Abstract
βΒ2-crystallin (CRYBB2) is expressed at an increased level in the postnatal lens cortex and is associated with cataracts. Improved understanding of the underlying biology of cataracts is likely to be critical for the development of early detection strategies and new therapeutics. The present study aimed to identify long non-coding RNAs (lncRNAs) and mRNAs associated with CRYBB2 knockdown (KO)-induced cataracts. RNAs from 3 non-treated mice and 3 CRYBB2 KO mice were analyzed using the Affymetrix GeneChip Mouse Gene 2.0 ST array. A total of 149 lncRNAs and 803 mRNAs were identified to have upregulated expression, including Snora73b, Klk1b22 and Rnu3a, while the expression levels of 180 lncRNAs and 732 mRNAs were downregulated in CRYBB2 KO mice, including Snord82, Snhg9 and Foxn3. This lncRNA and mRNA expression profile of mice with CRYBB2 KO provides a basis for studying the genetic mechanisms of cataract progression.
Keywords: cataract, βB2-crystallin, long non-coding RNA, mRNA
Introduction
Congenital cataracts area common cause of blindness, with the incidence estimated to be 1–6/10,000 infants in most populations (1,2). There are 13,000–200,000 patients with bilateral congenital cataract who go blind each year worldwide, with an increase of 2,000–40,000 per year (3). The primary clinical manifestation of the disease is the occurrence of lens opacity in the first year (4). Although surgical techniques and visual prognosis have improved, congenital cataracts remain the leading cause of visual disability in children worldwide (5). Previous studies have revealed that almost one-third of congenital cataracts are caused by genetic mutations (6), and 13 genes have previously been confirmed to be associated with congenital cataracts (7). These include crystallin genes [αA-crystallin (CRYAA), αB-crystallin, βΑ1-crystallin (CRYBA1), βB1-crystallin, βB2-crystallin (CRYBB2), γC-crystallin and γD-crystallin (CRYGD)], membrane transport protein genes [major intrinsic protein (MIP), gap junction protein (GJ)A3 and GJA8], a cytoskeletal protein gene [beaded filament structural protein 2 (BFSP2)], and transcription factor genes (paired-like homeodomain 3 and heat shock transcription factor 4).
Evidence indicates that gene expression in the lens epithelium is significantly altered during cataract formation. Sheets et al (8) reported the downregulation of CRYAA and CRYBA1/CRYBA3 and the upregulation of the receptor tyrosine kinase adhesion-related kinase (ARK) in the Emory mouse, a well-characterized model of age-dependent cataracts. Furthermore, metallothionein-IIA, osteonectin and ARK are upregulated in cataractous lenses relative to transparent lenses (9–11). Ruotolo et al (12) identified extensive downregulation of genes, including GCS1, GRB7, FST and POLR2E, in the lens associated with the development of age-related cataracts in humans. Although these changes in gene expression are informative, further gene identifications are required to elucidate the molecular mechanism of cataract formation. Crystallins, CRYBB2 in particular, are considered to act primarily as structural proteins of the lens (13). Previously, it was demonstrated that the relative amounts of CRYBB2 protein expression in the lens change markedly, increasing from 12 to 24% (14), suggesting that CRYBB2 serves a contributive function in lens development. Moreover, targeted knockout (KO) of CRYBB2 in mice has been demonstrated to induce age-related (15) and congenital cataracts (16); however, its functional significance is not yet known.
Long non-coding RNAs (lncRNAs) are defined as non-coding RNA molecules >200 nucleotides in length with limited protein coding potential (17,18). Previous studies have indicated that lncRNAs are deregulated in numerous diseases and associated with a wide range of biological processes, such as proliferation, apoptosis and cell migration (19,20). Recently, some lncRNAs have been identified to serve critical functions in eye development and diseases. Shen et al (21) reported that 38 lncRNAs were differentially expressed between transparent and cataractous lenses, among which one of the most abundant lncRNAs, myocardial infarction associated transcript, was specifically upregulated in the plasma fraction of whole blood and the aqueous humor of cataract patients. However, the function of lncRNAs in human lenses remains unknown.
In the present study, differences in lncRNA and mRNA expression between the lenses of untreated mice and CRYBB2 KO-induced cataract mice models were evaluated. A total of149 lncRNAs and 803 mRNAs were identified whose expression was upregulated, while the expression levels of a further 180 lncRNAs and 732 mRNAs were downregulated in CRYBB2 KO mice lenses. These findings suggest a potential function for these lncRNAs and mRNAs in cataract formation.
Materials and methods
Animals
A total of 3 male wild type (WT) and 3 male CRYBB2 KO BALB/c mice (age, 12 weeks old; weight, 25 g) were provided by in Genious Targeting Laboratory, Inc. (Ronkonkoma, NY, USA) (22). Mice with targeted disruption of the CRYBB2 gene were generated at the company by inserting a neo expression cassette to replace the first and second exons, preventing the production of a functional transcript from this locus. Mice were maintained in an animal facility at 25°C, with a relative humidity of 60–70%, under a 12-h light/dark cycle with free access to food and water at the Laboratory Animal Center of the Changhai Hospital, Second Military Medical University (Shanghai, China). All procedures were carried out in accordance with the Chinese legislation on the Use and Care of Laboratory Animals and the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research (23) and were approved by the Institutional Animal Care and Use Committee of Changhai Hospital, Second Military Medical University (Shanghai, China).
