Abstract
We examined the histological distribution of the lesions and the viral antigen associated with the virus and virus RNA in multisystemic organs in the early stages of foot-and-mouth disease virus (FMDV) O/JPN/2010 infection in pigs. Characteristic lesions commonly observed in pigs with FMD arise following inoculation with 106 tissue culture infectious dose (TCID)50/ml of FMDV O/JPN/2010 in pigs at 3 days post inoculation (dpi) by a natural infectious route. However, none of the six pigs inoculated with 103 TCID50/ml of FMDV O/JPN/2010 showed any evidence of infection up to 6 dpi. Immunohistochemical detection for the FMDV antigen and terminal deoxynucleotidyl transferase-mediated dUTP-nick end labeling (TUNEL) showed that FMDV predominantly infected prickle cells in the stratum spinosum in the tongue, coronet and bulb of the heel, and caused these infected cells to undergo cell death by apoptosis. However, there was no evidence that FMDV O/JPN/2010 infected epithelial/epidermal basal cells in the basal layer. Epithelial lesions with viral antigen in the tongue were distributed in the dorsal surface but not in the papillae, corpus linguae or inferior surface of the tongue. Non-suppurative myocarditis and epithelial lesions in the esophagus with FMDV antigen were observed in all three pigs examined at 3 dpi.
Keywords: foot-and-mouth disease virus, histopathology, immunohistochemistry, pig
Foot-and-mouth disease (FMD) is a highly contagious and economically devastating transboundary viral disease that affects cloven-hooved animals such as cattle, pigs, deer, goats, sheep and buffalo [1]. FMD is caused by the foot-and-mouth disease virus (FMDV) of the Aphthovirus genus and Picornaviridae family, and has seven distinct serotypes [1]. The FMDV serotypes O, A and Asia1 are currently prevalent in Southeast Asia (SEA) [16]. FMDV serotype O is the most prevalent and is divided into several topotypes according to molecular analysis of the structural protein VP1 [16]. In 2010, FMD epidemics caused by the FMDV serotype O SEA topotype devastated the livestock industry in many Asian countries [16], including Japan [23]. The FMDV serotype O SEA topotype virus has continued to circulate throughout many Asian countries since 2010 [27]. Previously we reported an experimental study using serotype O SEA topotype FMDV isolated in the 2010 epidemic in Japan from cows, goats [25] and pigs [12, 14] by a virologic approach. However, there are currently a limited number of reports on the pathology and viral distribution in host animals infected with FMDVs of SEA topotype.
In several forms of FMD, infected pigs exhale more virus particles than infected cattle [3]. Therefore, a FMD outbreak may spread rapidly if it occurs at a pig farm in an FMD-free country where vaccination is not practiced [10, 17]. In the FMD outbreak in Japan in 2010, confirmed cases increased markedly after a FMD outbreak occurred at a pig farm [23]. Early detection and notification are the most important factors for rapid and effective control of FMD in a FMD-free county, such as Japan. Many studies have examined the early pathogenesis of FMDV infection by experimentally infecting pigs with FMDV [2, 11, 19, 22, 32, 33]. FMDV primarily infects and replicates in the oropharyngeal tonsils in pigs [4, 30, 32, 33]. Viremia regularly follows FMDV infections, resulting in characteristic vesicular lesions in target organs including the foot and tongue [4, 30]. However, it is still unclear how these characteristic vesicular lesions develop in pigs. Immunohistochemical detection of viral antigens in formalin-fixed paraffin-embedded tissue enables the detection and localization of viral antigens within cells and tissues, and is a simple and rapid method for studying viral organ tropism and pathogenesis. However, few reports have examined the immunohistochemical distribution of FMDV antigens across the entire body of FMDV-infected pigs [19]. We previously examined experimental infection with FMDV O/JPN/2010 in pigs using intradermal inoculation and contact exposure for 11 days [12], and intraoral and intranasal inoculation for 14 days [14]. However, detailed histological examination and immunohistochemical distribution of the viral antigen associated with the presence of the virus and virus RNA in multisystemic organs in the early stages of FMDV O/JPN/2010 infection have not been examined.
