Abstract
Objective
Presumptive formats to initiate childhood vaccine discussions (e.g. “Well we have to do some shots.”) have been associated with increased vaccine acceptance after one visit compared to participatory formats (e.g. “How do you feel about vaccines?”). We characterize discussion format patterns over time and the impact of their repeated use on vaccine acceptance.
Methods
We conducted a longitudinal prospective cohort study of children of vaccine-hesitant parents enrolled in a Seattle-based integrated health system. After the child’s 2, 4, and 6 month visits, parents reported the format their child’s provider used to begin the vaccine discussion (presumptive, participatory, or other). Our outcome was the percentage of days under-immunized of the child at 8 months old for 6 recommended vaccines. We used linear regression and generalized estimating equations to test the association of discussion format and immunization status.
Results
We enrolled 73 parent/child dyads and obtained data from 82%, 73%, and 53% after the 2, 4, and 6 month visits, respectively. Overall, 65% of parents received presumptive formats at ≥1 visit and 42% received participatory formats at ≥1 visit. Parental receipt of presumptive formats at 1 and ≥2 visits (vs. no receipt) was associated with significantly less under-immunization of the child, while receipt of participatory formats at ≥2 visits was associated with significantly more under-immunization. Visit-specific use of participatory (vs. presumptive) formats was associated with a child being 10.1% (95% CI: 0.3, 19.8; P=.04) more days under-immunized (amounting to, on average, 98 more days under-immunized for all 6 vaccines combined).
Conclusions
Presumptive (vs. participatory) discussion formats are associated with increased immunization.
Keywords: Immunization, Health Communication, Preventive Health Services
Parental refusal or delay of childhood vaccines is both prevalent1–3 and a key contributor to the persistence of outbreaks of vaccine-preventable disease (VPD).4 Children who are underimmunized due to parental vaccine refusal or delay have higher inpatient admission and emergency department utilization rates,5 increased morbidity,6–8 and have died from vaccine-preventable disease.9 As a result, increasing parent acceptance of childhood vaccines to reduce VPD is a top public health priority in the US.10
The identification of provider communication strategies that are effective at improving parental acceptance of childhood vaccines is critical to addressing this priority. Pediatric providers are consistently cited by parents as the most important influence on their vaccine decision-making.11 In addition, parents report changing their mind about delaying or refusing a vaccine after their child’s provider addressed their concerns or gave them reassurance.1,12
In previous work, we identified the format used to initiate the childhood vaccine discussion as a potentially effective vaccine communication strategy.13–15 In particular, provider use of presumptive (e.g. “Well we have to do some shots.”) rather than participatory (e.g. “How do you feel about vaccines today?”) formats was associated with increased parental vaccine acceptance of childhood vaccines.13,14 Limitations of this previous work, however, included observing only a single vaccine visit and measuring parental verbal acceptance of vaccines rather than actual child immunization status. In the present study, we sought to address these limitations by characterizing provider discussion format over time and its association with a child’s immunization status. We hypothesized that use of presumptive formats for initiating the childhood vaccine discussion over multiple visits would be associated with increased immunization.
Methods
We conducted a longitudinal prospective cohort study from August 2014 – March 2017 in which we followed children of vaccine-hesitant parents (VHPs) over 3 health supervision visits at an integrated health care delivery organization (Group Health; GH) that covers approximately 600,000 members in Washington and Idaho states (in February 2017, GH became Kaiser Permanente). We associated parent-reported provider discussion format of the childhood vaccine discussion during these visits with the timeliness of immunizations received by the child by 8 months of age. The GH Human Subjects Review Committee formally reviewed and approved all study procedures, and the Washington State Institutional Review Board formally reviewed and approved study procedures involving Washington State Immunization Information System (WAIIS) data.
Study Participants
Trained research assistants approached parents of singleton newborns born at ≥35 weeks gestational age either in-person prior to discharge from the GH newborn nursery in Seattle, WA or by phone within 6 weeks of their child’s birth to participate in a study about how doctors and parents talk about vaccines. Phone recruitment was used exclusively after the nursery closed in January 2015. Parents were eligible if they were a) ≥18 years old, b) English-speaking, c) GH members planning to have their child receive health supervision through 8 months of age with a GH pediatrician or family physician as well as accompany their child to the 2, 4, and 6 month health supervision visits, and d) a VHP (defined as a score of ≥50 [on a 0-100 scale] on the validated Parent Attitudes about Childhood Vaccines [PACV] survey administered at the time of enrollment, consistent with previous studies in which PACV scores ≥50 were associated with significantly higher under-immunization compared to scores <502,3). We restricted eligibility to VHPs because they represent an important population for interventions aimed at improving vaccine acceptance.16 We included only parents of newborns in order to focus on provider vaccine communication behaviors used at the first health supervision visits when the majority of the primary series of recommended vaccines begin.17 Lastly, we excluded parents who planned to have another person (e.g. a grandparent) solely accompany the child to a visit since there was no reliable way to follow up with non-parents. Parents provided written informed consent upon in-person enrollment and oral consent upon phone enrollment.