RNA extraction
Following the sacrifice of the mice, the lenses were collected and RNA was isolated from the lenses of mice using the Chomczynski method (24) and was further purified using an RNeasy MinElute Clean-up kit (Qiagen GmbH, Hilden, Germany). The RNA concentration was measured with a Nanodrop spectrophotometer (Thermo Fisher Scientific, Inc., Waltham, MA, USA). The A260/A280 ratio was 1.8–2.0 and the quality of the RNA was verified by agarose gel electrophoresis.
Microarray processing
LncRNA and mRNA expression profiling was performed using the Affymetrix GeneChip Mouse Gene 2.0 ST array (Thermo Fisher Scientific, Inc.), according to the manufacturer's protocols. Intensities of target hybridization to respective probe features were detected by laser scanning of the array. First, quantile normalization of the microarray data of the 3 untreated and 3 CRYBB2 KO mice was performed. The data was then log2-scale transformed. Hierarchical clustering of the lncRNA and mRNA profiles was performed using Cluster 3.0 software (http://bonsai.hgc.jp/~mdehoon/software/cluster/software.htm) (25). The normalized expression values of the lncRNAs and mRNAs were centered on the median before unsupervised hierarchical clustering was performed. Clustering was performed with complete linkage and centered Pearson correlation. To estimate the accuracy of the measurements, the coefficient of variance for each measured parameter was determined.
Statistical analysis
Statistical analyses were performed with the use of GraphPad Prism 5 software (GraphPad Software, Inc., La Jolla, CA, USA). The Significance Analysis of Microarray method was used to identify significant gene expression changes between CRYBB2 KO mice and controls (26). P<0.05 was considered to indicate a statistically significant difference.
Results
Analysis of lncRNA expression patterns in CRYBB2 KO mice
The lncRNA expression profiles of lens tissues were compared using unsupervised hierarchical clustering in 3 untreated and 3CRYBB2 KO mice. As demonstrated in Fig. 1, in total, 329 lncRNAs with a coefficient of variance >0.10 were selected for clustering analysis. Hierarchical clustering of these 329 lncRNAs based on centered Pearson correlation indicated notable differential lncRNA expression in CRYBB2 KO and untreated mice (Fig. 1). Among these lncRNAs, 17 exhibited at least a two-fold change in the CRYBB2 KO mice compared with the untreated mice (all upregulated in CRYBB2 KO mice). A total of 149 out of 329 lncRNAs were upregulated in CRYBB2 KO mice compared with the untreated mice (Table I presents the top 20 most upregulated lncRNAs), whereas 180 out of 329 lncRNAs were downregulated in CRYBB2 KO mice compared with the untreated mice (Table II presents the top 20 most downregulated lncRNAs).
Figure 1.
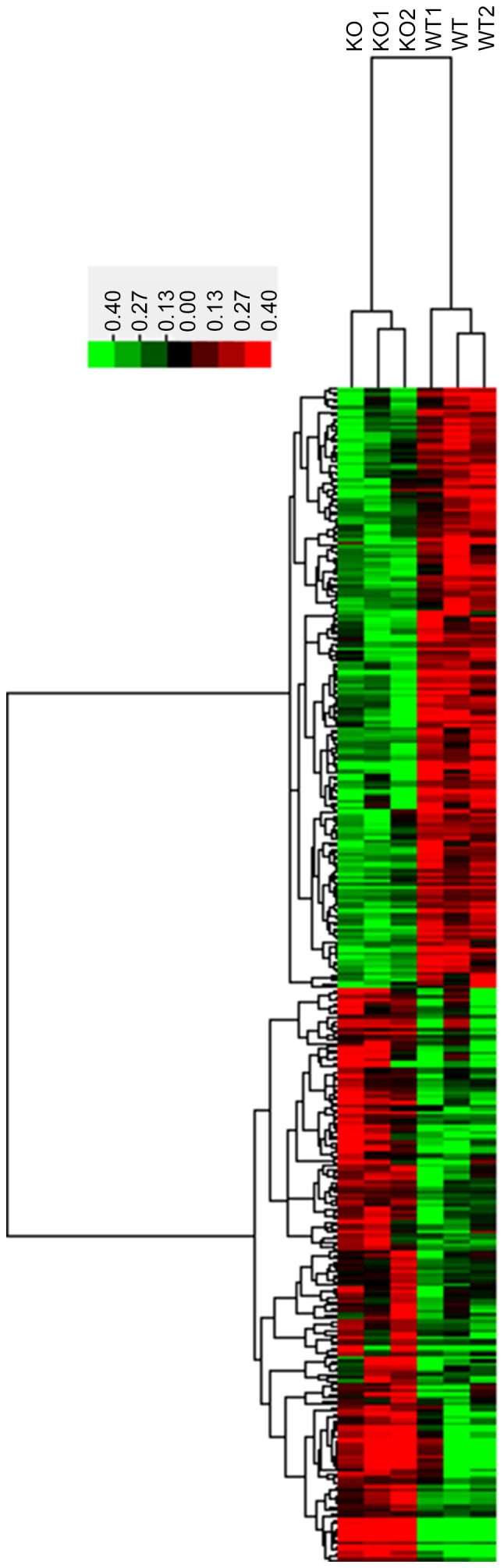
Differential expression of lncRNAs in WT and βB2-crystallin KO cataractous lens samples. Heatmaps were generated from hierarchical cluster analysis to indicate differential expression. The color scale illustrates the relative expression level of lncRNAs across different samples. Red denotes upregulation; green denotes downregulation. KO, knockout; WT, wild type; lncRNA, long non-coding RNA.