Here, we describe the developmental characteristics of FMD lesions in pigs infected with FMDV O/JPN/2010 by intraoral inoculation. We examined the pathology and distribution of FMDV across the entire body of pigs in the early stages of infection using FMDV serotype O SEA topotype isolated from the 2010 epidemic in Japan.
MATERIALS AND METHODS
Virus
The isolation and passage histories of the FMDV O/JPN/2010 isolate (O/JPN/2010-1/14c) were described in our previous study [12].
Experimental design
Pigs aged 24 days old were anesthetized with 2.0 mg of xylazine (Celactal, Bayer Yakuhin, Osaka, Japan) and 20 mg of pentobarbital (Somnopentyl, Kyoritsu Seiyaku, Tokyo, Japan) per kg before inoculation with FMDV.
Six pigs (numbers 2–7) were intraorally inoculated with a syringe containing 1 ml of a 106 tissue culture infectious dose (TCID)50/ml of the FMDV O/JPN/2010 isolate while another six pigs (numbers 8–13) were intraorally inoculated with 1 ml of 103 TCID50/ml of the same virus. One pig (number 1) was necropsied before inoculation as a non-infected control.
Pigs 2, 5, 6, 8 and 9 were examined clinically at 1 day post inoculation (dpi). Pigs 3, 4, 7, 10 and 11 were examined clinically at 3 dpi. Pigs 12 and 13 were examined clinically at 6 dpi. Clinical samples of sera, saliva and nasal swabs were collected from the pigs daily. Detailed methods of the collection and preparation of clinical samples used in this study were described previously [14]. Rectal temperatures were taken daily. After clinical assessment, the pigs were euthanized by injection of sodium pentobarbital and subjected to necropsy examination. These methods were conducted in animal rooms and laboratories in a high-containment facility at our institute in Kodaira, Tokyo. The experiments were approved by the Animal Ethics Committee of the National Institute of Animal Health (NIAH), Japan (authorization number: 13-084).
Histopathology and immunohistochemistry
Tissue samples for microscopy examination were collected from the lip; tongue; soft palate; tonsil of the soft palate; lingual tonsil; oropharynx mucosa including the paraepiglottic tonsil; nasopharynx mucosa including the pharyngeal tonsil; larynx; trachea; lung; esophagus; stomach; ileum; rectum; liver; spleen; kidney; heart; pancreas; gallbladder; bladder; thyroid gland; thymus; mandibular lymph node; parotid lymph node; retropharyngeal lymph node; cervical lymph node; inguinal lymph node; popliteal lymph node; mesenteric lymph node; mandibular gland; parotid gland; diaphragm; brachial skeletal muscle; femoral skeletal muscle; and skin of the snout, shoulder, leg, coronet, bulb of the heel and accessory digit of each pig. Tissues were fixed in 10% neutral phosphate buffered formalin, processed according to routine procedures and embedded in paraffin wax. Sections were stained with hematoxylin and eosin (H&E).
For immunohistochemistry (IHC), dewaxed sections were processed using the Universal Immuno-enzyme Polymer method with a HISTFINE simple stain Max PO (M) kit (Nichirei, Tokyo, Japan) according to the manufacturer’s instructions. Sections were labeled using a monoclonal antibody specific for FMDV serotype O (72C1, diluted 1 in 8, NIAH, Japan) [21] and counterstained with hematoxylin.
Cryo-sections of the tongue and skin of the coronet were processed to confirm the specificity of the 72C1 monoclonal antibody using immunofluorescence (IF). Samples of the tongue and skin of the coronet were frozen in liquid nitrogen. The FMDV antigen in cryo-sections was detected by IF using a primary antibody of the 72C1 and an anti-mouse immunoglobulin (Ig) G (H + L) fragment (Alexa Fluor® 488 conjugate, diluted 1 in 1,000; Cell Signaling Technology Japan K.K., Tokyo, Japan). Sections were mounted with ProLong® Gold Antifade Reagent with DAPI (Cell Signaling Technology Japan K.K.).
Detection of cells with a DNA strand break associated with apoptosis
To identify cells with a DNA strand break (characteristic of apoptosis), paraffin wax-embedded sections of the tongue and skin of the coronet, bulb and snout from all pigs were subjected to terminal deoxynucleotidyl transferase-mediated dUTP-nick end labeling (TUNEL) [9] using an Apoptag Kit (Chemicon International, Tokyo, Japan) according to the manufacturer’s instructions.