Instrument Development and Evaluation
Prior to the start of the study, we developed and validated a parent self-report instrument to identify the format their child’s doctor used to initiate the vaccine discussion. All instrument items were written according to accepted guidelines for survey development,18 formatted according to accepted criteria for reducing non-response and avoiding measurement error,19 and initially revised after pretesting with a convenience sample of 44 English-speaking parents. We revised the instrument further (e.g. response categories for each initiation format listed were changed from a 5-point Likert scale from strongly agree to strongly disagree to ‘choose only one’) and created a telephone script to accompany its administration to improve its accuracy after further pretesting it with VHPs (N=20).
We tested the accuracy of the final version of the parent self-report instrument (Appendix 1) by administering it to a separate cohort of VHPs (N=77) of 1-19 month old children after a videotaped health supervision visit with one of 17 participating pediatric providers at 12 non-GH Seattle primary care practices. Of these 77 VHPs, 49 were administered the instrument via phone within 3 days of the visit. This validation study was described generally to parents as one seeking to understand how their child’s doctor talks with them during their child’s check-up in order to minimize the likelihood that participants altered their behavior to meet observer expectations. We compared parent-reported discussion format to the gold standard of observed discussion format using the videotapes of the same visits. Two of us (DJO, JDR) separately coded videotaped encounters using our previously developed interaction coding scheme for the childhood vaccine discussion13,20 and then met to compare coding. We used the kappa statistic (k) to measure inter-rater agreement.21 After the first 20% of encounters, a k of ≥0.7 was reached, suggesting greater than moderate agreement.22 The remaining encounters were coded by the principal investigator (DJO), with a second investigator (JDR) co-coding a random selection of encounters to ensure coding accuracy. The accuracy of the phone-administered parent instrument in identifying provider use of participatory and presumptive discussion formats was 77% (95% Confidence Interval [CI]: 60%, 90%).
Data Collection
Parent demographics were collected at baseline, including parent age, relationship to child, marital status, educational level, household income, ethnicity, race, and number of children in the household. Using computer-assisted telephone interviewing software and standardized interview procedures23 consistent with those developed and used during the validation study of the parent self-report instrument, trained research assistants administered the instrument to enrolled parents 7 days a week beginning the day of their child’s 2, 4, and 6 month health supervision visits. Similar to the validation study of the instrument, the initial attempts for contacting parents by phone were made within 3 days of their child’s visit in order to minimize measurement error resulting from recall bias,24 with the window for contacting parents ending 1 week post-visit. The average number of days from visit to phone call was 2.4 days for the 2 month visit, 2.1 days for the 4 month visit, and 3.2 days for the 6 month visit. There were 12 instances in which the 1 week window was exceeded because parents called back outside this timeframe after our final attempt to reach them was made on or before day 7. There was no change in our results when excluding these late interviews, so they were retained. With each post-visit call, research assistants confirmed that the child of the enrolled parent just completed a health supervision visit and that the parent accompanied the child to the visit. Parents were also asked whether shots were discussed at the visit, and for those who responded no, questions about the format used to initiate the vaccine discussion were skipped. Lastly, whether the visit was with the child’s regular doctor and whether a first-time vaccine discussion with the doctor occurred was also recorded because these factors may influence provider communication behaviors and child immunization status.13,14
Outcome
When a child of an enrolled parent reached 8 months, 0 days, we assessed their immunization status using their GH electronic immunization record for each of the 16 doses of the 6 vaccines recommended at that age since birth: 2 doses of hepatitis B vaccine, 3 doses of rotavirus vaccine, 3 doses of diphtheria, tetanus toxoids, and acellular pertussis vaccine, 3 doses of Haemophilus influenzae type b vaccine, 3 doses of pneumococcal conjugate vaccine, and 2 doses of inactivated poliovirus vaccine.17 GH immunization records have been electronic since 1991, have been updated from the GH electronic medical record system since 2005, and receive data from WAIIS monthly on vaccinations received outside GH to ensure completeness.25 GH immunization records are also typically updated with WAIIS data on patients who dis-enroll from GH until the patient joins a new health plan. However, to ensure immunization record accuracy, we separately obtained WAIIS data of child participants whose parents provided data from at least 1 visit and didn’t remain continuous GH members (defined as a <63 day gap in membership from enrollment until 8 months old of age) in order to perform an additional cross-reference with GH immunization records.
