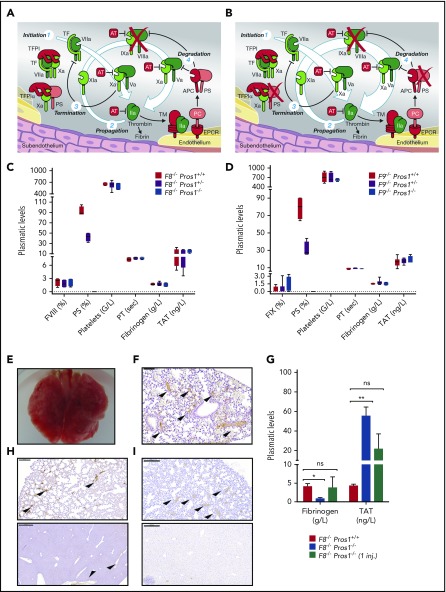
Figure 1.
Loss of X-ase activity rescues Pros1−/−mice. (A) Schematic model of thrombin generation in hemophilic condition. One of the major coagulation complexes is the intrinsic tenase (X-ase) complex.45 X-ase comprises activated FIX (FIXa) as the protease, activated FVIII as the cofactor, and factor X (FX) as the substrate. Although the generation or exposure of TF at the site of injury is the primary event in initiating coagulation via the extrinsic pathway, the intrinsic pathway X-ase is important because of the limited amount of available active TF in vivo and the presence of TFPI, which when complexed with activated FX (FXa), inhibits the TF/activated factor VII (FVIIa) complex46 (Figure 1A). Thus, sustained thrombin generation depends upon the activation of both FIX and FVIII47 (Figure 1A). This process is amplified because FVIII is activated by both FXa and thrombin, and FIX is activated by both FVIIa and activated factor XI (FXIa), the latter factor being previously activated by thrombin. Consequently, a progressive increase in FVIII and FIX activation occurs as FXa and thrombin are formed. (B) The experimental approach to enhancing thrombin generation in severe hemophilia A and B by targeting Pros1. (C-D) Murine model validation and evaluation of DIC hematologic parameters in hemophilic adult mice with and without Pros1 deficiency: PS (antigenic), FVIII (coagulant activity), or FIX (coagulant activity) plasma levels in F8−/−Pros1+/+, F8−/−Pros1+/−, and F8−/−Pros1−/− adult mice (C) and in F9−/−Pros1+/+, F9−/−Pros1+/− and F9−/−Pros1−/− adult mice (D) (n = 5 per group); platelets (n = 7 per group), fibrinogen (n = 8 per group), PT (n = 6 per group), and TAT (n = 6 per group) in hemophilia A group (C) and platelets (n = 5 per group), fibrinogen (n = 4 per group), PT (n = 4 per group), and TAT (n = 4 per group) in hemophilia B group (D). (E-F) Macroscopic image of lungs from F8−/−Pros1−/− mice 24 hours after a single intravenous injection of 2 U/g recombinant FVIII (Advate) infusion (E) and corresponding microscopic evaluation of fibrin clots (indicated by arrowheads) in lung section (F). (G-I) Recombinant FVIII (Advate) administration in F8−/−Pros1+/+ and F8−/−Pros1−/−: plasma levels of fibrinogen and TAT at 24 hours after 5 injections of 0.3 U/g Advate IV (injection time points: 1 hour before catheter insertion and 1 hour, 4 hours, 8 hours, and 16 hours after catheter insertion) (n = 3) (G, open and solid columns) and 24 hours after a single IV injection in F8−/−Pros1−/− (n = 3) (G, dashed column), and representative immunohistochemistry allowing the detection of fibrin clots (indicated by arrowheads) in lungs and liver sections in F8−/−Pros1−/− 24 hours after 0.3 U/g repeated IV injections of Advate (H) and after a single IV injection of 0.3 U/g Advate IV (I). All data are expressed as means ± SEM. *P < .05; **P < .005. AT, antithrombin; EPCR, endothelial protein C receptor; inj., injection; ns, not significant. Scale bars: 200 μm.