Cell culture, virus isolation and titration
Clinical samples and macerated tissue samples were subjected to virus isolation and titration. Tissue samples for virus isolation and titration were collected from the tongue, soft palate, tonsil of the soft palate, oropharynx mucosa including the paraepiglottic tonsil, nasopharynx mucosa including the pharyngeal tonsil, larynx, trachea, lung, esophagus, stomach, ileum, rectum, spleen, heart, thymus, mandibular lymph node, parotid lymph node, retropharyngeal lymph node, cervical lymph node, mandibular gland, parotid gland, skin of the snout and skin of the coronet of the right forefoot of pigs.
The LFBK-αvβ6 cells were used for virus isolation and titration as described previously [13]. Virus isolation was performed according to the OIE Manual as described previously [13, 14].
RNA extraction and RT-PCR
The same samples collected for virus isolation and titration were subjected to RNA extraction and reverse transcriptase-polymerase chain reaction (RT-PCR). Viral RNAs were extracted from samples using a High Pure Viral RNA kit (Roche Diagnostics, Basel, Switzerland) according to the manufacturer’s instructions and as described in our previous study [13, 14].
FMDV-specific genes were detected from the extracted RNAs by RT-PCR using FM8 and FM9 primers [28] as described in our previous study [13, 14].
RESULTS
Clinical signs and gross lesions
In pigs inoculated with 106 TCID50/ml of FMDV O/JPN/2010, vesicular lesions were clinically observed on the feet of one in six pigs (pig No. 2) at 1 dpi, and on the feet (Figs. 1a and 1b) or tongue (Fig. 1c) of three pigs (pig Nos. 3, 4 and 7) at 3 dpi (Table 1). At first, lesions on the feet appeared as blanched patches in the interdigital space between the hooves. These blanched patches spread to the coronary band, bulb of the heel and accessory digits and then developed into vesicles on one or more feet (Fig. 1a, 1b and 1d). Ruptured vesicles were observed on the feet from 2 dpi (Fig. 1c and 1d). At necropsy, the cut surface of the coronet cross section revealed that the vesicles had formed under the blanched patches at 1 dpi despite not being visible clinically (Fig. 1e). Moreover, the vesicles on the bulb of the heel and coronet had extended and merged in some feet at 3 dpi (Fig. 1f). On the tongue, blanched vesicles were observed on the dorsal surface along the midline in two pigs (pig Nos. 4 and 7) at 3 dpi (Fig. 1g). Ruptured vesicles were located on the lingual aspect of the lower lip along the midline in two pigs (pig Nos. 4 and 7) at 3 dpi (Fig. 1h). In the oral cavity, there were no gross lesions on the surface of the corpus linguae, inferior surface of the tongue or sublingual floor of the oral cavity, hard palate, soft palate, tonsil and laryngopharynx at necropsy. There were no vesicular lesions on the snout, upper lips or teats in any examined pigs. Salivation was not observed in any pigs. Although the rectal temperature as measured by an electronic thermometer was normal for all pigs, we felt fervescence when we touched the feet and body of all pigs during sampling at 1 dpi. Mild pneumonia and severe bronchopneumonia with multifocal abscesses appeared in pig No. 2 and No. 4 at necropsy, respectively. There were no gross lesions in the other organs.
Fig. 1.
Gross lesions in pigs inoculated with 106 tissue culture infectious dose (TCID)50/ml of foot-and-mouth disease virus (FMDV) O/JPN/2010. (a and b) Vesicles (arrows) in the interdigital space (a) and on the bulb of the heel (b) of the left forefoot of pig No. 3 at 3 dpi. (c) Ruptured vesicle in the interdigital space of the left forefoot of pig No. 4 at 3 dpi. (d) White vesicles (black arrows) and a ruptured vesicle (white arrow) on the left forefoot of pig No. 4 at 3 dpi. (e and f) Cut surface of a cross section of the coronet of pig No. 2 at 1 dpi (e) and pig No. 4 at 3 dpi (f). At 1 dpi, vesicles in the coronet are observed at the side or ridge (arrows). At 3 dpi, vesicular lesions had developed all around the coronet. (g) A blanched vesicle on the dorsal surface of the tongue along the midline in pig No. 7 at 3 dpi. (h) A ruptured vesicle on the lingual aspect of the lower lip along the midline in pig No. 4 at 3 dpi.