We expressed child immunization status as the percentage of days under-immunized from birth to 8 months, 0 days for all 6 vaccines combined using a method adapted from a prior study.26 This measure captures both refusal and delay in receipt of recommended doses by calculating the difference between the age in days the dose was received and the latest age in which it should have been received according to the recommended immunization schedule. If a dose was never received, the maximum number of days late a child could be for that dose was their age in days at 8 months, 0 days (244 days) minus the latest age in days in which that dose should have been received. To obtain the percentage of days under-immunized from 0 – 8 months, we summed the days late for each dose of the 6 vaccines and divided this by the maximum number of days a child could be late if they had received none of the total 16 doses for the 6 vaccines by 8 months (967 days). We calculated the percentage of days under-immunized from birth to 4 months, 0 days and birth to 6 months, 0 days in the same manner to determine the maximum number of days a child could be late if they had received none of the recommended doses for the 6 vaccines by 2 months (235 days) and 4 months (601 days), respectively. Lastly, to account for use of a catch-up immunization schedule after a first late dose, we performed a lenient calculation of days under-immunized by adding 28 additional days to the minimum interval number of days between doses for that vaccine.
Data Analysis
For all analyses, the child was the unit of analysis. Our primary exposure variable was the parent-reported discussion format providers used to initiate the childhood vaccine discussion, categorized as presumptive, participatory, or other (Appendix 1). We used descriptive statistics to summarize parent and visit characteristics as well as our predictor and outcome variables at each time point and overall. We assessed the bivariate relationships between parent and visit characteristics and our predictor variable using Pearson’s χ2 tests (or Fisher’s exact tests) and these same characteristics and our outcome variable using t-tests. In these analyses, we dichotomized PACV scores as 50-69 and 70-100, consistent with a prior PACV validation study in which higher under-immunization was associated with higher PACV score tier.3 We also assessed for a dose-response relationship between immunization status and number of visits (categorized as 0, 1, or ≥2 visits) in which a parent was exposed to presumptive or participatory formats by performing a nonparametric test for trend across both groups.
Parent and visit characteristics that were associated (P<0.10) with discussion format and immunization status in bivariate analysis and were not narrowly distributed27 were retained in multivariate models (parent age, race). We also included variables selected a priori as potential confounders (first-time vaccine discussion, child birth order).13,14,28 We used linear regression to test the association of the number of visits (with 0 visits as the referent group) in which a parent was exposed to a particular discussion format and percent days under-immunized at 8 months after accounting for parent and visit characteristics. Since there was no significant association found between exposure to ‘other’ formats and immunization status, we only present regression results from exposure to presumptive and participatory formats. Also, we found no significant difference when using the lenient calculation of the percentage of days under-immunized, so we only present regression results using the standard calculation. Lastly, we examined the association between the use of a participatory (vs. presumptive) format and immunization status at each visit. For these inferences, we applied generalized estimating equations and robust standard errors to account for repeated assessments within children.29
Results
We enrolled 73 parent/child dyads and obtained data from 82%, 73%, and 53% of participants after the 2, 4, and 6 month visits, respectively (Figure 1). Among parent participants, 92% were mothers, 69% were ≥30 years old, and 58% were white (Table 1). The primary reasons for lost to follow-up were that child participants never had subsequent health supervision visits after enrollment or parent participants were unable to be reached by phone after a visit. Overall, 92% (N=67) of parent participants provided discussion format data from ≥1 visit and 25% (N=18) from all 3 visits.
Figure 1. Study Population.
