 2.15 (1.99–2.32) | <0.001 |
| Type of anesthesia | <0.001 | |||
| General anesthesia | 2,313 (64.7%) | Ref | ||
| Regional anesthesia | 311 (8.7%) | 0.69 (0.60–0.79) | <0.001 | |
| Monitored anesthesia care | 944 (26.4%) | 1.47 (1.35–1.61) | <0.001 | |
| Local anesthesia | 8 (0.2%) | 8.99 (3.31–24.40) | <0.001 | |
| Postoperative ICU admission | 626 (17.5%) | <0.001 | 6.38 (5.73–7.10) | <0.001 |
SD, standard deviation; cBMI, conventional body mass index; mBMI, modified body mass index; ASA, American Society of Anesthesiologists; ICU, intensive care unit.
Table 3.
Multivariate logistic regression analysis for 1-year mortality regarding three preoperative variables (cBMI, mBMI, and albumin).
| Variable | Multivariate model | P-value |
|---|---|---|
| Odds Ratio (95% CI) | ||
| Type of operation | ||
| Non-cardiovascular surgery | Ref | |
| Cardiovascular surgery | 0.674 (0.526–0.857) | 0.002 |
| Gender | ||
| Male | Ref | |
| Female | 0.547 (0.50–0.60) | <0.001 |
| Age (year) | 1.03 (1.03–1.04) | <0.001 |
| Diabetes mellitus | 1.03 (0.92–1.15) | 0.621 |
| Hypertension | 0.71 (0.64–0.78) | <0.001 |
| Ischemic Heart Disease | 0.86 (0.74–0.99) | 0.035 |
| Neurologic Disease | 0.97 (0.83–1.14) | 0.738 |
| ASA class | ||
| I | Ref | |
| II | 2.83 (2.44–3.29) | <0.001 |
| III | 5.58 (4.66–6.70) | <0.001 |
| IV, V, VI | 11.37 (7.43–17.26) | <0.001 |
| History of general anesthesia | Ref (No) | |
| 1.228 (1.17–1.40) | <0.001 | |
| Type of anesthesia | ||
| General anesthesia | Ref | |
| Regional anesthesia | 0.53 (0.45–0.61) | <0.001 |
| Monitored anesthesia care | 1.09 (0.98–1.22) | 0.100 |
| Local anesthesia | 2.80 (0.70–10.10) | 0.131 |
| Postoperative ICU admission | Ref (No) | |
| 1.64 (1.42–1.90) | <0.001 | |
| cBMI (kg/m2) | 0.94 (0.88–1.01) | 0.087 |
| Interaction (cBMI * Albumin) | 0.98 (0.97–1.00) | 0.076 |
| Albumin | 0.38 (0.25–0.58) | <0.001 |
cBMI, conventional body mass index; mBMI, modified body mass index; IHD, ischemic heart disease; NUD, neurologic disease; ASA, American Society of Anesthesiologists; ICU, intensive care unit.
Comparison of preoperative cBMI, mBMI, and albumin according to ROC curve
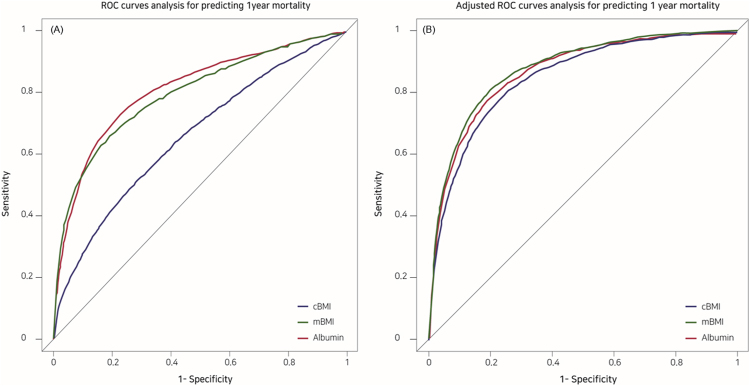
Figure 3A presents the ROC curves that show the risk for postoperative 1-year mortality with respect to preoperative cBMI, mBMI, and albumin. Figure 3B presents the ROC curves that predict the risk for postoperative 1-year mortality with respect to the preoperative cBMI, mBMI, and albumin after adjusting for the covariates (type of operation, gender, age, history of diabetes mellitus, hypertension, and ischaemic heart disease, ASA classification, history of general anesthesia, type of anesthesia, postoperative ICU admission). Prior to adjusting for the covariates, the AUC were cBMI Model = 0.659, mBMI Model = 0.797, albumin Model = 0.811, and overall Model = 0.820 (cBMI + albumin + cBMI * albumin); Table 4. After adjustment for the covariates, AUC was highest with the overall Model [cBMI + albumin + cBMI * albumin; 0.885, 95% CI (0.878–0.891)], followed by mBMI [0.879, 95% CI (0.873–0.886)], albumin [0.878, 95% CI (0.872–0.885)], and cBMI [0.853, 95% CI (0.846–0.860)]; Table 4. In addition, covariate adjusted AUC of mBMI was higher than cBMI (P < 0.001), while covariate adjusted AUC of mBMI and albumin was not significantly different in DeLong’s test (P = 0.558).