Table I.
Top 20 upregulated long non-coding RNAs in βB2-crystallin KO mice.
| Probe set ID | P-value | Fold-change (KO/WT) | MGI gene symbol | Gene description | GenBank accession no. |
|---|---|---|---|---|---|
| 17296979 | 2.00×10−7 | 5.84 | Gm10409 | Predicted gene 10409 | NR_033121 |
| 17303368 | 1.00×10−6 | 5.21 | Gm3002 | α-takusan pseudogene | NR_033388 |
| 17296602 | 2.60×10−6 | 3.75 | Gm3020 | Predicted gene 3020 | NR_033117 |
| 17430831 | 3.57×10−5 | 3.55 | Snora73b | Small nucleolar RNA, H/ACA box 73b | NR_028513 |
| 17303147 | 1.70×10−6 | 3.52 | Gm3591 | Predicted gene 3591 | XR_141206 |
| 17434158 | 6.77×10−3 | 3.51 | – | – | ENSMUST00000169242 |
| 17337152 | 4.01×10−3 | 3.38 | – | – | ENSMUST00000174425 |
| 17424407 | 1.21×10−2 | 2.98 | 4933409K07Rik | RIKEN cDNA 4933409K07 gene | NR_033123 |
| 17413061 | 3.08×10−3 | 2.87 | – | – | ENSMUST00000169242 |
| 17480922 | 3.86×10−4 | 2.68 | Mir139 | MicroRNA139 | NR_029791 |
| 17302054 | 3.04×10−2 | 2.49 | Snora31 | Small nucleolar RNA, H/ACA box 31 | NR_028481 |
| 17232731 | 3.02×10−2 | 2.4 | Rnu3a | U3A small nuclear RNA | NR_002842 |
| 17412952 | 9.63×10−3 | 2.32 | Gm3893 | Predicted gene 3893 | NR_033506 |
| 17342996 | 2.00×10−3 | 2.21 | Gm16197 | Predicted gene 16197 | NR_036469 |
| 17421488 | 1.19×10−2 | 2.06 | – | – | ENSMUST00000172415 |
| 17348121 | 3.41×10−2 | 2.04 | 4833419F23Rik | RIKEN cDNA4833419F23 gene | NR_040328 |
| 17347279 | 3.47×10−2 | 2.01 | – | – | ENSMUST00000157334 |
| 17430833 | 1.42×10−3 | 1.98 | Snora73a | Small nucleolar RNA, H/ACA box 73a | NR_028512 |
| 17221923 | 2.92×10−2 | 1.91 | – | – | ENSMUST00000083191 |
| 17523680 | 4.81×10−4 | 1.89 | Mir101c | MicroRNA101c | NR_039546 |
KO, knockout; WT, wild type; MGI, mouse genome informatics database.
Table II.
Top 20 downregulated long non-coding RNAs in βB2-crystallin KO mice.
| Probe set ID | P-value | Fold-change (KO/WT) | MGI gene symbol | Gene description | GenBank accession no. |
|---|---|---|---|---|---|
| 17251898 | 4.62×10−2 | 0.86 | Mir324 | MicroRNA 324 | NR_029758 |
| 17547715 | 4.84×10−2 | 0.86 | – | – | ENSMUST00000117972 |
| 17468138 | 4.35×10−2 | 0.85 | – | – | ENSMUST00000145420 |
| 17329209 | 4.58×10−2 | 0.85 | A830060N17 | Uncharacterized LOC328646 | NR_046162 |
| 17365718 | 3.37×10−2 | 0.84 | – | – | ENSMUST00000162724 |
| 17403967 | 3.43×10−2 | 0.84 | – | – | ENSMUST00000158662 |
| 17362668 | 3.62×10−2 | 0.84 | – | – | ENSMUST00000169060 |
| 17315735 | 3.71×10−2 | 0.84 | – | – | ENSMUST00000160698 |
| 17527984 | 4.10×10−2 | 0.84 | A730043L09 | Uncharacterized protein A730043L09 | NR_040769 |
| 17278612 | 4.40×10−2 | 0.84 | Mir342 | MicroRNA342 | NR_029771 |
| 17448958 | 2.17×10−2 | 0.83 | – | – | ENSMUST00000158856 |
| 17269866 | 3.17×10−2 | 0.83 | – | – | ENSMUST00000102272 |
| 17225173 | 3.59×10−2 | 0.83 | Snord82 | Small nucleolar RNA, C/D box 82 | NR_002851 |
| 17230480 | 3.98×10−2 | 0.83 | – | – | ENSMUST00000128545 |
| 17532275 | 4.53×10−2 | 0.83 | – | – | ENSMUST00000082463 |
| 17345664 | 4.84×10−2 | 0.83 | – | – | ENSMUST00000122623 |
| 17280661 | 2.01×10−2 | 0.82 | F730043M19Rik | RIKEN cDNA F730043M19 gene | NR_015602 |
| 17395003 | 2.17×10−2 | 0.82 | – | – | ENSMUST00000133525 |
| 17342024 | 2.90×10−2 | 0.82 | Snhg9 | Small nucleolar RNA host gene (non-protein coding) 9 | NR_027900 |
| 17232800 | 3.09×10−2 | 0.82 | – | – | ENSMUST00000104610 |
KO, knockout; WT, wild type; MGI, mouse genome informatics database.
