Abstract
This work describes the formulation of a functional yogurt-like product based on fermented maize with added phytosterols and its oxidative stability during cold storage. The technological challenge was to stabilize 3.5% esterified phytosterols (between 2 and 3 g of free sterols) in a low-fat emulsion and to preserve the obtained product throughout processing and storage. The natural bioactive compounds: lutein, zeaxanthin, β-cryptoxanthin, β-carotene and γ-tocopherol were detected in the yogurt, and remained stable during 12 days of refrigeration. Higher content of C18:1 n-9 and C18:3 n-3 (six and ninefold, respectively) were obtained in samples with phytosterols. This was desirable from a nutritional point of view, but at the same time it induced lipid oxidation that was 1.4-fold higher in the product with phytosterols than in the controls. The use of a multivariate approach served to find descriptors which were related to treatments, and to explain their behavior over time.
Electronic supplementary material
The online version of this article (10.1007/s13197-018-3102-5) contains supplementary material, which is available to authorized users.
Keywords: Phytosterols, Maize-yogurt, Antioxidants, Volatiles
Introduction
Cereals contribute as much as 70% of the energy intake in developing countries. Therefore, they may be taken into account for the design of new functional foods within a food security context. These foods may prevent chronic diseases which are rising as a consequence of the intake of extra calories, coming mainly from cheaper foodstuffs (Kearney 2010). Obesity, metabolic syndrome, and cholesterol associated illnesses may be a direct consequence of an unbalanced high-caloric diet.
Concerning hypercholesterolemia, it has been shown that an intake of 1–3 g/day of vegetable sterols can produce a 10–15% reduction in LDL-cholesterol (Ling and Jones 1995; Hallikainen and Uusitupa 1999). They diminish the incorporation of meal cholesterol into the chylomicrons (Amiot et al. 2011) and moreover, milk with phytosterols has been used to control hypertriglyceridemia in children (Blumenfeld Olivares et al. 2015). However, the typical Western diet contains only 150–400 mg/day and does not reach desired levels to obtain the aforementioned health benefits. Consequently, supplemental doses are required. The American Health Association recommends 2 g per day (reviewed in Rondanelli et al. 2013).
Incorporation of phytosterols into food has been typically associated with Western dietary products, mainly vegetable margarines, fermented milk and yogurt, cereal bars, and processed meat products. However, hypercholesterolemia has become a worldwide concern, affecting also developing countries where the population does not have access to these types of foods. Therefore, it is of great importance that these products become accessible across all demographic and social conditions (Gorton et al. 2010).
For this purpose, a strategy may be the fortification of fermented staple meals with phytosterols for making cholesterol-lowering products. But at the same time, the addition of high amounts of vegetable lipids can affect the oxidative stability of the foods producing undesirable changes in their properties.
Another important aspect of cereal fermentation is the biopreservation to maintain food safety by pathogen inhibition, especially by lactic acid production (Kohajdova 2015). In addition, some maize and sorghum based fermented foods have exhibited antioxidant capacity, and this could be used to improve its preservation (Oboh and Amusan 2009).
Alternatively, foods containing live active bacteria may exhibit probiotic activity (Gullón et al. 2015). For that reason, this product may be accepted not only by traditional consumers but it may gain new consumers who appreciate the properties of newly introduced foods, as demonstrated for Akpan, a traditional beverage made from a fermented cereal gruel called “ogi” (Akissoé et al. 2014).
Therefore, the aim of the present work was to obtain a functional fermented maize product enriched with phytosterols, which is similar to the African-indigenous food, but with the cholesterol lowering benefits (between 2 and 3 g of free phytosterols per daily portion). At the same time, we describe the oxidation process in the product regarding the fatty acids composition and the natural antioxidants such as vitamin E and maize carotenoids to ensure the presence of bioactive molecules throughout storage.
Research into this aspect constitutes a challenge to harnessing a traditional food, making this type of product supportive of a healthy diet for a broad spectrum of consumers that appreciate low-fat, gluten-free and phytosterols-enriched fermented food with lactic acid bacteria.
Materials and methods
Preparation of maize yogurt
Experimental maize yogurt was prepared as indicated by Sacca et al. (2012) with esterified phytosterols. Briefly, 200 g yellow maize (Zea mays var. Flynn, product from France) was soaked in distilled water for 15 h at 50 °C. Samples were wet milled at 5000 rpm (Grindomix GM200, France) for 1 min. The obtained paste was mixed up with 3 volumes of distilled water and wet filtered through a 315-mesh sieve.
The liquid was recovered and skimmed milk powder (commercial, 1.5% fat) and sugar were added to final proportions of 3 and 15% respectively. These ingredients have been shown to improve the acceptability of the product (Akissoé et al. 2014).
Esterified phytosterols (Advasterol™ Ester, AOM, Argentina) were added to a final proportion of 2.2 g of free phytosterols/125 g product (P series). Non-supplemented samples remained as controls (C). The preparations were pasteurized at 65 °C for 20 min. After cooling at 37 °C, Lactobacillus casei (CNCM I-4592, Lesaffre Company, France) was added in a proportion of 5.6 × 106 CFU/g. Batch fermentation was performed in an oven at 37 °C for 20 h.
After fermentation, 1/5 (100 g) of gruel (ogi) was taken apart, and the rest was cooked at 90 °C under continuous stirring for 10 min. Thereafter, the uncooked portion was added, well mixed and the individual samples were placed into 125 mL glass containers, capped aseptically and stored in darkness at 4 °C to simulate retail conditions during 2; 5; 9 and 12 days. Samples were kept at − 20 °C after storage.
Phytosterols
All standards and chemicals were obtained from Sigma-Aldrich, Saint-Quentin Fallavier, France (otherwise stated). Phytosterols were extracted as indicated in Slavin and Yu (2012) with modifications. Briefly, 0.5 g of each preparation were mixed with methanol: methyl tert-butyl ether (MTBE) (20:80) and homogenized at a speed of 6 m/s with a FastPrep®-24 (MP Biomedicals, Santa Ana, USA). Thereafter, samples were placed in glass tubes with 2 mL of ethanol 1% butylated-hydroxytoluene and 1.8 mL of 12N KOH and saponified for 2 h at 70 °C. Samples were extracted twice with 10 mL of n-hexane and after evaporation of the organic phase, were suspended in methanol: MTBE (20:80) and injected in a reverse-phase HPLC system (Agilent 1200 series Agilent, Massy, France) fit to an Uptisphere-C8 column of 250 mm × 4.6 mm id, 5 µm (Interchim, Montluçon, France). The mobile phase was methanol:water (92:8) at 1 mL min −1 with UV detection at 210 nm. RP-HPLC allows the accurate quantification of phytosterols without further derivatization, as required for the classical GC methodology (Slavin and Yu 2012). The disadvantage is that without a mass detection, campesterol and stigmasterol are not identified separately. But for the purpose of this work, they count as total free phytosterols.
pH
Aliquots of 0.5 g of each sample were mixed with an equal volume of distilled water at 20 °C, according to the lineouts of the normative for pH determination (NMX-F-317-S-1978) in foods.