Figure 3.
ROC curves (A) and covariate-adjusted ROC curves (B) showing the risk of postoperative 1-year mortality with respect to preoperative cBMI, mBMI, and albumin.
Table 4.
Comparison of three preoperative factors (cBMI, mBMI, and albumin) by ROC curve.
| Variable | AUC | 95% Confidence Interval | ||
|---|---|---|---|---|
| Lower Limit | Upper Limit | |||
| cBMI Model | Not adjusted | 0.659 | 0.648 | 0.670 |
| Adjusted* (1) | 0.853 | 0.846 | 0.860 | |
| mBMI Model | Not adjusted | 0.797 | 0.787 | 0.807 |
| Adjusted* (2) | 0.879 | 0.873 | 0.886 | |
| Albumin Model | Not adjusted | 0.811 | 0.802 | 0.820 |
| Adjusted* (3) | 0.878 | 0.872 | 0.885 | |
| Overall Model** | Not adjusted | 0.820 | 0.811 | 0.829 |
| Adjusted* (4) | 0.885 | 0.878 | 0.891 | |
*Model: adjusted covariates (type of operation, gender, age, history of diabetes mellitus, hypertension, ischemic heart disease, neurologic disease, American society of anesthesiologists classification, history of general anesthesia, type of anesthesia, postoperative intensive care unit admission).
**Overall Model: cBMI + albumin + interaction (cBMI * albumin). DeLong’s test for two ROC curve, (1) vs (4): Z = 16.673, P < 0.001, (2) vs (4): Z = 8.753, P < 0.001, (3) vs (4): Z = 6.670, P < 0.001, (2) vs (3): Z = 0.586, P = 0.558.
Discussion
Our study showed that low cBMI, mBMI, and albumin are risk factors of postoperative 1-year mortality; however, when considering the interaction between cBMI and albumin, albumin was the only significant factor of postoperative 1-year mortality. Furthermore, adding cBMI to albumin does not increase the validity of the AUC of albumin in predicting postoperative 1-year mortality. These findings are important and meaningful, because they were obtained from the general population of more than 80,000 patients over five years at a 1,360-bed tertiary care hospital. Furthermore, this is the first study that used mBMI in general population cohorts; mBMI has been previously used for liver transplantation patients.
The first thing to consider when interpreting our findings is the superiority between serum albumin and cBMI in representing patients’ malnutrition status. A previous study reported that for patients on dialysis, albumin was an independent risk factor of operative mortality or morbidity, while cBMI was not3. In contrast, low cBMI was associated with mortality in patients undergoing cardiac surgery, while serum albumin was not14. Therefore, it is imperative to identify the index with a greater sensitivity for postoperative prognosis. We verified that albumin is a better index than cBMI in predicting postoperative prognosis. The reason for this superiority of albumin is that it reflects protein energy malnutrition unlike cBMI, which simply reflects the nutritional status of patients15. Low serum albumin is a measure of exacerbated immunity16 and a risk factor of increased postoperative infection17,18. Therefore, serum albumin is a better predictor of postoperative prognosis than cBMI.