Analysis of mRNA expression patterns in CRYBB2 KO mice
The mRNA expression profiles of lens tissues were compared using unsupervised hierarchical clustering in the 3 untreated and 3 CRYBB2 KO mice. In total, 1,535 mRNAs with a coefficient of variance >0.10 were selected for clustering analysis. Hierarchical clustering of these 1,535 mRNAs based on centered Pearson correlation indicated notable differential mRNA expression between CRYBB2 KO and untreated mice (Fig. 2). Among these mRNAs, 52 exhibited at least a two-fold change in the CRYBB2 KO mice compared with the untreated mice (all upregulated in CRYBB2 KO mice). A total of 803 out of 1,535 mRNAs were upregulated in CRYBB2 KO mice compared with the untreated mice (Table III presents the top 20 most upregulated mRNAs), whereas 732 out of 1,535 mRNAs were downregulated (Table IV presents the top 20 most downregulated mRNAs).
Figure 2.
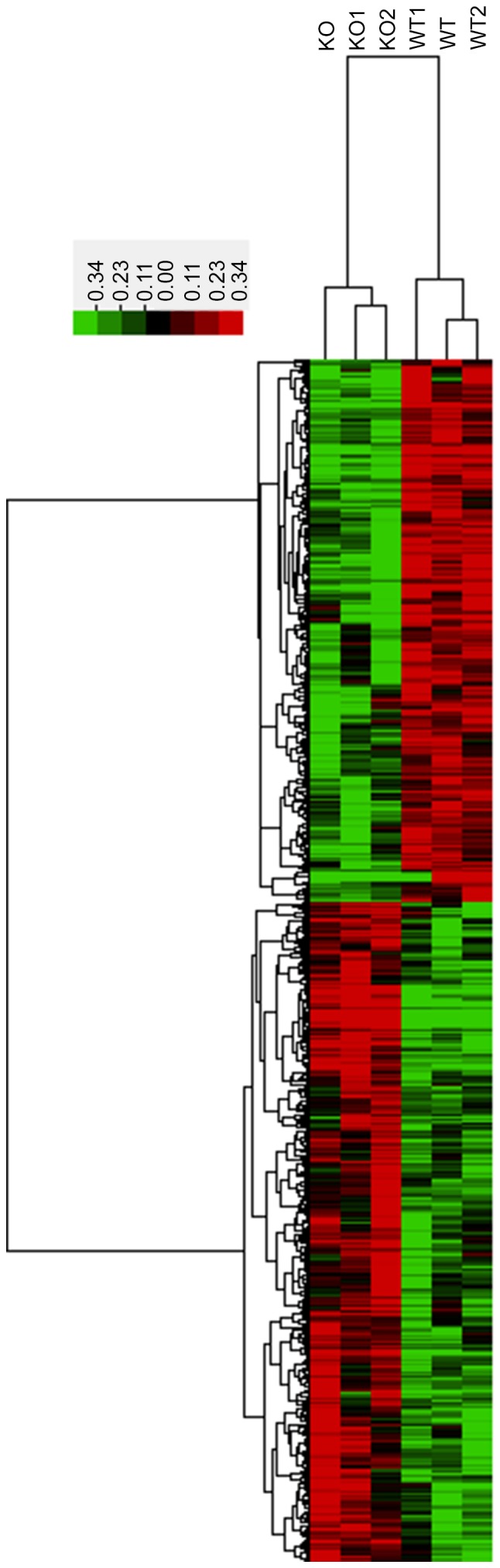
Differential expression of mRNAs in WT and βB2-crystallin KO cataractous lens samples. Heatmaps were generated from hierarchical cluster analysis to indicate differential expression. The color scale illustrates the relative expression level of mRNAs across different samples. Red denotes upregulation; green denotes downregulation. KO, knockout; WT, wild type.
Table III.
Top 20 upregulated mRNAs in βB2-crystallin KO mice.