The electrode was calibrated at pH 7 and 4 immediately before the measurement and pH from each stage of the process was recorded.
Fat, proteins and fatty acids composition
Total fat was determined by Soxhlet extraction with petroleum ether and proteins were determined by the Kjeldahl method. Dry matter was determined by heating to constant weight and carbohydrates by difference (AOAC 1980). The fatty acid methyl esters (FAME) were prepared according to Descalzo et al. (2005), and measured using a gas chromatograph (Varian CP-3800) with a flame ionization detector, using a capillary column CP-Sil 88 100 m × 0.25 mm × 0.20 µm, Varian CP7489, (Varian, California, USA), with nitrogen as carrier gas. The split ratio was 1:10. The column temperature was programmed at 70 °C for 4 min, increased from 70 to 170 °C at 13 °C min −1, and then increased from 170 to 200 °C at 1 °C min −1.
The injector and detector temperature was 250 °C. Individual fatty acids were identified by comparing relative retention times with standards (PUFA-2 Animal Source and Grain Fatty acid Methyl Ester Mix, Supelco, Pennsylvania, USA). The analytical results are expressed as percentages of total fat extracted.
The peroxidation index (PI) was calculated as follows (Witting and Horwitt 1964): (%monoenoic acid * 0.025) + (%dienoic acid * 1) + (%trienoic acid * 2) + (%pentaenoic acid * 6).
Carotenoids and tocopherols
The extraction procedure was adapted from Rossetti et al. (2010). Aliquots of 0.5 g were placed in a glass tube, with 2 mL of pyrogallol 1% in ethanol and mixed with vortex for 1 min, saponification with 12N KOH for 30 min at 70 °C. Samples were extracted twice with 5 mL n-hexane, evaporated and injected in an HPLC system Ultimate 3000 (Dionex, Tokyo, Japan) with a fluorimetric detector. For separation, an HALO® C18 coreshell column (250 × 4.6 mm i.d., 5 µm, Interchim, Montluçon France) was used with ethanol/methanol (60:40) at a flow of 1 mL/min. Fluorescence detection was set at 296 nm (excitation) and 330 nm (emission). Reverse-phase HPLC combined with an isocratic mobile-phase allowed a better fluorescent detection of low quantities of tocopherols as compared with a direct phase system, consequently the method of saponification and RP-HPLC was preferred (Lee et al. 2017).
Carotenoids were analyzed by reverse-phase HPLC, as described in Dhuique-Mayer et al. (2016) using an Agilent 1100 series system (Agilent, Massy, France), equipped with a YMC C30 column 250 × 4.6 mm i.d. 5 µm (YMC, Kyoto, Japan). The mobile phases used were H2O as eluent A, methanol as eluent B and MTBE (Methyl tert-butyl ether) as eluent C, at 1 mL min−1 and an injection volume of 20 µL. The following gradient program was used: 0-5 min, 40% A, 60% B; 5–10 min, 20% A, 80% B (initial condition); 10–60 min, 4% A, 81% B, 15% C; 60–71 min, 4% A, 11% B, 85% C; 71–72 min 100% B, and back to the initial condition. Absorbance was monitored using an Agilent 1100 photodiode array detector.
Calibration and recovery
Calibration curves with commercial standards (Sigma-Aldrich, France) were performed. Standards were diluted in methanol: MTBE (20:80) for carotenes and sterols and ethanol for tocopherols, within the following ranges: from 0.001 to 0.05 mg/mL for campesterol, β-sitosterol and stigmasterol and detection at 210 nm. From 0.0001 to 0.005 mg/mL for β-carotene, lutein, β-cryptoxhantin and zeaxanthin (detection at 445 nm), and 0.00005 to 0.01 mg/mL for α- and γ- tocopherol (detection by fluorescence at 296–330 nm). The peak area (Y), concentration (X, mg/20 μl) and the mean of three replicates ± standard deviation were calculated. The linearity for the bioactive compounds was good with r2 > 0.99.
Compounds chosen for recovery were: β-sitosterol (0; 0.01; 0.02; 0.04 mg/mL), β-carotene (0; 0.0002; 0.0005 and 0.001 mg/mL) and α-tocopherol (0; 0.001; 0.002 and 0.005 mg/mL). Control (C) samples were spiked with the three concentrations (or none as control) of each compound by triplicate and then extracted as described above, and the recovery was calculated as the mean concentration ± standard deviation. All assays were performed within the linear range of quantification. The mean recovery was 95.1 ± 4.8% for β-sitoesterol; 101 ± 4.6% for α-tocopherol and 112 ± 5.2% for β-carotene.
Lipid oxidation TBARS (Thiobarituric acid reactive substances) assay
The methodology was adapted from Rossetti et al. (2010). Briefly, 0.5 g of each sample was added 0.4 mL ethanol:butylated hydroxytoluene. (BHT, 0.1%), vortexed and added to 0.4 mL of TBA (thiobarbituric acid) solution prepared in TCA (trichloroacetic acid), to final concentrations of 0.01 M TBA and 10% TCA respectively.
The samples were incubated at 70 °C for 1 h. Then, they were centrifuged al 6000×g for 10 min, filtered through PVDF syringe filters (pore 0.45 µ) and read in the spectrophotometer (Specord® Analytik Jena AG, Germany) at 450 and 532 nm. The values were compared with a calibration curve of TEP (1,1,3,3,tetraethoxypropane) expressed in moles. A stock solution of 1 × 10−3 M was prepared in distilled water and standard solutions were prepared in the range from 2.5 × 10−9 to 1.25 × 10−8 mol/mL. A turbidity blank was prepared for each sample with the sample mixed with TCA only, and was subtracted from the total optical density of colored samples.
TBARS were calculated as follows
where OD is the optical density at 532 nm, TEP is used as standard for malonaldehyde (MDA) equivalents 1 mol TEP = 1 mol MDA reacting with TBA; 72.06 g/mol is the molecular weight of malonaldehyde, with a recovery of 85% for the system and 1 · 106 conversion mg/kg.
Ferric reducing ability power (FRAP)
FRAP values were obtained by comparing the absorbance change at 593 nm in test reaction mixtures with FeIII and 2,4,6-Tris(2-pyridyl)-s-triazine (TPTZ) for ferric reducing activity. The samples (1 g) were centrifuged at 6000×g for 10 min, aliquots of 41.5 µL were placed into 1 mL of the FRAP reagent, and incubated for 15 min at 30 °C (adapted from Rossetti et al. 2010). Absorbance was recorded with a spectrophotometer (Specord® Analytik Jena AG, Jena, Germany); and the values obtained were compared with a calibration curve performed with a TROLOX standard (6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid) solution between 0 and 1000 µm. The FRAP activity was related to the concentration of reducing power in each sample and expressed in μM of TROLOX equivalents.
