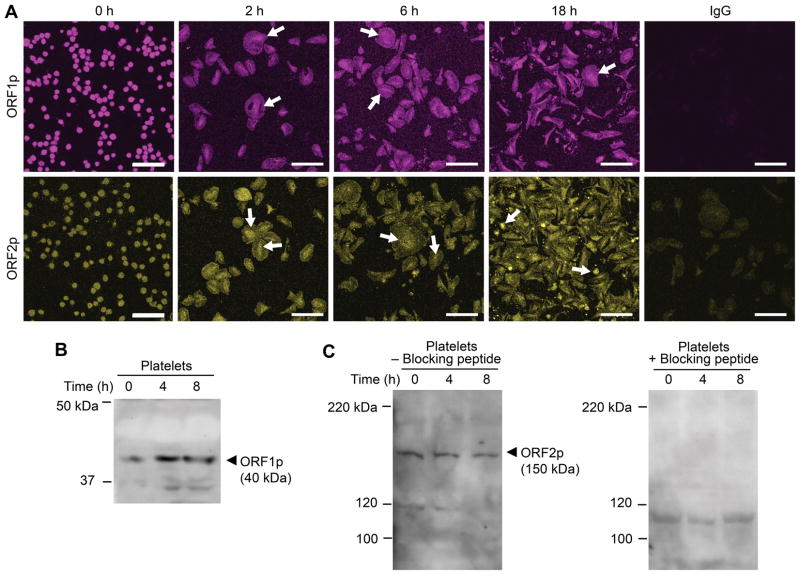
Figure 2. Human platelets express ORF1 and ORF2 protein.
(A) Freshly isolated platelets were fixed in suspension immediately (baseline, 0 h) or adhered to immobilized fibrinogen in the presence of thrombin for 2, 6, or 18 hours. Immunofluorescence staining with an anti-L1 ORF1p (top row, magenta stain), and anti-L1 ORF2p (bottom row, yellow stain) antibody, respectively, demonstrates expression of both L1-encoded proteins ORF1p and ORF2p in platelets, irrespective of presence or absence of thrombin stimulation. White arrows indicate the time-dependent concentration of L1 proteins in central cell areas, rich in mRNA and ribosomes. The isotype specific IgG control is shown on the far right (scale bars = 10 μm). This figure is representative of 3 independent experiments. (B) Immunoblot analysis of platelet lysates using the anti-L1 ORF1p antibody. Freshly isolated platelets were immediately fixed in suspension (t=0 h) or allowed to adhere to immobilized fibrinogen in the presence of thrombin for 4 or 8 hours. The cells were then lysed and proteins separated by SDS-PAGE electrophoresis. (C) Immunoblot analysis of platelet lysates using the anti-L1 ORF2p antibody. Platelets were treated as in B, however the samples were split and processed in parallel in the absence (left panel) or presence (right panel) of an ORF2p quenching peptide. Presence of the blocking peptide prevents the detection of L1 ORF2p with the anti-L1 ORF2p antibody. Panels B and C are representative of 4 and 5 independent experiments, respectively.