| Probe set ID | P-value | Fold-change (KO/WT) | MGI gene symbol | Gene description | GenBank accession no. |
|---|---|---|---|---|---|
| 17477347 | 3.60×10−2 | 7.22 | Klk1b22 | Kallikrein 1-related peptidase b22 | NM_010114 |
| 17303018 | 1.00×10−7 | 7.14 | Gm3500 | Predicted gene 3500 | NM_001256886 |
| 17296943 | 2.52×10−4 | 5.78 | – | – | ENSMUST00000163719 |
| 17303117 | 1.00×10−7 | 5.58 | Gm3696 | Predicted gene 3696 | ENSMUST00000167923 |
| 17296896 | 4.00×10−7 | 5.14 | Gm5796 | Predicted gene 5796 | NM_001029930 |
| 17548311 | 1.22×10−5 | 4.56 | Gm3579 | Predicted gene 3579 | AY140896 |
| 17373996 | 1.35×10−4 | 4.53 | BC048594 | cDNA sequence BC048594 | BC048594 |
| 17331088 | 4.22×10−5 | 4.48 | Gm19797 | Predicted gene 19797 | XM_003085996 |
| 17303024 | 3.66×10−4 | 4.13 | Gm10021 | Predicted gene 10021 | AK084071 |
| 17413352 | 5.14×10−3 | 3.46 | Car9 | Carbonic anhydrase 9 | ENSMUST00000030183 |
| 17296849 | 9.00×10−6 | 3.4 | Gm2897 | Predicted gene 2897 | NM_001177715 |
| 17335467 | 1.21×10−2 | 3.22 | Cdkn1a | Cyclin-dependent kinase inhibitor 1A (P21) | NM_007669 |
| 17412962 | 7.94×10−3 | 3.2 | Gm3893 | Predicted gene 3893 | BC059060 |
| 17496857 | 2.59×10−2 | 3.16 | Cox6a2 | Cytochrome c oxidase, subunit VI a, polypeptide 2 | NM_009943 |
| 17466743 | 9.47×10−4 | 3.13 | Npvf | Neuropeptide VF precursor | ENSMUST00000031853 |
| 17331078 | 3.77×10−4 | 3.06 | Tmem45a | Transmembrane protein 45a | NM_019631 |
| 17296836 | 2.00×10−6 | 3.01 | Gm5458 | Predicted gene 5458 | NM_001024706 |
| 17280292 | 5.05×10−4 | 2.93 | – | – | ENSMUST00000169148 |
| 17296595 | 2.52×10−5 | 2.9 | D830030K20Rik | RIKEN cDNA D830030K20 gene | ENSMUST00000169218 |
| 17303315 | 5.57×10−5 | 2.78 | Gm5797 | Predicted gene 5797 | ENSMUST00000100886 |
KO, knockout; WT, wild type; MGI, mouse genome informatics database.
Table IV.
Top 20 downregulated mRNAs in βB2-crystallin KO mice.
| Probe set ID | P-value | Fold-change (KO/WT) | MGI gene symbol | Gene description | GenBank accession no. |
|---|---|---|---|---|---|
| 17256388 | 3.86×10−2 | 0.86 | Ttc25 | Tetratricopeptide repeat domain 25 | NM_028918 |
| 17283203 | 4.91×10−2 | 0.86 | Foxn3 | Forkhead box N3 | ENSMUST00000046859 |
| 17357502 | 4.94×10−2 | 0.86 | Cpsf7 | Cleavage and polyadenylation specific factor 7 | NM_172302 |
| 17440775 | 4.31×10−2 | 0.86 | Dao | D-amino acid oxidase | ENSMUST00000112292 |
| 17473796 | 4.34×10−2 | 0.86 | Rps5 | Ribosomal protein S5 | NM_009095 |
| 17528663 | 4.79×10−2 | 0.86 | Polr2m | Polymerase (RNA) II (DNA directed) polypeptide M | NM_178602 |
| 17231003 | 4.52×10−2 | 0.85 | Mfsd7b | Major facilitator superfamily domain containing 7B | NM_001081259 |
| 17219005 | 4.68×10−2 | 0.85 | Creg1 | Cellular repressor of E1A-stimulated genes 1 | NM_011804 |
| 17225580 | 4.98×10−2 | 0.85 | Olfr1415 | Olfactory receptor 1415 | NM_001011525 |
| 17256716 | 4.83×10−2 | 0.85 | Rundc1 | RUN domain containing 1 | NM_172566 |
| 17291143 | 3.31×10−2 | 0.85 | – | – | AK029074 |
| 17304186 | 4.98×10−2 | 0.85 | Plac9 | Placenta specific 9 | NM_207229 |
| 17358640 | 4.57×10−2 | 0.85 | Mbl2 | Mannose-binding lectin (protein C) 2 | ENSMUST00000025797 |
| 17368499 | 4.89×10−2 | 0.85 | Dbh | Dopamine beta hydroxylase | ENSMUST00000000910 |
| 17425160 | 3.58×10−2 | 0.85 | Erp44 | Endoplasmic reticulum protein 44 | NM_029572 |
| 17451345 | 4.98×10−2 | 0.85 | 2900026A02Rik | RIKEN cDNA 2900026A02 gene | NM_172884 |
| 17501800 | 3.53×10−2 | 0.85 | Hapln4 | Hyaluronan and proteoglycan link protein 4 | NM_177900 |
| 17499224 | 3.82×10−2 | 0.85 | F10 | Coagulation factor X | NM_001242368 |
| 17504309 | 3.96×10−2 | 0.85 | Ccdc113 | Coiled-coil domain containing 113 | NM_172914 |
| 17502071 | 4.01×10−2 | 0.85 | – | – | ENSMUST00000050921 |
KO, knockout; WT, wild type; MGI, mouse genome informatics database.