We showed that mBMI has an equal predictive power to serum albumin with postoperative 1-year mortality. In addition, when interactions between albumin and cBMI were considered, albumin was the only independent predictor of postoperative 1-year mortality. It means that serum albumin was the most important factor for predicting 1-year mortality rather than cBMI. Previous studies have hypothesised that mBMI would be superior to cBMI in predicting mortality because it also measures the effects of fluid accumulation; therefore, mBMI would be more important for patients with ascites or edema2,10. Similarly, our study showed that mBMI is a stronger predictor of postoperative mortality is than cBMI in postoperative patients. We hypothesised that mBMI would be better than cBMI and serum albumin because mBMI has the characteristics of cBMI and albumin used in predicting postoperative prognosis. However, our study showed that low mBMI was not a better indicator of postoperative 1-year mortality than serum albumin, and adding cBMI to albumin does not increase the validity of the AUC of albumin in predicting postoperative 1-year mortality. Furthermore, mBMI was also not an independent risk factor when considering the interactions among variables. Based on this, we suggest that mBMI is a more appropriate index than only cBMI for use in patients undergoing surgery. Moreover, using mBMI is not recommended for predicting postoperative 1-year mortality rather than serum albumin; because serum albumin is the strongest index for predicting 1-year mortality after surgery.
In addition, there is an important consideration for using mBMI; unlike albumin levels or cBMI, which are currently the standard indices, there are no clear classification criteria for the normal ranges of mBMI. Tanaka et al. classified mBMI into 6 groups (<600, 600–800, 800–1000, 1000–1200, 1200–1400, and >1400)11. Suhr et al. reported that mortality was high in the liver transplantation group with mBMI <60012. However, no other study has classified mBMI more appropriately and investigated its association with mortality. Since mBMI involves a wider range of measurements (600–1400) than albumin or cBMI, the OR of mBMI seems smaller than that of cBMI or albumin. Considering this wide range of mBMI, further studies are warranted to identify appropriate classifications of mBMI for its clinical application.
Our study has a few limitations. First, the retrospective design may have led to selection or detection bias. However, this analysis based on all general surgical adult populations, included a large sample size of more than 80,000.
Second, the generalizability of our findings is limited because we reviewed the medical records at a single center. Third, serum albumin levels were checked at different time points across patients, so there may be differences in the measurements according to the disease severity. Finally, we could not take into consideration the surgical techniques used and postoperative care during the study period of 5 years. Nevertheless, our study is meaningful in that it is the first study that compared the association of mBMI, albumin, and cBMI with 1-year mortality in general surgical patients.
In conclusion, our study showed that low albumin was the strongest and independent risk factor of postoperative 1-year mortality compared to cBMI or mBMI. Adding cBMI to albumin does not increase the validity of the AUC of albumin in predicting postoperative 1-year mortality.
Methods
This study was a retrospective observational study and was approved by the Institutional Review Board at the SNUBH (Approval Number: B1705/395-106). The requirement for written informed consent was waived by the IRB, and this manuscript adheres to the applicable STROBE guidelines. The medical records of adult patients who were admitted to the SNUBH between January 2011 and December 2015 and underwent an elective or emergency surgical procedure were collected. Patients with inaccurate or incomplete medical records were excluded from the analysis. When one patient underwent two or more surgeries, only the medical record for the final surgery was included. The SNUBH is a 1,360-bed tertiary care academic hospital, where about 150 elective or emergency surgical operations are performed in 38 operating rooms on average every day. Furthermore, the hospital has been keeping and managing medical records since 2003 using an electronic medical system.
Definition of mBMI
We defined and calculated mBMI using the method suggested by a previous study10,11. The mBMI was calculated by multiplying cBMI (kg/m2) with preoperative serum albumin level (g/L). We used heights and weights taken before the surgery or at the time of admission and serum albumin level determined at the day closest to the surgery and at least 4 weeks prior to the surgery.
Data Collection and outcome
The following medical records were collected for the study: gender, age (year), height (cm), weight (kg), cBMI (kg/m2), preoperative blood laboratory test results [haemoglobin (g/dL), white blood cell count (*1000/µL), platelet count (*1000/µL), prothrombin time (INR), aspartate aminotransferase (IU/L), alanine aminotransferase (IU/L), glucose (mg/dL), activated partial thromboplastic time (sec), blood urea nitrogen (mg/dL), creatinine (mg/dL), sodium (mmol/L), potassium (mmol/L)], Cormack grade, [history of diabetes mellitus, hypertension, ischaemic heart disease, and neurologic disease], American society of anesthesiologists (ASA) classification, history of general anesthesia, type of operation and anesthesia, postoperative ICU admission, and death date.