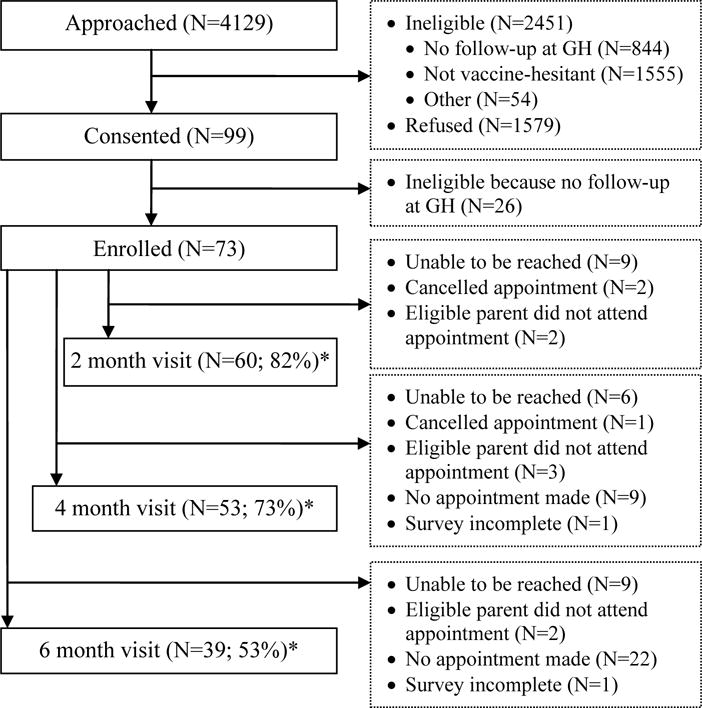
*Of these 152 total data points from the 2, 4 and 6 month visits, there were 19 instances (12%) in which no initiation format data was obtained because the parent reported that no shots were discussed (and therefore the initiation format questions were skipped). Of the remaining 133 data points, there were 2 instances (1%) in which the parent couldn’t remember the initiation format used or responded that shots were not needed (responses 4 and 5 in Appendix A). Therefore, 131 data points were included in analysis.
Table 1.
Parent and Visit Characteristics (N=73)
| Characteristic | N (%) |
|---|---|
| Relationship to Child | |
| Mother | 67 (92) |
| Parent Marital Status | |
| Married or living with a partner | 65 (89) |
| Parent Ageˆ | |
| ≥30 years | 50 (69) |
| Parent Education | |
| ≥4 year college degree | 39 (53) |
| Household Incomeˆ | |
| >$75,000 | 32 (46) |
| Parent Raceˆ | |
| American Indian/Alaskan Native | 2 (3) |
| Asian | 4 (6) |
| Black or African American | 3 (4) |
| Native Hawaiian or other Pacific Islander | 2 (3) |
| White | 41 (58) |
| More than one race | 16 (22) |
| Other | 3 (4) |
| Parent Ethnicity | |
| Hispanic | 11 (15) |
| No. Children in Household | |
| 1 | 34 (47) |
| Child birth order | |
| First born | 37 (51) |
| First-time vaccine discussion | |
| At 2 month visitˆ | 32 (58) |
| At 4 month visitˆ | 11 (26) |
| At 6 month visitˆ | 6 (19) |
| Saw child’s regular doctor | |
| At 2 month visitˆ | 52 (88) |
| At 4 month visitˆ | 42 (79) |
| At 6 month visitˆ | 32 (82) |
| PACV Score: mean (range) | 63.4 (50-93.3) |
| 50-69 | 48 (66) |
| 70-100 | 25 (34) |
Missing data
At each time point, presumptive formats were most frequently used and ‘other’ formats the least frequently used (Figure 2), with these ‘other’ formats consisting primarily of parent (rather than doctor) initiated formats (e.g. “I brought it up” or “I started to talk about the shots before my doctor did”). Among parents with discussion format data from ≥1 visit, 40% (N=27) were exposed only to presumptive formats, 16% (N=11) only to participatory formats, and 10% (N=7) to other formats; 33% (N=22) were exposed to a mix of discussion formats. There were 65% of parents (N=44) who received presumptive formats at ≥1 visit and 42% (N=28) who received participatory formats at ≥1 visit. A significantly higher proportion of White (vs. non-White) parents (49% vs. 15%; P=.008) and parents ≥30 years old (vs. <30 years old; 42% vs. 14%; P=.03) had 0 visits involving presumptive formats, and a significantly higher proportion of mothers (vs. non-mothers; 62% vs. 0%; P=.007) had 0 visits involving participatory formats.
Figure 2.