Table 1. Gross lesions in pigs inoculated with 106 tissue culture infectious dose (TCID)50/ml of foot-and-mouth disease virus O/JPN/2010.
| Pig No. | Site | 0 dpi | 1 dpi | 2 dpi | 3 dpi |
|---|---|---|---|---|---|
| 2 | Tongue | − | − | ||
| Lip | − | − | |||
| Right forefoot | − | + | |||
| Left forefoot | − | − | |||
| Right hindfoot | − | + | |||
| Left hindfoot | − | + | |||
| 5 | Tongue | − | − | ||
| Lip | − | − | |||
| Right forefoot | − | − | |||
| Left forefoot | − | − | |||
| Right hindfoot | − | − | |||
| Left hindfoot | − | − | |||
| 6 | Tongue | − | − | ||
| Lip | − | − | |||
| Right forefoot | − | − | |||
| Left forefoot | − | − | |||
| Right hindfoot | − | − | |||
| Left hindfoot | − | − | |||
| 3 | Tongue | − | − | − | − |
| Lip | − | − | − | − | |
| Right forefoot | − | − | + | + | |
| Left forefoot | − | − | − | + | |
| Right hindfoot | − | − | − | + | |
| Left hindfoot | − | − | − | − | |
| 4 | Tongue | − | − | − | + |
| Lip | − | − | − | + | |
| Right forefoot | − | − | + | + | |
| Left forefoot | − | − | + | + | |
| Right hindfoot | − | − | − | + | |
| Left hindfoot | − | − | − | + | |
| 7 | Tongue | − | − | − | + |
| Lip | − | − | − | + | |
| Right forefoot | − | − | − | − | |
| Left forefoot | − | − | − | − | |
| Right hindfoot | − | − | − | − | |
| Left hindfoot | − | − | − | − | |
dpi: day(s) post inoculation; +: vesicular or erosive/ulcerative lesion positive. The site showing lesion positive are colored yellow.
No clinical signs or gross lesions associated with FMDV infection were observed in any of the six pigs inoculated with 103 TCID50/ml of FMDV O/JPN/2010.
Histological and immunohistochemical examination in the tongue of pigs inoculated with 106 TCID50/ml of FMDV O/JPN/2010
At 1 dpi, no prominent histological lesions were observed in the tongue of the three examined pigs. However, the viral antigen was immunohistochemically detected in the papilla of the connective tissue and prickle cells surrounding the papilla of the connective tissue in the upper layer of the stratum spinosum in the dorsal surface along midline (Fig. 2a). In serial sections stained with TUNEL, TUNEL-positive reaction was prominent in the lamellar in the upper to middle layer of the stratum spinosum along the midline of the dorsal epithelium, in the absence of other histological abnormalities. At 3 dpi, a histological abnormality was observed and comprised of necrosis of individual prickle cells in the middle to bottom layer of the stratum spinosum in the dorsal epithelium along the midline, and was associated with the presence of the viral antigen (Fig. 2b and 2c). The individually necrotic cells seemed to fuse together to form small vesicles (Fig. 2b) and developed into large vesicles in the stratum spinosum. The distribution of the viral antigen corresponded closely to the distribution of epithelial lesions in the tongue (Fig. 2c and 2f). Papilla of the connective tissue were also cavitated, and were filled with necrotic cell debris and vesicular fluid, and coalesced to become continuous with the leading vesicle (Fig. 2d). The severely affected vesicular lesion in the epithelium of the tongue developed a spongiform appearance (Fig. 2e and 2f). Viral antigen was not detected in the stratum corneum.
Fig. 2.