Only preoperative blood laboratory test results obtained within 1 month before the surgery were used. The type of operation was classified into cardiovascular surgery and non-cardiovascular surgery. Cardiovascular surgery was defined as cardiac surgery or major vascular surgery involving cardiopulmonary bypass. All medical records were collected by a medical record technician of the SNUBH’s medical informatics team, who was blinded from the aims of this study; the main researchers were also blinded from the data until the final statistical outcomes were available. Moreover, the accurate dates of death of all patients (as of July 1, 2017) were obtained with approval from the Ministry of the Interior and Safety in Korea.
The primary outcome evaluated was the value of preoperative mBMI as a prognostic factor for postoperative 1-year mortality. Therefore, we compared mBMI with cBMI and albumin levels.
Statistical Methods
The baseline characteristics of all patients are presented as percentage (%) or mean and standard deviation. Continuous variables were analysed using the t-test and categorical variables were analysed using the chi-square test to evaluate the association of each variable with 1-year survival or death. The risk for postoperative 1-year mortality with respect to the three variables (cBMI, mBMI, and albumin) was analysed using restricted cubic spline, and the odds ratios (ORs) for postoperative 1-year mortality for each variable were computed using univariate and multivariate logistic regression analysis. We performed univariate logistic regression analysis to determine factors associated with postoperative 1-year mortality individually. We then selected covariates significant at P < 0.05 in the univariate logistic model to be included in the multivariate logistic regression analysis. At multivariate logistic regression analysis, we built two models considering the interaction among cBMI, mBMI, and albumin. Finally, the power of each of the three variables (cBMI, mBMI, and albumin) for explaining postoperative 1-year mortality was computed using the receiver operating characteristic (ROC) curve; areas under the curve (AUC) were compared and tested using the DeLong’s test. Additionally, we used ROC curves, after adjusting for the covariates that affected postoperative 1-year mortality, to compute adjusted AUC for each variable. The covariates adjusted for in the AUC were selected from the univariate logistic regression analysis based on the criterion of P < 0.05.
The power of the sample size of our study was verified by the PASS 15 program. From the area under the curve (AUC) of 0.8, to detect the difference of 0.05 of the AUC from the event rate of 4.4%, 15,000 patients were adequate to yield 80% power and an alpha error of 0.05. Therefore, our study was determined to have sufficient power to detect a difference of 0.05 from an AUC of 0.8 in predicting 1-year mortality. Statistical analyses were performed using IBM SPSS (Version 23.0; IBM Corp., Armonk, NY, USA) and R (Version 3.3.2 with R packages; http://www.R-project.org) software, with statistical significance set at P < 0.05.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
Electronic supplementary material
Author Contributions
Tak Kyu Oh contributed to the study design, and drafted the first manuscript; Jaebong Lee analysed the data. Jung-Won Hwang, Sang-Hwan Do, Jin Hee Kim, Young-Tae Jeon and Kooknam Kim contributed to the acquisition of data and provided critical revision of the manuscript. In-Ae Song contributed to the study design and provided critical revision of the manuscript. All authors read and approved the final manuscript.
Competing Interests
The authors declare no competing interests.