Discussion
In the present study, the lncRNA and mRNA profiles of untreated and CRYBB2 KO cataractous lenses were evaluated. A total of 149 lncRNAs and 803 mRNAs were identified to be upregulated, while 180 lncRNAs and 732 mRNAs were identified to be downregulated in CRYBB2 KO mice lenses, implying a potential role of these lncRNAs and mRNAs in cataract formation.
In previous research, an increasing number of lncRNAs have been identified and associations between lncRNAs and numerous diseases, including cardiovascular and neurodegeneration diseases, have been reported (27). The roles of lncRNAs in cancer development are being studied (28,29). However, the function of lncRNAs in disease, particularly in cataracts, has not yet been reported. To the best of our knowledge, the current study presents the first report on differential lncRNA expression in a cohort of mice with or without CRYBB2 KO. Through an analysis of lenses, it was identified that 329 lncRNAs were differentially expressed in CRYBB2 KO and untreated mice, suggesting that lncRNAs may serve critical functions in cataract formation. Among the top 20 most upregulated lncRNAs, five were predicted genes and a further six were unnamed. Among the top 20 most downregulated lncRNAs, 13 lncRNAs were unnamed and the others were known (identified with Mouse Genome Informatics gene symbols). These results indicated that these lncRNAs were linked with CRYBB2-associated cataract formation. Notably, the expression changes of lncRNAs in the upregulated group (maximum change, 5.84-fold) were higher compared with those in the downregulated group (maximum change, 0.86-fold), suggesting a higher susceptibility of lncRNAs to be upregulated rather than downregulated in cataracts.
Differential mRNA expression was also examined in the cohort of mice with or without CRYBB2 KO. Through an analysis of lenses, it was identified that 1,535 mRNAs were differentially expressed between CRYBB2 KO and untreated mice. Among the top 20 most upregulated mRNAs, 10 mRNAs were predicted genes and two mRNAs were unnamed. These results indicated that these mRNAs may serve critical functions in cataract formation. Notably, the expression changes of mRNAs in the upregulated group (maximum change, 7.22-fold) were higher compared with those in the downregulated group (maximum change, 0.86-fold), suggesting a higher susceptibility of mRNAs to be upregulated rather than downregulated in cataracts.
A previous limited microarray survey with a panel of cell cycle-regulated genes illustrated that irradiation with protons altered the gene expression pattern of human lens epithelial cells (30), such as cyclin-dependent kinase inhibitor 1 (CDKN1A), which codes for a protein that is involved in several pathways functionally associated with linear energy transfer-responsive radiation damage. Cytochrome C oxidase 6A2 (COX6A2) was identified to be upregulated during cataract development in mice with a mutation in MIP, a functional water channel that serves a key role in establishing lens fiber cell architecture and is associated with inherited and age-related forms of cataracts (31). Consistent with these results, the present study also identified upregulated expression of CDKN1A and COX6A2 in a CRYBB2 KO-induced cataract mice model. Furthermore, Fas-mediated apoptosis in human lens epithelial cells of cataracts is associated with diabetic retinopathy (32), suggesting a role for Fas in cataract formation, which is contrary to the finding of the present study that Fas was downregulated in a CRYBB2 KO-induced cataracts mouse model (data not shown). BFSP2, a gene for a lens-specific beaded filament structural protein, was down-regulated in CRYBB2 KO-induced cataract mice (33,34), which is in agreement with the findings of the present study: BFSP2 expression is restricted to the lens fiber cells, and a deletion mutation of BFSP2 is associated with cataracts. CRYGD mutation has previously been observed to cause autosomal dominant congenital cerulean cataracts, suggesting an inhibitory role of CRYGD in cataract formation (35). This is consistent with the current findings that CRYGD is downregulated in a CRYBB2 KO-induced cataracts mouse model (data not shown).
The present study has some limitations, including the relatively small number of mice in each cohort, and the fact that only RNA samples from the lens were utilized for hybridizations. Furthermore, the differentially expressed lncRNAs and mRNAs require further clarification in future investigations.
In conclusion, knowledge of the changes in lncRNA and mRNA expression associated with cataracts may contribute to a better understanding of the opacification process. The findings of the present study demonstrate that there are notable lncRNA and mRNA differences between mice with or without CRYBB2 KO induction. The data indicate that the response of the lens to the development of CRYBB2 KO-related cataract is characterized by an extensive upregulation of numerous mRNAs and lncRNAs.
Acknowledgements
Not applicable.
Funding
This study was supported by grants from the National Science Foundation of China (grant nos. 81300748 and 81170834).
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Authors' contributions
YJ conceived of and designed the experiments. KX, H-XR and W-JL performed the experiments and analyzed the data. W-JL and KX obtained the reagents, materials and analysis tools. YJ and W-JL wrote the study. All authors read and approved the final study.