Volatile compounds determination
Solid Phase Micro Extraction (SPME) was carried out over samples of 0.5 g, sealed in 10 mL vials with 15 min incubation at 60 °C and 30 min trapping with a tri-phased fibre 50/30 µm (DVB, Carboxen, PDMS, SUPELCO, Bellefonte, PA, USA).
A tandem gas chromatograph 6890/MSD 5973N/Gerstel Multipurpose Sample MPS-2 was used for volatiles compounds analysis (Agilent Technologies, Palo Alto, USA). Injection was carried out in splitless mode at 250 °C for 60 s for desorption. Separation was performed on a DB-WAX polar column (30 m × 0.25 mm × 0.25 µm, J&W, Palo Alto, USA). The temperature program was: 40 °C for 1 min; 3 °C min −1 from 40 to 170 °C, then 10 °C min −1 up to 240 °C and held for 10 min. Mass spectral were recorded in EI + mode at 70 eV within 40–300 Da. Source temperature was 250 °C with hydrogen as carrier gas at 1.2 mL min−1 at constant flow.
Vials of butanol solution at 0.01 mg/L were injected, every five samples, to standardize analysis and to compensate fiber aging for more reliable qualitative results. A quantification with internal standards was not performed, rather a comparison between treatments was done in a semi-quantitative way. Clustering analysis was applied to illustrate the whole volatile picture. For technical reasons day 12 is missing. Data were acquired and processed with Mass Hunter B06 analysis (Agilent Technologies, Palo Alto, USA). Volatile compounds were identified through their mass spectra (NIST version 2011; National Institute of Standards and Technology, Gaithersburg, MD, USA) and their Retention Indices (RI) when available. RIs were determined by linear interpolation following the injection of n-alkanes (C8–C20), Sigma-Aldrich (St. Louis, MO, USA).
Statistics
The data are presented as mean ± standard deviation for three replicates. Statistical analysis was conducted using one way analysis of variance (ANOVA). One-way ANOVA was used for the phytosterols content and repeated measurements was applied for other individual variables, followed by Tukey’s multiple range tests to determine any differences between group means at the (p < 0.05) level.
Volatiles were transformed to their z scores for the heat map:
where x is the measured variable, µ the mean and σ the standard deviation.
Each set of variables was submitted to a factorial discriminant analysis (FDA) with stepwise selection method. The selection method made it possible to consider all dependent variables to be within the model from the beginning, and after each iteration step, non-discriminant variables were selectively excluded. The selected variables were correlated using the Partial Least Squares (PLS) multivariate approach in order to describe the interaction between pro- and antioxidants, and their relationship with the samples during storage. All data were analyzed using software XLSTAT®, version 2015.5.01.23654 (Addinsoft, Paris, France).
Results and discussion
Free phytosterols content in functional yogurt-like maize product
The product obtained was semi-liquid, yellowish, with a smooth creamy texture and a sour yogurt taste as expected for this type of food (Sacca et al. 2012).
At first sight, no differences had been perceived between control (C) and phytosterol added (P) samples after processing (Fig. 1). However, during the formulation the product was not homogeneous; fatty droplets were found on the surface of P, but not on C samples. In order to homogenize the product, a 15-min heating and mixing step was applied; with a temperature shift between 37 °C and 90 °C. After cooking for 10 min at 90 °C, it acquired a uniform texture probably due to the emulsification and trapping of oily drops into the gelatinized starch.
Fig. 1.

Aspect of maize yogurt-like product with (P) and without (C) addition of esterified phytosterols
The chemical composition and oxidative status of P and C products is presented in Table 1.
Table 1.
Chemical composition of maize fermented product with (P) or without (C) esterified phytosterols
| Variable | Treatment C | Treatment P | p |
|---|---|---|---|
| β-sitosterol (g/kg) | 0.062 ± 0.005 | 8.230 ± 0.793 | < 0.001 |
| Campesterol + Stigmasterol (g/kg) | 0.022 ± 0.003 | 14.137 ± 0.924 | < 0.001 |
| Zeaxantine (mg/kg) | 0.349 ± 0.018 | 0.348 ± 0.030 | NS |
| Luteine (mg/kg) | 0.463 ± 0.021 | 0.440 ± 0.031 | NS |
| β-carotene (mg/kg) | 0.024 ± 0.001 | 0.029 ± 0.003 | 0.018 |
| β-cryptoxantine (mg/kg) | 0.034 ± 0.001 | 0.026 ± 0.002 | 0.003 |
| β + δ tocopherol (mg/kg) | ND | 3.168 ± 0.411 | – |
| γ-tocopherol (mg/kg) | 1.952 ± 0.450 | 5.662 ± 1.704 | < 0.001 |
| α-tocopherol (mg/kg) | ND | ND | – |
| TBARS (mg eq MDA/kg) | 0.112 ± 0.029 | 0.156 ± 0.014 | 0.034 |
| FRAP (μM) | 429.1 ± 71.11 | 372.6 ± 58.65 | NS |
| Protein (%) | 1.41 ± 0.05 | 1.45 ± 0.05 | NS |
| Fat (%) | 1.21 ± 0.02 | 4.18 ± 0.09 | < 0.001 |
| Carbohydrates (%) | 14.60 ± 1.51 | 15.40 ± 0.97 | NS |
| Water (%) | 83.0 ± 6.90 | 79.1 ± 5.98 | NS |
p indicates significant differences obtained by ANOVA
NS non-significant, ND not detected
According to the results, one portion of 125 g of P yogurt may contain 2.70 g of total free phytosterols (β-sitosterol, campesterol and stigmasterol) which remained stable during storage (supplementary Fig. 1). This is consistent with the cholesterol lowering recommendations, as reviewed by the European Atherosclerosis Consensus Panel in Gylling et al. (2014). This concentration is higher than the one found in some commercial products of similar characteristics (Saraiva et al. 2011; Semeniuc et al. 2015). Endogenous phytosterols’ content in C samples is low to exhibit a functional cholesterol-lowering action and should be enriched in order to design functional foods. P and C also contained 1.4% protein and 15% carbohydrates. The average fat content was 1.2 and 4.8% of total fat for C and P series respectively. Micronutrients such as zeaxanthin, lutein, β-carotene and β-cryptoxanthin, were also present in trace amounts in both P and C. On the other hand, P samples were richer in tocopherols (β+δ and γ- isomers), probably found as by-products of phytosterols preparations. These naturally occurring compounds may act as antioxidants in the body by scavenging harmful free radicals, and for that reason it is desirable to conserve these properties in the processed foods.
Fatty acids profile and stability of phytosterols
Oxidative stability depends mainly on fat components in the food, probably coming from maize, milk and phytosterols. In fact, fatty acids used to esterify phytosterols rendered different profiles in the P and C samples, which affected the stability of the product. As shown in Table 2, the C samples had higher content of saturated fatty acids C12, C14, C16 and C18 (p < 0.0001).
Table 2.