Discussion Format Over Time (N=131)
Child participants were under-immunized for a mean of 39.2% of days between birth and 8 months of age (standard deviation: 41.2), with 19% remaining completely un-immunized. White (vs. non-White) parents had children who were significantly more under-immunized at 8 months of age (mean values of 51.9% [95% CI: 39.0%, 64.9%] vs. 24.3% [95% CI: 10.7%, 37.9%], respectively; P=.005), and Hispanic (vs. non-Hispanic) parents had children who were significantly less under-immunized at 8 months of age (mean values of 16.5% [95% CI: 2.3%, 30.6%] vs. 43.2% [95% CI: 32.3%, 54.0%], respectively; P=.046). Only PACV score was associated with being completely unimmunized at 8 months of age, with a higher proportion of parents who scored 70-100 (vs. 50-69) having an un-immunized child at 8 months of age (40% vs. 8.3%, respectively; P=.003). Use of participatory (vs presumptive) formats at the 2-month visit was associated with a child being more underimmunized after the 2-month visit (mean values, 60.7% [95% CI, 39.9, 81.5] vs 25.7% [95% CI, 13.1, 38.3], respectively; P = .003), and there was a higher proportion of 4- or 6-month visits in which providers used participatory (vs presumptive) formats with parents who had children who were completely unimmunized after the 2-month visit (30% vs 5%, respectively; P = .02).
There was a dose-response relationship between immunization status and the number of visits involving presumptive formats, with significantly less under-immunization associated with an increasing number of visits involving presumptive formats (P=.003). There was also a dose-response relationship between immunization status and the number of visits involving participatory formats, with significantly more under-immunization associated with an increasing number of visits involving participatory formats (P=.001). In unadjusted regression analyses, children of parents exposed to 1 and ≥2 visits involving presumptive formats were significantly less under-immunized from birth to 8 months of age compared to children of parents who were exposed to 0 visits (Figure 3). Correspondingly, children of parents exposed to 1 and ≥2 visits involving participatory formats were significantly more under-immunized from birth to 8 months of age compared to children of parents who were exposed to 0 visits. After adjusting for demographic and visit characteristics, these associations remained significant except for parents exposed to only 1 visit involving participatory formats. Among all parents, visit-specific use of participatory (vs. presumptive) formats was associated with a child being on average 10.1% (95% CI: 0.3, 19.8; P=.04) more days under-immunized.
Figure 3.

Immunization Status by Discussion Format
Discussion
Our finding that presumptive (vs. participatory) formats to initiate the childhood vaccine discussion are associated with increased immunization over several visits is consistent with our previous results obtained after observing single encounters.13–15 It is also consistent with recent results in the adolescent vaccine context.30,31 Taken together, these studies suggest that presumptive formats for initiating the vaccine discussion are a more effective communication strategy than participatory formats for improving parental vaccine acceptance.
Our results are also noteworthy because of previous concerns that presumptive formats to initiate vaccine discussions may result in decreased vaccine uptake over time by eroding trust or generating feelings of resentment.32 There are several possible explanations for why we did not find decreased vaccine uptake with the repeated use of presumptive formats. First, presumptive formats establish vaccine acceptance as the reference point, or default, for parents’ decision-making. Defaults themselves are influential because they leverage several cognitive biases underlying human decision-making, such as omission and status quo biases,33 which make it difficult to depart from default reference points.34 It may be that these cognitive biases simply prevail in their influence on parental vaccination behavior over other counteracting factors, such as feelings of resentment. Second, presumptive formats need not feel presumptive to parents. Rather, they can be delivered using a variety of positive interpersonal skills, such as sitting at the same level as parents, maintaining eye contact, using a non-judgmental tone, speaking in an unhurried manner, and allowing parents to respond.35 It may be that providers in our study used these techniques when employing presumptive formats, thereby minimizing any negative impact these initiations might have on factors important to vaccine acceptance, such as trust. Lastly, it is possible that vaccine uptake is indeed negatively affected but follow-up beyond 8 months is needed for this effect to be detectable or a longer exposure to presumptive formats (i.e. more than 3 visits) is required. However, this seems unlikely since the 3 visits we observed include the majority of the primary series of vaccines.
We also found that more providers used participatory (vs. presumptive) formats in subsequent visits with parents who had children who were completely un-immunized after the 2 month visit. This is perhaps not surprising because it can feel awkward to begin a subsequent vaccine conversation with a parent who has previously refused vaccines for their child by presuming the parent will now accept them. The importance of beginning the vaccine discussion in these scenarios with a vaccine recommendation, however, deserves further study.