Ethics approval and consent to participate
All procedures were carried out in accordance with the Chinese legislation on the Use and Care of Laboratory Animals and the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research (23) and were approved by the Institutional Animal Care and Use Committee of Changhai Hospital, Second Military Medical University (Shanghai, China).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Reddy MA, Francis PJ, Berry V, Bhattacharya SS, Moore AT. Molecular genetic basis of inherited cataract and associated phenotypes. Surv Ophthalmol. 2004;49:300–315. doi: 10.1016/j.survophthal.2004.02.013. [DOI] [PubMed] [Google Scholar]
- 2.Holmes JM, Leske DA, Burke JP, Hodge DO. Birth prevalence of visually significant infantile cataract in a defined US population. Ophthalmic Epidemiol. 2003;10:67–74. doi: 10.1076/opep.10.2.67.13894. [DOI] [PubMed] [Google Scholar]
- 3.Foster A, Gilbert C, Rahi J. Epidemiology of cataract in childhood: A global perspective. J Cataract Refract Surg. 1997;23(Suppl 1):S601–S604. doi: 10.1016/S0886-3350(97)80040-5. [DOI] [PubMed] [Google Scholar]
- 4.Peng CH, Liu JH, Woung LC, Lin TJ, Chiou SH, Tseng PC, Du WY, Cheng CK, Hu CC, Chien KH, Chen SJ. MicroRNAs and cataracts: Correlation among let-7 expression, age and the severity of lens opacity. Br J Ophthalmol. 2012;96:747–751. doi: 10.1136/bjophthalmol-2011-300585. [DOI] [PubMed] [Google Scholar]
- 5.You C, Wu X, Zhang Y, Dai Y, Huang Y, Xie L. Visual impairment and delay in presentation for surgery in chinese pediatric patients with cataract. Ophthalmology. 2011;118:17–23. doi: 10.1016/j.ophtha.2010.04.014. [DOI] [PubMed] [Google Scholar]
- 6.Gao X, Cheng J, Lu C, Li X, Li F, Liu C, Zhang M, Zhu S, Ma X. A novel mutation in the connexin 50 gene (GJA8) associated with autosomal dominant congenital nuclear cataract in a Chinese family. Curr Eye Res. 2010;35:597–604. doi: 10.3109/02713681003725831. [DOI] [PubMed] [Google Scholar]
- 7.Chen C, Sun Q, Gu M, Liu K, Sun Y, Xu X. A novel Cx50 (GJA8) p. H277Y mutation associated with autosomal dominant congenital cataract identified with targeted next-generation sequencing. Graefes Arch Clin Exp Ophthalmol. 2015;253:915–924. doi: 10.1007/s00417-015-3019-x. [DOI] [PubMed] [Google Scholar]
- 8.Sheets NL, Chauhan BK, Wawrousek E, Hejtmancik JF, Cvekl A, Kantorow M. Cataract-and lens-specific upregulation of ARK receptor tyrosine kinase in Emory mouse cataract. Invest Ophthalmol Vis Sci. 2002;43:1870–1875. [PMC free article] [PubMed] [Google Scholar]
- 9.Hawse JR, Padgaonkar VA, Leverenz VR, Pelliccia SE, Kantorow M, Giblin FJ. The role of metallothionein IIa in defending lens epithelial cells against cadmium and TBHP induced oxidative stress. Mol Vis. 2006;12:342–349. [PMC free article] [PubMed] [Google Scholar]
- 10.Gilmour DT, Lyon GJ, Carlton MB, Sanes JR, Cunningham JM, Anderson JR, Hogan BL, Evans MJ, Colledge WH. Mice deficient for the secreted glycoprotein SPARC/osteonectin/BM40 develop normally but show severe age-onset cataract formation and disruption of the lens. EMBO J. 1998;17:1860–1870. doi: 10.1093/emboj/17.7.1860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hawse JR, Hejtmancik JF, Horwitz J, Kantorow M. Identification and functional clustering of global gene expression differences between age-related cataract and clear human lenses and aged human lenses. Exp Eye Res. 2004;79:935–940. doi: 10.1016/j.exer.2004.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ruotolo R, Grassi F, Percudani R, Rivetti C, Martorana D, Maraini G, Ottonello S. Gene expression profiling in human age-related nuclear cataract. Mol Vis. 2003;9:538–548. [PubMed] [Google Scholar]
- 13.Pauli S, Söker T, Klopp N, Illig T, Engel W, Graw J. Mutation analysis in a German family identified a new cataract-causing allele in the CRYBB2 gene. Mol Vis. 2007;13:962–967. [PMC free article] [PubMed] [Google Scholar]
- 14.Lou D, Tong J, Zhang L, Chiang SW, Lam DS, Pang C. A novel mutation in CRYBB2 responsible for inherited coronary cataract. Eye (Lond) 2009;23:1213–1220. doi: 10.1038/eye.2008.222. [DOI] [PubMed] [Google Scholar]
- 15.Zhang J, Li J, Huang C, Xue L, Peng Y, Fu Q, Gao L, Zhang J, Li W. Targeted knockout of the mouse betaB2-crystallin gene (Crybb2) induces age-related cataract. Invest Ophthalmol Vis Sci. 2008;49:5476–5483. doi: 10.1167/iovs.08-2179. [DOI] [PubMed] [Google Scholar]
- 16.Mothobi ME, Guo S, Liu Y, Chen Q, Yussuf AS, Zhu X, Fang Z. Mutation analysis of congenital cataract in a Basotho family identified a new missense allele in CRYBB2. Mol Vis. 2009;15:1470–1475. [PMC free article] [PubMed] [Google Scholar]