Fatty acids profile in fermented maize yogurt-like product with (P) or without (C) added esterified phytosterols, in fresh (day 0) or 12-days stored product (day12) at 4 °C
| Variable | Serie C day 0 | Serie P day 0 | Serie C day 12 | Serie P day 12 | Treatmenta | Day | Day * treatment |
|---|---|---|---|---|---|---|---|
| Fatty acids b (%) | |||||||
| C12:0 | 1.273 ± 0.068a | 0.573 ± 0.021b | 1.653 ± 0.001a | 0.603 ± 0.006b | < 0.0001 | 0.004 | < 0.0001 |
| C14:0 | 6.573 ± 0.592a | 1.647 ± 0.02b1 | 6.980 ± 0.036a | 1.610 ± 0.046b | < 0.0001 | 0.111 | 0.301 |
| C15:1 | 23.763 ± 0.635a | 8.100 ± 0.110b | 24.573 ± 0.525a | 8.120 ± 0.115b | < 0.0001 | 0.002 | 0.238 |
| C16:0 | 1.303 ± 0.006a | 0.440 ± 0.010b | 0.943 ± 0.059a | 0.230 ± 0.010b | < 0.0001 | < 0.0001 | 0.008 |
| C18:0 | 8.250 ± 0.096a | 2.707 ± 0.229b | 8.010 ± 0.115a | 2.473 ± 0.136b | < 0.0001 | 0.791 | 0.202 |
| C18:1 n-9 | 22.653 ± 0.095a | 46.463 ± 0.506b | 29.007 ± 0.216a | 44.550 ± 1.465b | < 0.0001 | < 0.0001 | 0.133 |
| C18:2 n-6 | 17.053 ± 0.058b | 16.867 ± 0.127b | 19.467 ± 0.454a | 19.280 ± 0.344a | 0.399 | < 0.0001 | 0.926 |
| C18:3 n-3 | 1.380 ± 0.017b | 4.040 ± 0.062a | 1.357 ± 0.083b | 1.477 ± 0.038c | < 0.0001 | < 0.0001 | < 0.0001 |
| C18:3 n-6 | 0.283 ± 0.006b | 0.273 ± 0.015b | 0.367 ± 0.015a | 0.374 ± 0.172a | 0.899 | < 0.0001 | < 0.0001 |
| C20:5 n-3 | 0.543 ± 0.025a | 0.460 ± 0.026b | nd | nd | 0.004 | – | – |
| n-6/n-3 | 9.014 ± 0.042b | 3.810 ± 0.050c | 14.642 ± 0.580a | 15.595 ± 0.122a | < 0.0001 | < 0.0001 | < 0.0001 |
| Peroxidation index | 35.224 ± 0.353b | 41.894 ± 0.480a | 36.308 ± 0.822b | 45.394 ± 0.423a | < 0.0001 | < 0.0001 | 0.010 |
aPr > |t| is indicated for treatment
bFatty acids are presented in percentage of total fat extracted and expressed as mean ± standard deviation
MUFA C18:1 n-9 and polyunsaturated (PUFA) and C18:2 n-3 were higher in the P samples than in the C samples (p < 0.0001). The presence of MUFA and PUFA, especially oleic and α-linolenic acids, is a desirable characteristic from a nutritional point of view. In addition, higher percentages of α-linolenic acid (C18:3 n-3) may help achieve the ideal dietary n-6/n-3 PUFA ratio (Simopoulos 2002) with a positive consumers’ perception. However, the higher content of PUFA raised the peroxide index (p < 0.0001) in the P samples (Table 2).
After 12 days at 4 °C, C18:3 n-3 descended drastically in P samples and C20:5 n-3 disappeared in both samples. This accounted for the modification of the n-6/n-3 relationship. Whereas at time 0, it was improved in P respect to the C samples, after storage it augmented and both treatments showed almost the same ratio at expenses of the disappearance of n-3 fatty acids. It seems evident that the incorporation of esterified phytosterols changed the matrix composition and made the enriched samples more susceptible to oxidation.
Evolution of lipid oxidation; pH and total antioxidant activity in yogurt-like maize product
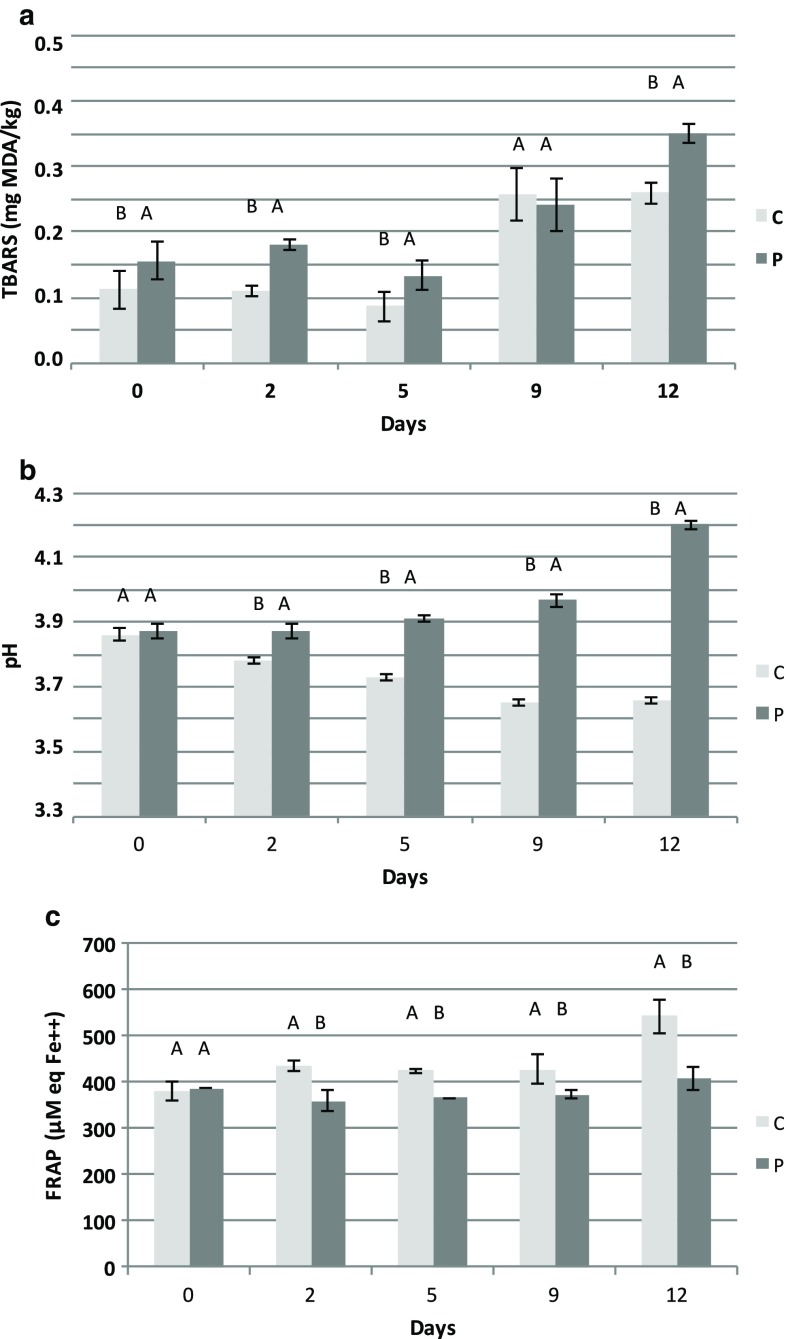
Lipid oxidation was monitored by the TBARS method (Fig. 2a), as well as by volatiles production. The TBARS value at 532 nm was chosen, because, as previously discussed by Semeniuc et al. (2015), many compounds may react with TBA, giving rise to colored compounds at 450 nm, especially aldehydes, lactic acid and other organic acids, which may interfere with the oxidation estimation.