Histology and immunohistochemical detection of the FMDV antigen in vesicular lesions on the tongue of pigs inoculated with 106 TCID50/ml of FMDV O/JPN/2010. (a) Viral antigen in the periphery of the papilla of connective tissue in pig No. 2 at 1 dpi. Immunohistochemistry (IHC), scale bar=100 µm. (b and c) Individual necrotic prickle cells and small vesicles in the middle to bottom layer of the stratum spinosum with FMDV antigen in pig No. 4 at 3 dpi. b: hematoxylin and eosin (H&E), scale bar=100 µm; c: IHC, scale bar=100 µm. (d) Papilla of connective tissue are cavitated and filled with necrotic cell debris in pig No. 4 at 3 dpi. H&E, scale bar=100 µm. (e and f) Vesicular lesion showing a spongiform appearance with FMDV antigen in pig No. 4 at 3 dpi. e: H&E, scale bar=200 µm; f: IHC, scale bar=100 µm.
Ulcers or erosions, rather than vesicular lesions observed in the tongue, were the main types of lesions observed in infected pigs (Fig. 3a and 3b). Ulcerative lesions appeared as severe necrosis and acantholysis of prickle cells penetrating the stratum spinosum. Epithelial cells in the basal layer were also affected, and the structure of the basal layer was destroyed in ulcerative lesions (Fig. 3c). In serial sections stained with IHC, the FMD viral antigen was prominent in the necrotic lesion penetrating into the stratum spinosum. However, the viral antigen was not detected in the epithelial cells of the basal layer (Fig. 3d). In serial sections stained with TUNEL, affected epithelial cells in the stratum spinosum were prominently labeled. In contrast, epithelial cells of the basal layer were negative for TUNEL (Fig. 3e). Epithelial lesions were only observed in the dorsal epithelium of the tongue. The papillae of the tongue however were intact despite the surrounding tissue being severely affected (Fig. 3f). The viral antigen was not detected in the papillae of the tongue despite prominent label in the surrounding epithelial cells of the stratum spinosum (Fig. 3g). The viral antigen was detected in the same areas of the tongue in cryo-sections and paraffin-embedded sections. In severe ulcerative lesions in the tongue, epithelial lesions extended into the muscle layer (Fig. 3a and 3b). Myositis composed of lymphocyte infiltration and necrosis of the muscle fibers, and prominent viral antigen labeling were observed in these lesions (Fig. 3h and 3i).
Fig. 3.
Histology, immunohistochemical detection of the FMDV antigen and terminal deoxynucleotidyl transferase-mediated dUTP-nick end labeling (TUNEL) reaction in ulcerative lesions on the tongue of pig No. 4 at 3 dpi following inoculation with 106 TCID50/ml of FMDV O/JPN/2010. (a and b) Ulceration, epithelial necrosis and myositis with FMDV antigen. a: H&E, scale bar=400 µm; b: IHC, scale bar=400 µm. (c, d and e) Epithelial necrosis in the stratum spinosum with FMDV antigen and TUNEL-positive reaction. No FMDV antigen or TUNEL-positive reaction is observed in the affected basal layer. c: H&E, scale bar=100 µm; d: IHC, scale bar=100 µm; e: TUNEL, scale bar=100 µm. (f and g) No lesion or FMDV antigen is observed in the papilla of the tongue. Surrounding epithelium is severely affected, with prominent FMDV antigen staining. f: H&E, scale bar=200 µm; g: IHC, scale bar=200 µm. (h and i) Myositis composed of lymphocyte proliferation and necrosis of muscle fibers with FMDV antigen. h: H&E, scale bar=100 µm; i: IHC, scale bar=100 µm.