- 17.Rinn JL, Chang HY. Genome regulation by long noncoding RNAs. Ann Rev Biochem. 2012;81:145–166. doi: 10.1146/annurev-biochem-051410-092902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wilusz JE, Sunwoo H, Spector DL. Long noncoding RNAs: Functional surprises from the RNA world. Genes Dev. 2009;23:1494–1504. doi: 10.1101/gad.1800909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mercer TR, Dinger ME, Mattick JS. Long non-coding RNAs: Insights into functions. Nat Rev Genet. 2009;10:155–159. doi: 10.1038/nrg2521. [DOI] [PubMed] [Google Scholar]
- 20.Ponting CP, Oliver PL, Reik W. Evolution and functions of long noncoding RNAs. Cell. 2009;136:629–641. doi: 10.1016/j.cell.2009.02.006. [DOI] [PubMed] [Google Scholar]
- 21.Shen Y, Dong LF, Zhou RM, Yao J, Song YC, Yang H, Jiang Q, Yan B. Role of long non-coding RNA MIAT in proliferation, apoptosis and migration of lens epithelial cells: A clinical and in vitro study. J Cell Mol Med. 2016;20:537–548. doi: 10.1111/jcmm.12755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang J, Huang C, Li W, Wang J, Weng W. Establishment of a βB2 crystallin gene knockout mice model. Acad J Second Mil Med Univ. 2006;27:1246–1249. [Google Scholar]
- 23.Delgado D, del Pozo-Rodríguez A, Solinís MÁ, Avilés-Triqueros M, Weber BH, Fernández E, Gascón AR. Dextran and protamine-based solid lipid nanoparticles as potential vectors for the treatment of X-linked juvenile retinoschisis. Hum Gene Ther. 2012;23:345–355. doi: 10.1089/hum.2011.115. [DOI] [PubMed] [Google Scholar]
- 24.Chomczynski P, Sacchi N. The single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction: Twenty-something years on. Nat Protoc. 2006;1:581–585. doi: 10.1038/nprot.2006.83. [DOI] [PubMed] [Google Scholar]
- 25.Eisen MB, Spellman PT, Brown PO, Botstein D. Cluster analysis and display of genome-wide expression patterns. Proc Natl Acad Sci USA. 1998;95:14863–14868. doi: 10.1073/pnas.95.25.14863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tusher VG, Tibshirani R, Chu G. Significance analysis of microarrays applied to the ionizing radiation response. Proc Natl Acad Sci USA. 2001;98:5116–5121. doi: 10.1073/pnas.091062498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chen G, Wang Z, Wang D, Qiu C, Liu M, Chen X, Zhang Q, Yan G, Cui Q. LncRNADisease: A database for long-non-coding RNA-associated diseases. Nucleic Acids Res. 2013;41:D983–D986. doi: 10.1093/nar/gks1099. (Database issue) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gupta RA, Shah N, Wang KC, Kim J, Horlings HM, Wong DJ, Tsai MC, Hung T, Argani P, Rinn JL, et al. Long non-coding RNA HOTAIR reprograms chromatin state to promote cancer metastasis. Nature. 2010;464:1071–1076. doi: 10.1038/nature08975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kogo R, Shimamura T, Mimori K, Kawahara K, Imoto S, Sudo T, Tanaka F, Shibata K, Suzuki A, Komune S, et al. Long noncoding RNA HOTAIR regulates polycomb-dependent chromatin modification and is associated with poor prognosis in colorectal cancers. Cancer Res. 2011;71:6320–6326. doi: 10.1158/0008-5472.CAN-11-1021. [DOI] [PubMed] [Google Scholar]
- 30.Chang PY, Bjornstad KA, Rosen CJ, McNamara MP, Mancini R, Goldstein LE, Chylack LT, Blakely EA. Effects of iron ions, protons and X rays on human lens cell differentiation. Radiat Res. 2005;164:531–539. doi: 10.1667/RR3368.1. [DOI] [PubMed] [Google Scholar]
- 31.Zhou Y, Bennett TM, Shiels A. Lens ER-stress response during cataract development in Mip-mutant mice. Biochim Biophys Acta. 2016;1862:1433–1442. doi: 10.1016/j.bbadis.2016.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Okamura N, Ito Y, Shibata MA, Ikeda T, Otsuki Y. Fas-mediated apoptosis in human lens epithelial cells of cataracts associated with diabetic retinopathy. Med Electron Microsc. 2002;35:234–241. doi: 10.1007/s007950200027. [DOI] [PubMed] [Google Scholar]
- 33.Jakobs PM, Hess JF, FitzGerald PG, Kramer P, Weleber RG, Litt M. Autosomal-dominant congenital cataract associated with a deletion mutation in the human beaded filament protein gene BFSP2. Am J Hum Genet. 2000;66:1432–1436. doi: 10.1086/302872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zhang L, Gao L, Li Z, Qin W, Gao W, Cui X, Feng G, Fu S, He L, Liu P. Progressive sutural cataract associated with a BFSP2 mutation in a Chinese family. Mol Vis. 2006;12:1626–1631. [PubMed] [Google Scholar]
- 35.Santana A, Waiswol M, Arcieri ES, de Vasconcellos Cabral JP, de Melo Barbosa M. Mutation analysis of CRYAA, CRYGC, and CRYGD associated with autosomal dominant congenital cataract in Brazilian families. Mol Vis. 2009;15:793–800. [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.


