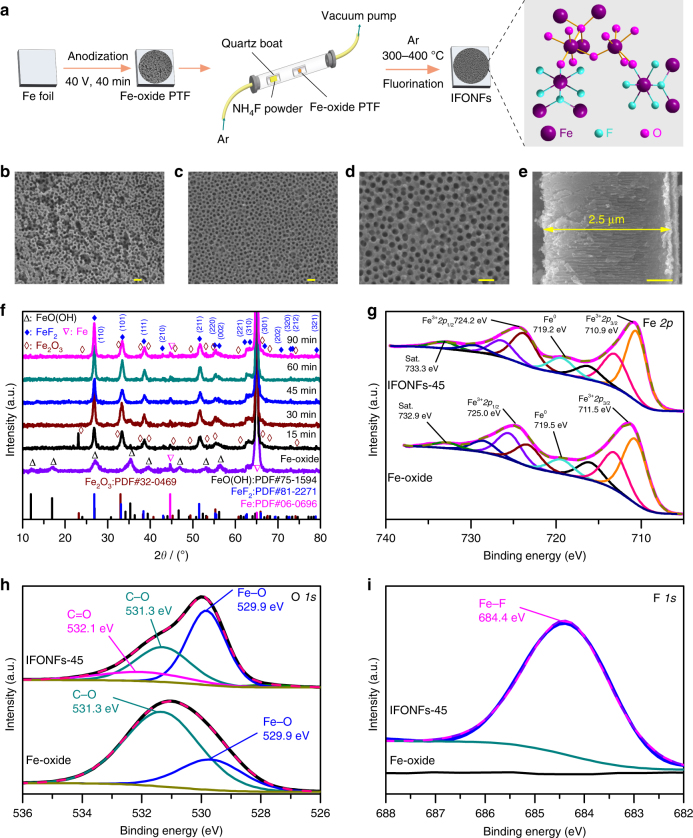
Fig. 1.
IFONFs fabricated from commercial Fe foil with anodization/fluorination process. a Schematic of the fabrication process for IFONFs, starting with a Fe foil. SEM top-view images of b Fe-oxide PTF and c IFONFs-45. Scale bar, 100 nm. d High-magnification SEM image of IFONFs-45. Scale bar, 100 nm. e SEM cross-sectional image of IFONFs-45. Scale bar, 500 nm. f XRD spectrum of Fe-oxide PTF and IFONFs synthesized with various Tfluorinated. For Fe-oxide PTF, the diffraction peaks at 2θ = 11.9°, 16.9°, 26.9°, 35.3°, 39.4°, 46.6°, 53.1°, and 56.2° suggests the formation of FeO(OH) (PDF#75–1594) with anodization. The Fe-oxide PTF anodically grown on Fe substrates consists of FeO(OH) and Fe, while the Fe peaks are from Fe foil. With short Tfluorinated of 15 min, the diffraction peaks of both Fe2O3 and FeF2 are sharp and intense, and Fe2O3 is obtained by annealing FeO(OH) in an argon atmosphere during fluorination47. With Tfluorinated of 45 min, the primary diffraction peaks associated with Fe2O3 are declined while both peaks for FeF2 (110) and (101) are distinguished, indicating a Tfluorinated-dependent phase transformation for iron fluoride-oxide in fluorination condition. Fe2O3 and FeF2 could coexist with fluorination, and FeF2 phase has a tetragonal structure with a P42/mnm space group (PDF#81–2271). High-resolution XPS in g Fe 2p, h O 1s, and i F 2p regions for Fe-oxide PTF and IFONFs-45, respectively. For IFONFs-45 in g, two core-level signals of Fe 2p located at ∼710.9 and 724.2 eV are attributed to Fe 2p3/2 and Fe 2p1/2, which are negatively shift ~0.6 and ~0.8 eV relative to those in the raw Fe oxide, respectively, owing to the bond formation of Fe and F elements. The satellite peaks at ∼719.9 and 733.3 eV are attributed to Fe0, suggesting the presence of elemental Fe on the surface. Moreover, the O 1s peak of IFONFs-45 in h is shifted to lower binding energy with respect to the Fe-oxide PTF, which attributes to the formation of FeF2 phase