Fig. 2.
Evolution of lipid oxidation (a), pH (b) and total antioxidant activity (c) in control (C) and phytosterol added (P) yogurt-like product during storage at 4 °C. Means and standard deviations are indicated for each point. Different letters indicate significant differences (p < 0.05) between samples using Tukey’s multiple comparisons test of repeated measures analysis
Oxidation remained below 0.12 and 0.2 mg MDA/kg for the C and P samples respectively within the first 5 days. However, TBARS were significantly higher in P (p < 0.05) than in C samples. This result is consistent with the higher susceptibility of the P samples to oxidation (p < 0.0001). At day 9, the onset of oxidation was similar for both treatments, but remained lower in the controls on day 12.
The maize yogurts registered lower TBARS levels (less than 1 mg MDA/kg sample) than flavored yogurts enriched with phytosterols (Semeniuc et al. 2015).
The oxidation of phytosterols is dependent on the matrix. Rudzińska et al. (2014) found that the content of phytosterols and phytostanols in margarines changed from 79 to 63 mg/g in enriched samples stored for 18 weeks at 4 °C. On the other hand, Botelho et al. (2014) showed that phytosterols in chocolate bars were stable during storage, probably due to the presence of high amounts of SFA and polyphenols in cacao. However, this is not the case in the present food because endogenous antioxidants are scarce (see below). In addition, the presence of esterified phytosterols and live microorganisms may contribute to changing its composition.
The initial pH did not differ between treatments (p > 0.05). Values of 3.86 and 3.88 for the C and P series respectively were found in the fresh product, as a result of the lactic fermentation due to production of organic acids. A pH value around 4 is desirable in order to protect food from pathogens (Gullón et al. 2015). However, pH changed significantly during storage (p < 0.0001). It increased in P samples and decreased in the controls (Fig. 2b). This may indicate that different formulations, P series having a higher vegetable fat content than C series, may induce differences within the food environment influencing the metabolism of microorganisms during storage. These microorganisms that were added after cooking (uncooked ogi), may account for the changes.
Results agree with Sacca et al. (2012) showing that Akpan contains live lactic acid bacteria used as starters, as well as other endogenous microorganisms, which transform the food during storage. For that reason, this product can be refrigerated for about 4–7 days, with a maximum storage time of 2 weeks. A modification in the processing of this type of foods may be envisaged in order to improve its stability and nutritional potential.
Total antioxidant activity was measured as reduction power. It was overall higher (p < 0.05) in C than in P samples (Fig. 2c). Surprisingly, this activity increased over time in the C samples, as a possible consequence of microbial metabolism during storage. Hur et al. (2014) postulated that some lactic acid bacteria with β-glucosidase activity could release phenolic antioxidants, and therefore possibly improve the total antioxidant capacity. It has been reported that yellow maize contains native phenolic antioxidant compounds (Oboh and Amusan 2009).Among food phenols, FRAP methodology is especially sensitive to gallic acid, guaiacol, catechins and anthocyanins (Wang et al. 2016). Phenolic compounds may promote antioxidant activity in the water-soluble phase by hydrogen atom transfer interacting with other phytochemicals during storage. This may be the case for guaiacol (C6H4(OH)(OCH3)) which was higher in C than in P samples (see supplementary Fig. 2).
Evolution of antioxidant fat-soluble vitamins
Major carotenoids in the maize yogurt were lutein (Fig. 3a), zeaxanthin (Fig. 3b) (non-provitamin A carotenoids), while smaller quantities were found for β-carotene (Fig. 3c) and β-cryptoxanthin (Fig. 3d) (pro-vitamin A carotenoids). Both the C and P showed similar content.
Fig. 3.
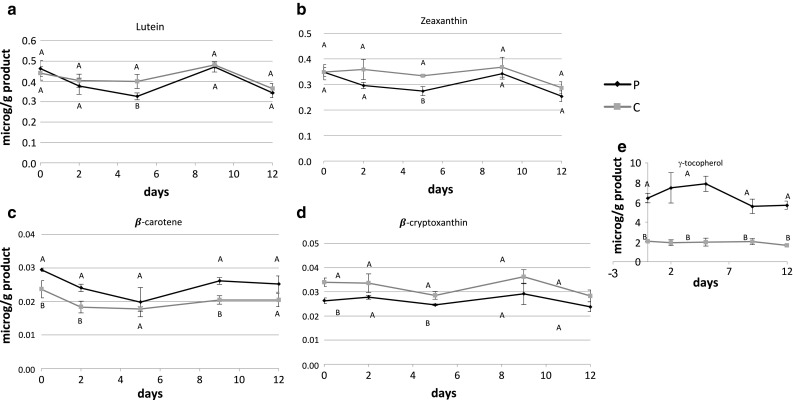
Evolution of fat-soluble vitamins and carotenoids in control (C) and phytosterol added (P) yogurt-like product during storage at 4 °C. Means and standard deviations are indicated for each point. Different letters indicate significant differences (p < 0.05) between samples using Tukey’s multiple comparisons test of repeated measures analysis
Lutein and zeaxanthin were conserved until day 9, and decreased similarly for P and C products on day 12. β-carotene and β-cryptoxanthin showed tenfold lower values than the former carotenoids and also seemed to be conserved during storage.
Despite their stability, provitamin A carotenoids are low in these products (Weber and Grune 2012). The maximum values were around 0.025 µg of β-carotene and 0.035 µg of β-cryptoxanthin per gram of product which correspond (in a portion of 125 g) approximately to 0.2% of the carotene amount consumed by adults daily in developed countries (Bechoff and Dhuique-Mayer 2017). Therefore, a fortification strategy could be contemplated in order to propose this type of food as a vitamin A source.
The tocopherols profile was different between the C and P series (Fig. 3e). P showed a significantly higher (p < 0.0001) content of γ- and β + δ-tocopherols (an average of 6.5 and 2.5 µg/g, respectively). Meanwhile, C samples showed low levels of γ-tocopherol (an average of 2 µg/g). The α-isomer was not detected in any of the products.
