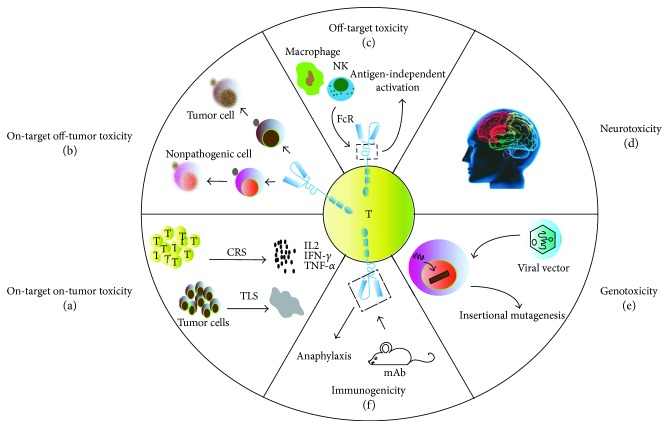
Figure 1.
Toxicities of T cells genetically modified with CARs. (a) On-target on-tumor toxicity. (a1) Effector T-cell activation and excessive cytokine release may result in cytokine release syndrome (CRS). (a2) High tumor load leads to massive destruction of tumor tissue, resulting in tumor lysis syndrome (TLS). (b) On-target off-tumor toxicity: the shared target antigen is also expressed on nonpathogenic cell, subsequently damaging healthy tissue. (c) Off-target toxicity: the extracellular crystallizable fragment (Fc) of CARs can interact with the Fc receptor (FcR) expressed on innate immune cells, leading to antigen-independent activation. (d) Neurotoxicity: manifestation ranges from confusion, delirium, aphasia to some degree of myoclonus, and seizure. (e) Genotoxicity: integrating viral vectors used to facilitate the stable expression in primary T cells may pose a potential risk of oncogenic insertional mutagenesis. (f) Immunogenicity: single-chain variable fragments (scFvs) derive from mouse monoclonal antibodies (mAbs), leading to severe immune response.