There are several limitations to this study. First, although provider discussion format was measured using a reasonably accurate parent-report instrument, there was likely still some residual measurement error. We did explore whether accuracy of parent-report was associated with parent socio-demographic factors (age, gender, race, education, household income, marital status, and number of children in the household) and found none to be statistically significant. Second, there was attrition over the course of the study resulting in missing data from 30% of observations. This attrition may have been exacerbated by the acquisition of GH by Kaiser Permanente, which was announced in the middle of our study (December 2015) and created uncertainty among GH membership.36 To assess for bias introduced by this attrition, we compared parent demographics and visit characteristics among participants with discussion format data from all 3 time points (N=18) with those who had missing data from at least one visit (N=55) and found no significant differences. Third, given our non-randomized study design, there may be unmeasured variables that are driving the observed association between use of participatory discussion formats and under-immunization. We conducted a sensitivity analyses in which we excluded parents with children who were completely un-immunized at 8 months, under the assumption that some of these unmeasured variables may be factoring more prominently with these parents. Our results, however, were similar: parents exposed to ≥2 visits involving participatory formats still had children who remained significantly more under-immunized at 8 months of age compared to parents exposed to 0 visits involving participatory formats (mean of 36.9%, 95% CI: 11.7, 62.1; P=.005). Fourth, the clinical significance of being 10.1% more days under-immunized from birth to 8 months—which amounts to, on average, 98 more days under-immunized for all 6 vaccines combined—is unclear since the risk of disease due to delay varies by vaccine/vaccine-preventable disease. However, under-immunized children are at some increased risk8 and missing early doses in a vaccine series is associated with missing later ones,37 contributing to more under-immunization over time. Lastly, we conducted our study at a single US institution, and therefore our results may not be generalizable.
Conclusions
Presumptive formats for initiating the childhood vaccine discussion were associated with increased immunization over time. Though these results require confirmation using a randomized trial study design, they add to the growing evidence-base for the effectiveness of presumptive initiation formats in the vaccine context. Studies are needed to evaluate the implementation of the presumptive format in clinical practice as well as interventions that combine the presumptive format with other strategies designed to facilitate parental acceptance of childhood vaccines.
What’s New.
The impact of provider discussion formats over time on childhood immunization status is unknown. We found that presumptive (vs. participatory) formats used at the 2, 4, and/or 6 month visits were associated with increased immunization at 8 months of age.
Acknowledgments
Funding Source: Research reported in this publication was supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development of the National Institutes of Health under award 1K23HD06947 (PI: Opel). The funding source had no role in study design; in the collection, analysis and interpretation of date; in the writing of the report; or in the decision to submit the article for publication.
Appendix 1. Parent-report instrument to identify the type of communication format providers used to initiate the childhood vaccine discussion
(Item Introduction) For this question, I will read you several scenarios. After I read them, please choose the one that best describes what happened during your visit.
| 1 | AT THIS VISIT, your child’s doctor STARTED to talk about the plan for shots by TELLING you which shots your child would be getting without asking your opinion. For example, if your child’s doctor FIRST started to talk about the plan for shots by saying something like “Well, we have to do some shots.” or “He’s due for DTaP, Hib and IPV today.”, choose this response. He or she simply told you what the plan was and didn’t ask you how you felt about that plan. |
| 2 | AT THIS VISIT, your child’s doctor STARTED to talk about the plan for shots by TELLING you which shots your child would be getting and then ASKING your opinion about that shot plan. For example, if your child’s doctor FIRST started to talk about the plan for shots by saying something like “So, he’s due for three shots today. Is that OK with you?”, choose this response. In this instance, he or she told you what the plan was but then asked you what you thought about it. |
| 3 | AT THIS VISIT, your child’s doctor STARTED to talk about the plan for shots by ASKING you what you wanted to do about shots. For example, if your child’s doctor FIRST started to talk about the plan for shots by saying something like “How do you feel about shots?” or “Are we going to do shots today?”, choose this response. |
| 4 | You don’t remember how your child’s doctor started talking about shots AT THIS VISIT. |
| 5 | Your child didn’t need any shots AT THIS VISIT. |
| 6 | None of the above. [interviewer write verbatim what parent said] For example, if you started to talk about the plan for shots first BEFORE your child’s doctor did, such as by saying “So we wanted to just do a few shots today” or “We’re going on vacation tomorrow so I think I would like to get all the vaccines today”, you would choose this response. |
Footnote: Responses 1 and 2 were coded as presumptive formats, response 3 as participatory format, and response 6 as ‘other’ format. Responses 4 and 5 were excluded as missing data.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflicts of Interest: None.