The γ- and β + δ-tocopherol isomers are part of the commercial preparation of esterified phytosterols, and may also serve as antioxidants. However, at those concentrations they were not sufficient to counteract the oxidation induced in the P samples, probably due to high PUFA composition.
Kmiecik et al. (2015) reported that synthetic tocopherols were effective to inhibit phytosterols oxidation in oil mixtures submitted to heating using concentrations of α-, β-, δ- and γ-tocopherol were 24, 0.1, 45 and 1.5 mg/100 g of oil, respectively. These values are tenfold higher than the ones reported in the present work, indicating that even P product is a poor source of vitamin E in terms of food functionality.
This work shows for the first time the levels of vitamin A and vitamin E in a yogurt-like maize-derived product. This is important because many authors cite yellow maize as a source of these vitamins. However, processing and storage will strongly affect their content in food, and this should be taken into account in order to produce foods with functional claims.
Similarly to phytosterols, antioxidant vitamins seemed to be stable during refrigeration and therefore antioxidant strategies should be applied in order to conserve the bioactive compounds within the matrix.
Formation of volatile compounds during storage
Some volatile compounds are formed during oxidation. Therefore, the relationship between volatiles production, the presence of esterified phytosterols and their evolution during storage was described by a clustering analysis in the supplementary Fig. 2. The heat map showed that C and P samples were clustered regarding their similarity of the released volatiles. In the heat map the samples (C or P) are displayed vertically and the volatiles are displayed horizontally (columns). Inside each cluster, fresh (C0 and P0) or stored samples (C9 and P9) were correctly classified.
Similarly expressed features are characterized by horizontal rectangles of homogeneous color along the map. The color corresponds to the level of significance with high (> 1 relative units in green color) and low values (< − 1 relative units in red). This map illustrates the general picture indicating the volatiles that are typical in each formulation.
Acetic acid was the main volatile with a significant increase over time (p < 0.001), followed by acetoin, but without changes during storage (p > 0.05). It has been reported that Lactobacillus casei, can produce lactic acid; diacetyl; acetoin and polysaccharides (Branen and Keenan 1971). Also this compound was found by Semeniuc et al. (2015) in milk yogurts with phytosterols and is related, as other organic acids, to the fermentation process.
Other organic acids, significantly contributed to clustering of control samples. For example, 3-methyl butanoic, butanoic, 2-methyl propanoic, octanoic and nonanoic acids and lactic acid methyl-ester were higher in the C samples than in the P samples (p < 0.001), and their relative quantities did not change over time. These acids may result from microbial metabolism rather than from lipid oxidation. Hexanoic and pentanoic acid, as well as their derivative alcohols, formed a cluster and also contributed to C classification. It has been previously reported that hexanoic acid and hexanol are related to lipid oxidation and deterioration of cereals (Lampi et al. 2015). They may also account for the differences in pH between the P and C groups.
Alcohols, 1-butanol, iso-pentanol, 1-heptanol were higher in the C samples than in the P samples, and may result from oxidation, as they (with the exception of 1-butanol) increased during storage.
Aldehydes were found mainly in P samples, indicating lipid oxidation. Particularly, (E)-2-hexenal and (E, E)-2, 4-heptadienal were found exclusively in the P samples, and may rise from degradation of n-3 fatty acids.
2-pentylfuran was found almost exclusively in the P samples, and may by caused by a combination of lipid oxidation and aldehyde degradation.
Other compounds such as 2, 3-heptanedione and 3-methyl butanol were exclusively from C samples whereas phenolic compounds; p-xylene and p-cymene were present exclusively in the P yogurts at late stages of storage. Interestingly, guaiacol was higher in the C samples than in the P samples (p < 0.001), and it may account for the antioxidant differences found between treatments.
In conclusion, volatile compounds profile was a useful tool for differentiating the P and C samples by means of phytosterols addition, fermentation, and lipid oxidation during storage. Indeed, volatile profile is a complementary tool to explain the oxidative process in this type of products where TBARS may have interferences. Volatiles may also be linked to sensory attributes. A sensory profile of the maize product Akpan, showed that its acceptance correlated with fermented odor and milky taste attributes, and that there were differences between European and African consumers for these attributes (Akissoé et al. 2014). As a functional food, it should also be considered to replace sugar. Normally, sugar and milk are both most commonly added to prepare maize fermented drinks, whereas the plain form (with no ingredient added) is the least common way of consuming them. Therefore, in a next step it is envisaged a consumer’s test evaluating the functional product.
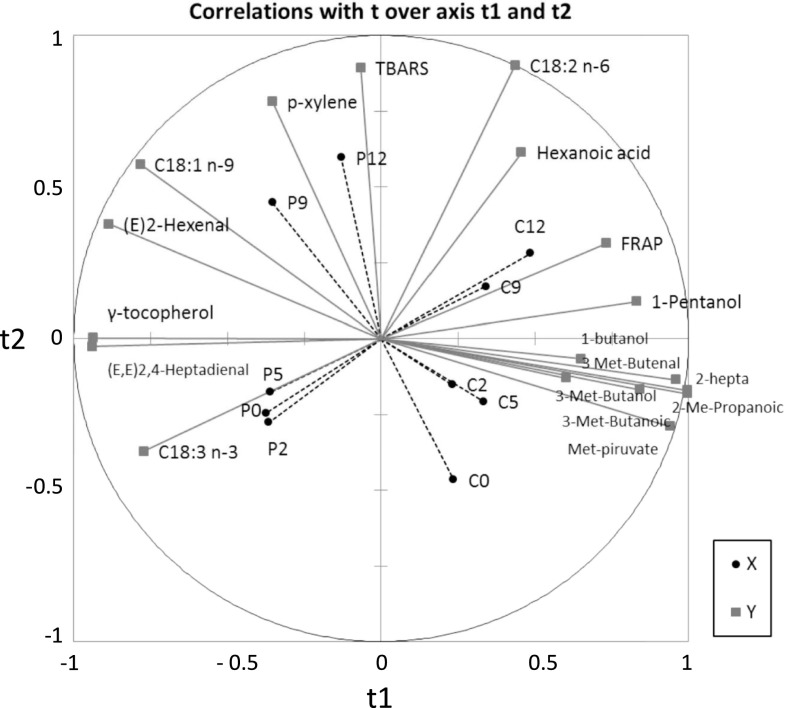
Partial least square (PLS) regression
PLS obtains a simultaneous map of the descriptors and the products over time. It also showed factors which are related to P or C treatments (X variables)
Each set of dependent Y variables (except for phytosterols content) was submitted first to a discriminant analysis with the stepwise descendent selection method in order to select the discriminant variables for treatments (P or C) and storage times (2, 5, 9 and 12 days).
The biochemical parameters were: FRAP and TBARS; fatty acids: C12, C14, C18:1 n-9, C18:2 n-6; C18:3 n-3 and antioxidants: γ-tocopherol and lutein. The volatiles were: p-xylene; 1-butanol; 2-heptanone; 3-methyl-2-butenal; 3-methyl-1-butanol; 3-methyl pyruvate; 1-pentanol; (E)-2-hexenal; 2-methyl-propanoic acid; 3-methyl-butanoic acid; hexanoic acid; (E,E)-2,4-heptadienal.
