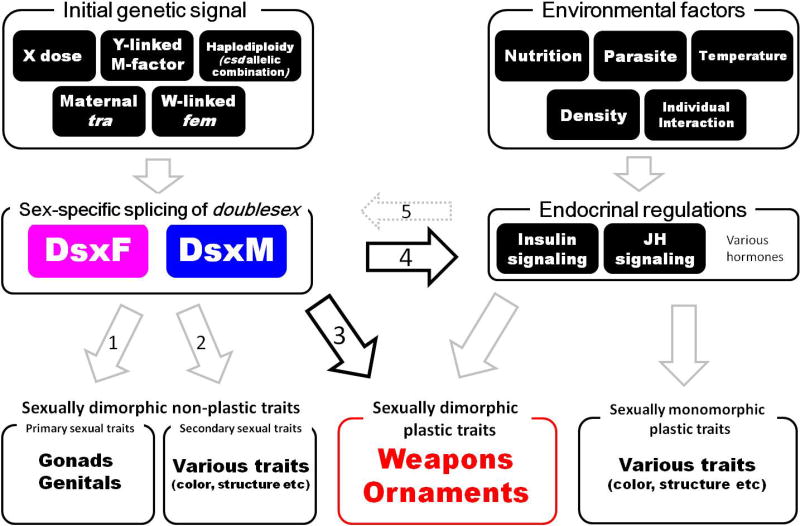
Figure 1.
Sexually dimorphic plastic trait(s) develop downstream of both sexual regulation (mediated by Dsx) and environmental regulation (mediated by hormonal pathway(s)).
A variety of initial genetic signals determine the sex through upstream initiation of the sex determination cascade in insects. Examples include, X chromosome dose in Drosophila melanogaster [72], maternal input of transformer (tra) mRNA in Nasonia vitripennis [73], W-linked fem piRNA in Bombyx mori [74], the Y-linked M-factor, Mdmd in Musca domestica [75], and haplodiploidy and the csd allelic combination in Apis mellifera [76]. However, in all studied insects, these different determination signals converge on the conserved transcription factor Dsx, which functions as a downstream master switch gene for both sex determination and differentiation (see text). Various environmental factors can affect an animal’s physiological state, typically via hormonal regulation, and these endocrine signals the expression of various plastic traits. Accordingly, many traits that are both sex-specific and plastic are also under the control of this endocrine regulation, and thus these endocrine signals interact with Dsx in a variety of ways to ultimately generate trait expression that is sensitive not only to an organism’s sex, but also their condition.
Arrows 1–4 indicate various outcomes of Dsx regulation across insects. Dsx can regulate sexually dimorphic traits that do not exhibit plastic responses, such as yolk protein for ovary development and the gene lozenge during Drophila female genital disk development ([77], arrow 1), or through regulation of the gene bric-a-brac during development of abdominal pigmentation in Drosophila ([78], arrow 2). Critically to this review, Dsx can regulate the expression of sexually dimorphic plastic traits in two ways, either directly through changes in expression levels of dsx as in dung beetle horns ([33], arrow 3), or through sex-specific splice variants regulating responsiveness to endocrine signals ([19], arrow 4). It is possible, as suggested by the morph-specific expression patterns of Dsx target genes in dung beetle horns [33] that dsx may itself be regulated by endocrine signals (arrow 5), although there is no direct evidence of this relationship. It is important to note that, while the specific genes targeted by Dsx during the regulation of sexually dimorphic plasticity are unknown, evidence from next-generation sequencing experiments have suggested that developmental toolkit genes such as hedgehog may be directly regulated by Dsx expression level (arrow 3), and it is likely that JH signaling genes may also be regulated by Dsx (arrow 4). However, in both cases these predictions need to be confirmed through both functional experiments and through the use of techniques such as gSELEX, which allow for more targeted investigation of the binding sites regulated by Dsx, as well as allowing the investigation of genes that are regulated by Dsx that are not involved in the development of sexually dimorphic plastic traits (arrows 1 and 2).