References
- 1.Gust DA, Darling N, Kennedy A, Schwartz B. Parents with doubts about vaccines: which vaccines and reasons why. Pediatrics. 2008;122:718–25. doi: 10.1542/peds.2007-0538. [DOI] [PubMed] [Google Scholar]
- 2.Opel DJ, Taylor JA, Mangione-Smith R, et al. Validity and reliability of a survey to identify vaccine-hesitant parents. Vaccine. 2011;29:6598–605. doi: 10.1016/j.vaccine.2011.06.115. [DOI] [PubMed] [Google Scholar]
- 3.Opel DJ, Taylor JA, Zhou C, Catz S, Myaing M, Mangione-Smith R. The relationship between parent attitudes about childhood vaccines survey scores and future child immunization status: a validation study. JAMA Pediatr. 2013;167:1065–71. doi: 10.1001/jamapediatrics.2013.2483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Phadke VK, Bednarczyk RA, Salmon DA, Omer SB. Association Between Vaccine Refusal and Vaccine-Preventable Diseases in the United States: A Review of Measles and Pertussis. JAMA. 2016;315:1149–58. doi: 10.1001/jama.2016.1353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Glanz JM, Newcomer SR, Narwaney KJ, et al. A population-based cohort study of undervaccination in 8 managed care organizations across the United States. JAMA Pediatr. 2013;167:274–81. doi: 10.1001/jamapediatrics.2013.502. [DOI] [PubMed] [Google Scholar]
- 6.Glanz JM, McClure DL, Magid DJ, Daley MF, France EK, Hambidge SJ. Parental refusal of varicella vaccination and the associated risk of varicella infection in children. Arch Pediatr Adolesc Med. 2010;164:66–70. doi: 10.1001/archpediatrics.2009.244. [DOI] [PubMed] [Google Scholar]
- 7.Glanz JM, McClure DL, O’Leary ST, et al. Parental decline of pneumococcal vaccination and risk of pneumococcal related disease in children. Vaccine. 2011;29:994–9. doi: 10.1016/j.vaccine.2010.11.085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Glanz JM, Narwaney KJ, Newcomer SR, et al. Association between undervaccination with diphtheria, tetanus toxoids, and acellular pertussis (DTaP) vaccine and risk of pertussis infection in children 3 to 36 months of age. JAMA Pediatr. 2013;167:1060–4. doi: 10.1001/jamapediatrics.2013.2353. [DOI] [PubMed] [Google Scholar]
- 9.Invasive Haemophilus influenzae Type B disease in five young children–Minnesota, 2008. MMWR. 2009;58:58–60. [PubMed] [Google Scholar]
- 10.Assessing the State of Vaccine Confidence in the United States: Recommendations from the National Vaccine Advisory Committee: Approved by the National Vaccine Advisory Committee on June 10, 2015. Public Health Rep. 2015;130:573–95. doi: 10.1177/003335491513000606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Freed GL, Clark SJ, Butchart AT, et al. Sources and perceived credibility of vaccine-safety information for parents. Pediatrics. 2011;127(Suppl 1):S107–12. doi: 10.1542/peds.2010-1722P. [DOI] [PubMed] [Google Scholar]
- 12.Fredrickson DD, Davis TC, Arnould CL, et al. Childhood immunization refusal: provider and parent perceptions. Family Med. 2004;36:431–9. [PubMed] [Google Scholar]
- 13.Opel DJ, Heritage J, Taylor JA, et al. The Architecture of Provider-Parent Vaccine Discussions at Health Supervision Visits. Pediatrics. 2013;132:1037–46. doi: 10.1542/peds.2013-2037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Opel DJ, Mangione-Smith R, Robinson JD, et al. The Influence of Provider Communication Behaviors on Parental Vaccine Acceptance and Visit Experience. Am J Public Health. 2015;105:1998–2004. doi: 10.2105/AJPH.2014.302425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hofstetter AM, Robinson JD, Lepere K, et al. Clinician-parent discussions about influenza vaccination of children and their association with vaccine acceptance. Vaccine. 2017;35:2709–15. doi: 10.1016/j.vaccine.2017.03.077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Leask J. Target the fence-sitters. Nature. 2011;473:443–5. doi: 10.1038/473443a. [DOI] [PubMed] [Google Scholar]