In the feet of pigs inoculated with 106 TCID50/ml of FMDV O/JPN/2010
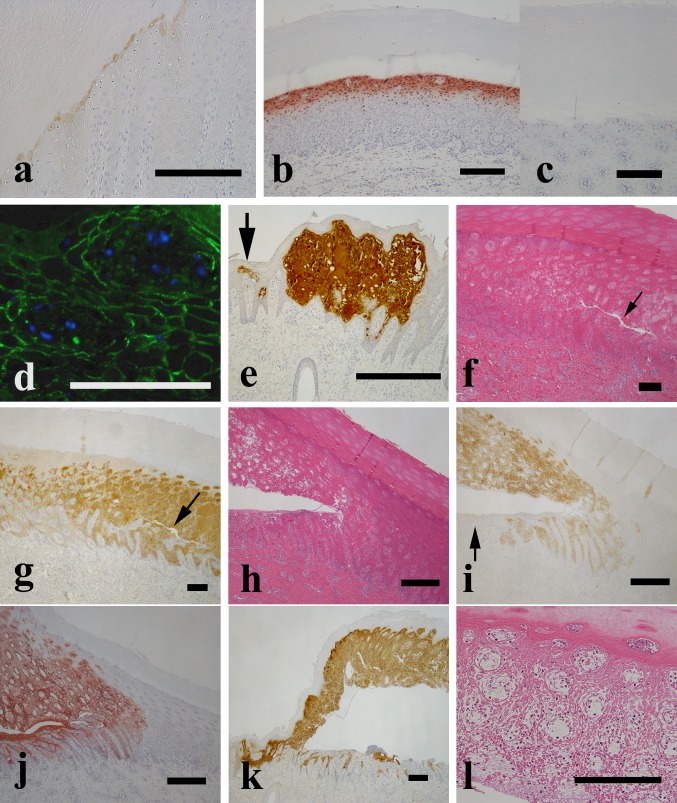
Viral antigen labeling was faint in histologically-normal areas of the epidermis, especially in the upper layer of the stratum spinosum lamellar (Fig. 4a). TUNEL-positive label was prominent in the lamellar of the upper layer of the stratum spinosum, which showed no other histological abnormalities (Fig. 4b), although some TUNEL positive reaction was observed at the upper layer of the stratum spinosum in the non-infected control pig (Fig. 4c). In cryo-sections, the viral antigen was first detected in prickle cells surrounding the dermal papilla in the upper layer of the stratum spinosum in the skin of the coronet at 1 dpi (Fig. 4d). The viral antigen was first detected in the cell membrane of the infected cells and seemed to expand into adjacent cells in a contagious manner (Fig. 4d). A histological abnormality comprising multifocal acantholytic degeneration of prickle cells surrounded by the dermal papillae and small regions of multifocal necrosis in the upper to middle layer of the stratum spinosum of the coronet were observed in the feet of some pigs at 1 dpi (Fig. 4e). The focal necrosis may have developed rapidly into massive necrosis as viral antigen levels increased in the stratum spinosum (Fig. 4e). A cleft subsequently appeared in the center of the necrotic lesion (Fig. 4f and 4g). Epidermal necrotic lesions with a cleft were TUNEL-positive and viral antigen-positive and further developed into vesicles following separation of the epidermis from the underlying tissue (Fig. 4h, 4i and 4j). Epidermal lesions developed further into large vesicles by the filling of the cavity with vesicular fluid (Fig. 4k). Acantholytic and necrotic cellular debris and fibrin were detected in the vesicular cavities. The viral antigen was prominently observed in the vesicular epidermis of the foot (Fig. 4k). Prickle cells in the vesicular epidermis were acantholytic, necrotic and loosely connected to the stratum corneum (Fig. 4l). Ulcers or erosions formed after vesicle rupture. In the ulcers or erosions, neutrophil and lymphocyte infiltration were observed, but hemorrhage was less prominent and no FMDV antigen was observed in ulcerative lesions.
Fig. 4.