As shown in Fig. 4, the first component clearly separated the P and C groups along the t1 axis. The second component t2 separated the storage times. The treatments were correctly separated by their chemical characteristics, as they are not concentrated in one part of the circle, but well distributed all around it. The C group recorded positive scores in t1 and was associated with the presence of organic acids and alcohols. Meanwhile, the P samples had negative scores in t1 and were associated with γ-tocopherol, PUFAs and aldehydes, as indicators of oxidation.
Fig. 4.
PLS regression of discriminant variables applied to control (C) and phytosterols added (P) yogurt-like product during storage at 4 °C. Y quantitative (biochemical) and X qualitative (treatments) variables are plotted in the two first components that account for 79.58% of the variability
For the second component, they were separated by the initial and final stages of refrigerated storage. In this case, fresh samples (day 0) of both products exhibited negative scores. There was no clear separation over days 0, 2 and 5 between each group, P or C. This is an expected result, as the evolution of the food was more pronounced at days 9 and 12. In this case, days 9 and 12 were associated with higher TBARS, hexanoic acid, C18:2 n-6 and p-xylene levels.
Therefore, multivariate analyses made it possible to describe associations between quantitative and qualitative data, in order to illustrate the evolution of the product formulated with or without phytosterols. Combinations of variables explained the differences attributable to changes either in composition or in oxidative stability and may be used to predict its behavior during storage.
Conclusion
This work proved the possibility of obtaining a potentially functional food, with an average concentration of 2.7 g of free phytosterols/125 g of final ready-to-eat product obtained by modification of a traditional process. In addition, the maize yogurt has a low content of milk, which may be an alternative for intolerance cases. Similarly, it may be suitable for the celiac community, and may also be a model for the use of other cereals, adapted to nutritional and territorial needs.
Addition of antioxidants and vitamin sources should be considered in order to augment its bioactive potential, as well as protect the product from oxidative damage. Other aspects may be also considered, such as monitoring microbial evolution during fermentation, processing and storage, to ensure that the product will be acceptable after longer storage times. Furthermore, bioaccessibility of functional molecules and consumers acceptance of the functional product are to be considered.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1: Phytosterols content in fermented maize yogurt-like product (P samples) during refrigerated storage. Different letters indicate significant differences (p < 0.05) between samples using Tukey’s multiple comparisons test of one-way ANOVA. (PDF 15 kb)
Supplementary Fig. 2: Heat map of volatile compounds determined in control (C) and phytosterol added (P) yogurt-like product during storage at 4 °C. Green color indicates high volatile values (> 1 relative units) and red color indicates low volatile values (< -1 relative units). (PDF 27 kb)
Acknowledgements
We wish to particularly thank Dr. Debora Primrose, a native speaker, for her revision of the English language and Safisis/Lesaffre for providing the Lactobacillus casei (CNCM I-4592).
Footnotes
Electronic supplementary material
The online version of this article (10.1007/s13197-018-3102-5) contains supplementary material, which is available to authorized users.
References
- Akissoé NH, Sacca C, Declemy AL, Bechoff A, Anihouvi VB, Dalodé G, Pallet D, Fliedel G, Mestres C, Hounhouigan JD, Tomlins KI. Cross-cultural acceptance of a traditional yoghurt-like product made from fermented cereal. J Sci Food Agric. 2014;95:1876–1884. doi: 10.1002/jsfa.6892. [DOI] [PubMed] [Google Scholar]
- Amiot MJ, Knol D, Cardinault N, Nowicki M, Bott R, Antona C, Borel P, Bernard JP, Duchateau G, Lairon D. Phytosterol ester processing in the small intestine: impact on cholesterol availability for absorption and chylomicron cholesterol incorporation in healthy humans. J Lipid Res. 2011;52:1256–1264. doi: 10.1194/jlr.M013730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- A.O.A.C. (1980) Official methods of analysis, 15th edn. Association of Official Analitycal Chemists. Kenneth Helrich, Ed., Washington DC
- Bechoff A, Dhuique-Mayer C. Factors influencing micronutrient bioavailability in biofortified crops. Ann NY Acad Sci. 2017;1390:74–87. doi: 10.1111/nyas.13301. [DOI] [PubMed] [Google Scholar]
- Blumenfeld Olivares JA, Martin ISM, Calle ME, Valdes CB, Arruche EP, Delgado EA, Ciudad MJ, Cabria MH, Yurita LC. Low-fat, fermented milk enriched with plant sterols, a strategy to reduce hypertriglyceridema in children, a double-blind, randomized placebo-cotrolled trial. Nutr Hosp. 2015;32:1056–1060. doi: 10.3305/nh.2015.32.3.9319. [DOI] [PubMed] [Google Scholar]
- Botelho PB, Galasso M, Dias V, Mandrioli M, Lobato L, Rodriguez-Estrada M, Castro I. Oxidative stability of functional phytosterol-enriched dark chocolate. LWT Food Sci Technol. 2014;55:444–451. doi: 10.1016/j.lwt.2013.09.002. [DOI] [Google Scholar]
- Branen AL, Keenan TW. Diacetyl and acetoin production by Lactobacillus casei. Appl Microbiol. 1971;22:517–521. doi: 10.1128/am.22.4.517-521.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Descalzo AM, Insani EM, Biolatto A, Sancho AM, García PT, Pensel NA, Josifovich JA. Influence of pasture or grain-based diets supplemented with vitamin E on antioxidant/oxidative balance of Argentine beef. Meat Sci. 2005;70:35–44. doi: 10.1016/j.meatsci.2004.11.018. [DOI] [PubMed] [Google Scholar]
- Dhuique-Mayer C, Servent A, Descalzo A, Mouquet-Rivier C, Amiot M-J, Achir N. Culinary practices mimicking a polysaccharide-rich recipe enhance the bioaccessibility of fat-soluble micronutrients. Food Chem. 2016;210:182–188. doi: 10.1016/j.foodchem.2016.04.037. [DOI] [PubMed] [Google Scholar]
- Gorton D, Bullen CR, Mhurchu CN. Environmental influences on food security in high income countries. Nutr Rev. 2010;68:1–29. doi: 10.1111/j.1753-4887.2009.00258.x. [DOI] [PubMed] [Google Scholar]