- 17.Committee On Infectious Diseases, American Academy of Pediatrics. Recommended Childhood and Adolescent Immunization Schedule-United States, 2017. Pediatrics. 2017;139 doi: 10.1542/peds.2016-4007. [DOI] [PubMed] [Google Scholar]
- 18.Sudman S, Bradburn NM. Asking questions. 1st. San Francisco: Jossey-Bass; 1982. [Google Scholar]
- 19.Dillman DA. Mail and internet surveys: the tailored design method. 2nd. Hoboken, NJ: Wiley; 2007. [Google Scholar]
- 20.Opel DJ, Robinson JD, Heritage J, et al. Characterizing providers’ immunization communication practices during health supervision visits with vaccine-hesitant parents: A pilot study. Vaccine. 2012;30:1269–75. doi: 10.1016/j.vaccine.2011.12.129. [DOI] [PubMed] [Google Scholar]
- 21.Burla L, Knierim B, Barth J, et al. From text to codings: intercoder reliability assessment in qualitative content analysis. Nurs Res. 2008;57:113–7. doi: 10.1097/01.NNR.0000313482.33917.7d. [DOI] [PubMed] [Google Scholar]
- 22.Bernard HR. Research methods in anthropology: qualitative and quantitative methods. 3rd. Walnut Creek, CA: AltaMira Press; 2002. [Google Scholar]
- 23.Groves RM. Survey methodology. Hoboken, NJ: J. Wiley; 2004. [Google Scholar]
- 24.DiMatteo MR, Robinson JD, Heritage J, et al. Correspondence among patients’ self-reports, chart records, and audio/videotapes of medical visits. Health Commun. 2003;15:393–413. doi: 10.1207/S15327027HC1504_02. [DOI] [PubMed] [Google Scholar]
- 25.Jackson ML, Henrikson NB, Grossman DC. Evaluating Washington State’s immunization information system as a research tool. Acad Pediatr. 2014;14:71–6. doi: 10.1016/j.acap.2013.10.002. [DOI] [PubMed] [Google Scholar]
- 26.Luman ET, Barker LE, Shaw KM, et al. Timeliness of childhood vaccinations in the United States: days undervaccinated and number of vaccines delayed. JAMA. 2005;293:1204–11. doi: 10.1001/jama.293.10.1204. [DOI] [PubMed] [Google Scholar]
- 27.Harrell FE. Regression Modeling Strategies: With Applications to Linear Models, Logistic Regression, and Survival Analysis. New York NY: Springer-Verlag New York, Inc; 2011. [Google Scholar]
- 28.Opel DJ, Mangione-Smith R, Taylor JA, et al. Development of a survey to identify vaccine-hesitant parents: The parent attitudes about childhood vaccines survey. Hum Vaccin. 2011;7:419–25. doi: 10.4161/hv.7.4.14120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fitzmaurice GM, Laird NM, Ware JH. Applied longitudinal analysis. Second. Wiley; 2011. [Google Scholar]
- 30.Sturm L, Donahue K, Kasting M, et al. Pediatrician-Parent Conversations About Human Papillomavirus Vaccination: An Analysis of Audio Recordings. J Adolesc Health. 2017;61(2):246–251. doi: 10.1016/j.jadohealth.2017.02.006. [DOI] [PubMed] [Google Scholar]
- 31.Brewer NT, Hall ME, Malo TL, et al. Announcements Versus Conversations to Improve HPV Vaccination Coverage: A Randomized Trial. Pediatrics. 2017;139(1) doi: 10.1542/peds.2016-1764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Leask J, Kinnersley P, Willaby HW, Danchin M. Presumptive initiations in vaccine discussions with parents: acquiescence but at what cost? [E-letter] Pediatrics. 2013 [Google Scholar]
- 33.Baron J, Ritov I. Reference points and omission bias. Organizational Behavior and Human Decision Processes. 1994;59:475–98. doi: 10.1006/obhd.1999.2839. [DOI] [PubMed] [Google Scholar]
- 34.Halpern SD, Ubel PA, Asch DA. Harnessing the power of default options to improve health care. NEJM. 2007;357:1340–4. doi: 10.1056/NEJMsb071595. [DOI] [PubMed] [Google Scholar]
- 35.Bryant KA, Wesley GC, Wood JA, et al. Use of standardized patients to examine physicians’ communication strategies when addressing vaccine refusal: a pilot study. Vaccine. 2009;27:3616–9. doi: 10.1016/j.vaccine.2009.03.048. [DOI] [PubMed] [Google Scholar]
- 36.Aleccia J. Group Health acquisition by Kaiser draws worry and praise. Seattle Times. 2015 Dec 5; [Google Scholar]
- 37.Strine TW, Luman ET, Okoro CA, et al. Predictors of age-appropriate receipt of DTaP dose 4. Am J Prev Med. 2003;25:45–9. doi: 10.1016/s0749-3797(03)00093-x. [DOI] [PubMed] [Google Scholar]


