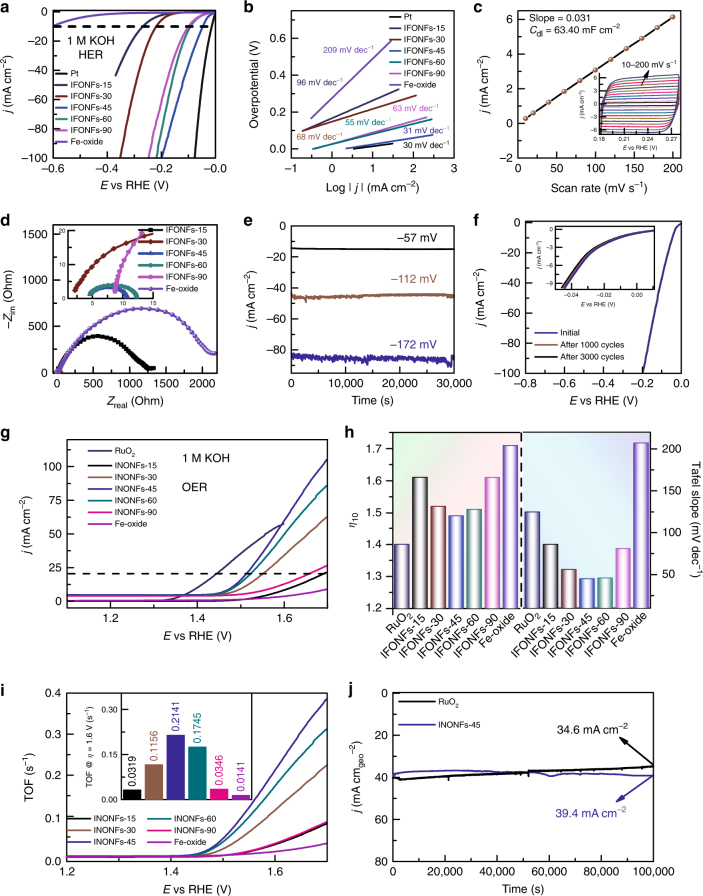
Fig. 4.
Electrochemical HER and OER activity of IFONFs. a Polarization curves measured at 50 mV s−1 in 1 M KOH aqueous solution, and b the corresponding Tafel slopes. c The capacitive current at 0.23 V (vs RHE) as a function of scan rate for IFONFs-45 (Δj0 = ja − jc). The inset is CVs for IFONFs-45 with different rates from 10 to 200 mV s−1. d Nyquist plots measured at a voltage −5 mV (vs RHE) over the frequency range 1000–0.01 Hz in 1 M KOH. The inset is enlarged area denoted by dash squares. e CV cycle-dependent current density at different potentials with 30,000 s in 1 M KOH aqueous solution. f Polarization curves before and after 3000 CV cycles of IFONFs-45 ranging from 0 to 0.2 V vs RHE. g LSV curves measured at 5 mV s−1 in 1 M KOH aqueous solution, h the corresponding η10 (left) and Tafel slopes (right) for RuO2, IFONFs and Fe-oxide catalysts. i TOF per oxidative iron site for IFONFs and Fe-oxide catalysts. The inset reveals the TOF values at η = 1.6 V. j Time-dependent current density curve of IFONFs-45 and RuO2 at a fixed overpotential of 1.56 and 1.52 V to drive 40 mA cm−2, respectively. dec, decade