- Gullón B, Pereira M, Mestres C, Hounhouigan J, Pallet D, Alonso JL, Pintado M. Assessment of prebiotic potential of Akpan-yoghurt-like product and effects on the human intestinal microbiota. J Funct Food. 2015;19:545–553. doi: 10.1016/j.jff.2015.09.026. [DOI] [Google Scholar]
- Gylling H, Plat J, Turley S, Ginsberg HN, Ellegård L, Jessup W, Jones PJ, Lütjohann D, Maerz W, Masana L, Silbernagel G, Staels B, Borén J, Catapano AL, De Backer G, Deanfield J, Descamps OS, Kovanen PT, Riccardi G, Tokgözoglu L, Chapman MJ. Plant sterols and plant stanols in the management of dyslipidaemia and prevention of cardiovascular disease. Atherosclerosis. 2014;232:346–360. doi: 10.1016/j.atherosclerosis.2013.11.043. [DOI] [PubMed] [Google Scholar]
- Hallikainen MA, Uusitupa MI. Effects of 2 low-fat stanol ester-containing margarines on serum cholesterol concentrations as part of a low-fat diet in hypercholesterolemic subjects. Am J Clin Nutr. 1999;69:403–410. doi: 10.1093/ajcn/69.3.403. [DOI] [PubMed] [Google Scholar]
- Hur SJ, Lee SY, Kim YC, Choi I, Kim GB. Effect of fermentation on the antioxidant activity in plant-based foods. Food Chem. 2014;160:346–356. doi: 10.1016/j.foodchem.2014.03.112. [DOI] [PubMed] [Google Scholar]
- Kearney J. Food consumption trends and drivers. Philos Trans R Soc Lond B Biol Sci. 2010;365:2793–2807. doi: 10.1098/rstb.2010.0149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kmiecik D, Korczak J, Rudzińska M, Gramza-Michałowska A, Hęś M, Kobus-Cisowska J. Stabilisation of phytosterols by natural and synthetic antioxidants in high temperature conditions. Food Chem. 2015;173:966–971. doi: 10.1016/j.foodchem.2014.10.074. [DOI] [PubMed] [Google Scholar]
- Kohajdova Z. Fermented cereal products. In: Ray BC, Montet D, editors. Microorganisms and fermentation of traditional foods. Boca Raton: CRC Press; 2015. pp. 78–107. [Google Scholar]
- Lampi AM, Damerau A, Li J, Moisio T, Partanen R, Forssell P, Piironen V. Changes in lipids and volatile compounds of oat flours and extrudates during processing and storage. J Cereal Sci. 2015;62:102–109. doi: 10.1016/j.jcs.2014.12.011. [DOI] [Google Scholar]
- Lee MJ, Feng W, Yang L, Chen Y-K, Chi E, Liu A, Yang CS. Methods for efficient analysis of tocopherols, tocotrienols and their metabolites in animal samples with HPLC-EC. J Food Drug Anal. 2017 doi: 10.1016/j.jfda.2017.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ling WH, Jones PJH. Dietary phytosterols: a review of metabolism, benefits and side effects. Life Sci. 1995;57:195–206. doi: 10.1016/0024-3205(95)00263-6. [DOI] [PubMed] [Google Scholar]
- NMX-F-317-S-1978 (1978) Determination of pH in foods. Normas mexicanas. Dirección General de Normas. http://www.colpos.mx/bancodenormas/nmexicanas/NMX-F-317-S-1978.PDF. Accessed 24 April 2016
- Oboh G, Amusan TV. Nutritive value and antioxidant properties of cereal gruels produced from fermented maize and sorghum. Food Biotechnol. 2009;23:17–31. doi: 10.1080/08905430802671899. [DOI] [Google Scholar]
- Rondanelli M, Monteferrario F, Faliva MA, Perna S, Antoniello N. Key points for maximum effectiveness and safety for cholesterol-lowering properties of plant sterols and use in the treatment of metabolic syndrome. J Sci Food Agric. 2013;93:2605–2610. doi: 10.1002/jsfa.6174. [DOI] [PubMed] [Google Scholar]
- Rossetti L, Langman L, Grigioni GM, Biolatto A, Sancho AM, Comerón E, Descalzo AM. Antioxidant status and odor profile in milk from silage or alfalfa-fed cows. Aus J Dairy Technol. 2010;65:3–9. [Google Scholar]
- Rudzińska M, Przybylski R, Wąsowicz E. Degradation of phytosterols during storage of enriched margarines. Food Chem. 2014;142:294–298. doi: 10.1016/j.foodchem.2013.07.041. [DOI] [PubMed] [Google Scholar]
- Sacca C, Adinsi L, Anihouvi V, Akissoé N, Dalode G, Mestres C, Jacobs A, Dlamini N, Pallet D, Hounhouigan DJ. Production, consumption, and quality attributes of Akpan—a yoghurt-like cereal product from West Africa. Food Chain. 2012;2:207–220. doi: 10.3362/2046-1887.2012.018. [DOI] [Google Scholar]
- Saraiva D, Castilho MC, Martins MR, Noronha da Silveira MI, Ramos F. Evaluation of phytosterols in milk and yogurts used as functional foods in Portugal. Food Anal Method. 2011;4:28–34. doi: 10.1007/s12161-010-9131-y. [DOI] [Google Scholar]
- Semeniuc CA, Vladimiro C, Mandrioli M, Muste S, Borsari A, Rodriguez-Estrada MT. Stability of flavoured phytosterol-enriched drinking yogurts during storage as affected by different packaging materials. Eur Food Res Technol. 2015;242:431–439. doi: 10.1007/s00217-015-2554-3. [DOI] [PubMed] [Google Scholar]
- Simopoulos AP. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed Pharmacother. 2002;56:365–379. doi: 10.1016/S0753-3322(02)00253-6. [DOI] [PubMed] [Google Scholar]
- Slavin M, Yu LL. A single extraction and HPLC procedure for simultaneous analysis of phytosterols, tocopherols and lutein in soybeans. Food Chem. 2012;135:2789–2795. doi: 10.1016/j.foodchem.2012.06.043. [DOI] [PubMed] [Google Scholar]
- Wang B, Huang Q, Venkitasamy C, Chai H, Gao H, Cheng N, Cao W, Lv X, Pan Z. Changes in phenolic compounds and their antioxidant capacities in jujube (Ziziphusjujuba Miller) during three edible maturity stages. LWT Food Sci Technol. 2016;66:56–62. doi: 10.1016/j.lwt.2015.10.005. [DOI] [Google Scholar]
- Weber D, Grune T. The contribution of beta-carotene to vitamin A supply of humans. Mol Nutr Food Res. 2012;56:251–258. doi: 10.1002/mnfr.201100230. [DOI] [PubMed] [Google Scholar]
- Witting LA, Horwitt MK. Effect of degree of fatty acid unsaturation in tocopherol deficiency-induced creatinuria. J Nutr. 1964;82:19–23. doi: 10.1093/jn/82.1.19. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Fig. 1: Phytosterols content in fermented maize yogurt-like product (P samples) during refrigerated storage. Different letters indicate significant differences (p < 0.05) between samples using Tukey’s multiple comparisons test of one-way ANOVA. (PDF 15 kb)
Supplementary Fig. 2: Heat map of volatile compounds determined in control (C) and phytosterol added (P) yogurt-like product during storage at 4 °C. Green color indicates high volatile values (> 1 relative units) and red color indicates low volatile values (< -1 relative units). (PDF 27 kb)