Footnotes
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-22886-6.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Norman K, Pichard C, Lochs H, Pirlich M. Prognostic impact of disease-related malnutrition. Clin Nutr. 2008;27:5–15. doi: 10.1016/j.clnu.2007.10.007. [DOI] [PubMed] [Google Scholar]
- 2.Pikul J, Sharpe MD, Lowndes R, Ghent CN. Degree of preoperative malnutrition is predictive of postoperative morbidity and mortality in liver transplant recipients. Transplantation. 1994;57:469–472. doi: 10.1097/00007890-199402150-00030. [DOI] [PubMed] [Google Scholar]
- 3.Chan M, Kelly J, Batterham M, Tapsell L. Malnutrition (subjective global assessment) scores and serum albumin levels, but not body mass index values, at initiation of dialysis are independent predictors of mortality: a 10-year clinical cohort study. J Ren Nutr. 2012;22:547–557. doi: 10.1053/j.jrn.2011.11.002. [DOI] [PubMed] [Google Scholar]
- 4.Hendifar A, et al. Influence of Body Mass Index and Albumin on Perioperative Morbidity and Clinical Outcomes in Resected Pancreatic Adenocarcinoma. PLoS One. 2016;11:e0152172. doi: 10.1371/journal.pone.0152172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Matsuoka, K. et al. Significance of Body Mass Index for Postoperative Outcomes after Lung Cancer Surgery in Elderly Patients. World J Surg, 10.1007/s00268-017-4142-0 (2017). [DOI] [PubMed]
- 6.Wu N, et al. The prognostic influence of body mass index, resting energy expenditure and fasting blood glucose on postoperative patients with esophageal cancer. BMC Gastroenterol. 2016;16:142. doi: 10.1186/s12876-016-0549-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Brunaud L, et al. Predictive factors for postoperative morbidity after laparoscopic adrenalectomy for pheochromocytoma: a multicenter retrospective analysis in 225 patients. Surg Endosc. 2016;30:1051–1059. doi: 10.1007/s00464-015-4294-7. [DOI] [PubMed] [Google Scholar]
- 8.Caras RJ, Lustik MB, Kern SQ, McMann LP, Sterbis JR. Preoperative Albumin Is Predictive of Early Postoperative Morbidity and Mortality in Common Urologic Oncologic Surgeries. Clin Genitourin Cancer. 2017;15:e255–e262. doi: 10.1016/j.clgc.2016.09.008. [DOI] [PubMed] [Google Scholar]
- 9.Puvanesarajah V, et al. Poor Nutrition Status and Lumbar Spine Fusion Surgery in the Elderly: Readmissions, Complications, and Mortality. Spine (Phila Pa 1976) 2017;42:979–983. doi: 10.1097/BRS.0000000000001969. [DOI] [PubMed] [Google Scholar]
- 10.Suhr O, Danielsson A, Holmgren G, Steen L. Malnutrition and gastrointestinal dysfunction as prognostic factors for survival in familial amyloidotic polyneuropathy. J Intern Med. 1994;235:479–485. doi: 10.1111/j.1365-2796.1994.tb01106.x. [DOI] [PubMed] [Google Scholar]
- 11.Tanaka T, Renner EL, Selzner N, Therapondos G, Lilly LB. The impact of obesity as determined by modified body mass index on long-term outcome after liver transplantation: Canadian single-center experience. Transplant Proc. 2013;45:2288–2294. doi: 10.1016/j.transproceed.2012.11.009. [DOI] [PubMed] [Google Scholar]
- 12.Suhr OB, Ericzon BG, Friman S. Long-term follow-up of survival of liver transplant recipients with familial amyloid polyneuropathy (Portuguese type) Liver Transpl. 2002;8:787–794. doi: 10.1053/jlts.2002.34386. [DOI] [PubMed] [Google Scholar]
- 13.Yamamoto S, et al. Liver transplantation for familial amyloidotic polyneuropathy (FAP): a single-center experience over 16 years. Am J Transplant. 2007;7:2597–2604. doi: 10.1111/j.1600-6143.2007.01969.x. [DOI] [PubMed] [Google Scholar]
- 14.Rapp-Kesek D, Stahle E, Karlsson TT. Body mass index and albumin in the preoperative evaluation of cardiac surgery patients. Clin Nutr. 2004;23:1398–1404. doi: 10.1016/j.clnu.2004.06.006. [DOI] [PubMed] [Google Scholar]
- 15.Blackburn GL, Harvey KB. Prognostic strength of nutritional assessment. Prog Clin Biol Res. 1981;77:689–697. [PubMed] [Google Scholar]
- 16.Gleeson MW, Dickson RC. Albumin gains immune boosting credibility. Clin Transl Gastroenterol. 2015;6:e86. doi: 10.1038/ctg.2015.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shi H, Na Q, Zhang X, Jiang X. Correlations between the levels of acute infection markers and serum albumin in elderly patients with hip fracture. Aging Clin Exp Res. 2017;29:435–441. doi: 10.1007/s40520-016-0585-7. [DOI] [PubMed] [Google Scholar]
- 18.Yuwen P, et al. Albumin and surgical site infection risk in orthopaedics: a meta-analysis. BMC Surg. 2017;17:7. doi: 10.1186/s12893-016-0186-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during the current study are available from the corresponding author on reasonable request.