Histology, immunohistochemical detection of the FMDV antigen and TUNEL reaction in vesicular lesions on the foot of pigs inoculated with 106 TCID50/ml of FMDV O/JPN/2010. (a) Viral antigen in histologically normal areas of the upper layer of the stratum spinosum in pig No. 5 at 1 dpi. IHC, scale bar=200 µm. (b) TUNEL-positive reaction was prominent in the lamellar in the upper layer of the stratum spinosum without other histological abnormalities in the bulb of the heel of pig No. 5 at 1 dpi. TUNEL, scale bar=200 µm. (c) Some TUNEL-positive reaction in the upper layer of the stratum spinosum in a non-infected control pig (pig No. 1). TUNEL, scale bar=200 µm. (d) FMDV antigen in a cryo-section of the skin of the coronet of pig No. 2 at 1 dpi. Viral antigen is detected in the cell membrane of infected prickle cells surrounding the dermal papilla in the upper layer of the stratum spinosum. Immunofluorescence (IF), scale bar=100 µm. (e) Small region of focal necrosis (arrow) and massive necrosis with FMDV antigen in the stratum spinosum of the skin of the coronet of pig No. 4 at 3 dpi. IHC, scale bar=400 µm. (f and g) Severe necrotic lesion with prominent FMDV antigen staining in the stratum spinosum of the skin of the bulb of the heel of pig No. 4 at 3 dpi. Note the cleft in the center of the necrotic lesion (arrow). f: H&E, scale bar=200 µm; g: IHC, scale bar=200 µm. (h, i and j) The epidermal necrotic lesion with the cleft is TUNEL-positive and viral antigen-positive and is continuing to develop into a vesicle by the separation of the epidermis from the underlying tissue in the skin of the bulb of the heel of pig No. 2 at 1 dpi. Note the lower FMDV antigen staining in the underlying tissue (arrow). h: H&E, scale bar=400 µm; i: IHC, scale bar=400 µm; j: TUNEL, scale bar=400 µm. (k) Viral antigen staining is prominent in the vesicular epidermis but lower in the underlying tissue of the skin around the accessory digit of pig No. 4 at 3 dpi. IHC, scale bar=400 µm. (l) Prickle cells in the vesicular epidermis were acantholytic and necrotic and loosely connected to the stratum corneum of the skin of the coronet of pig No. 2 at 1 dpi. H&E, scale bar=200 µm.
In the basal area, viral antigen and TUNEL-positive label were detected in the suprabasal prickle cells of the stratum spinosum, but not in basal cells (Fig. 4h). The epithelial cells in the stratum granulosm of the skin of the coronet and heel showed ballooning degeneration. Viral antigen was not detected in the stratum corneum (Fig. 4g). Similar vesicular or ulcerative lesions were observed in the skin of the bulb of the heel, and interdigital space of the hooves and accessory digits. In the supracoronary hair follicles, we observed acantholysis of the follicular epithelium and mural epithelium associated with the presence of the viral antigen. However, no histological lesions associated with the viral antigen were observed in haired skin from the leg. Lesions and the presence of the viral antigen and TUNEL-positive reaction in the skin of the foot were prominent and more severe than those observed in the tongue.
In the other organs of pigs inoculated with 106 TCID50/ml of FMDV O/JPN/2010
The distribution of histological lesions and immunohistochemical viral antigen associated with the presence of the virus and virus RNA in multisystemic organs in the early stages of infection with FMDV O/JPN/2010 are summarized in Table 3.
Table 3. The results of virus isolation, RT-PCR analysis, immunohistochemistry (IHC) and histology from tissue samples (virus isolation/RT-PCR/IHC/histological lesion).
| Pig No. | 2 | 5 | 6 | 3 | 4 | 7 |
|---|---|---|---|---|---|---|
| dpi | 1 | 1 | 1 | 3 | 3 | 3 |
| Tongue | −/−/+/− | −/−/+/− | −/−/+/− | −/+/+/+ | 3.1/+/+/+ | 5.8/+/+/+ |
| Tonsil of the soft palate | −/+/+/− | −/−/+/− | 4.3/+/+/− | −/+/+/− | 6.3/+/+/− | 3.3/+/+/− |
| Soft palate | 2.8/+/−/− | −/+/−/− | −/−/−/− | −/+/−/− | 4.3/+/−/− | 3.3/+/−/− |
| Oropharynx | 2.6/+/−/− | 2.6/+/−/− | −/−/−/− | 2.6/+/−/− | 4.3/+/+/− | 2.6/+/+/− |
| Nasopharynx | −/−/−/− | −/+/−/− | −/−/−/− | −/−/−/− | 4.8/+/−/− | 2.8/+/−/− |
| Larynx | 3.1/+/−/− | −/+/−/− | −/−/−/− | −/−/−/− | 5.3/+/−/− | 2.8/+/−/− |
| Trachea | −/−/−/− | −/−/−/− | −/−/−/− | −/−/−/− | 4.1/+/−/− | 2.6/+/−/− |
| Lung | −/−/−/− | −/+/−/− | −/−/−/− | −/−/−/− | 3.8/+/−/− | 2.6/+/−/− |
| Esophagus | −/−/−/− | −/+/−/− | −/−/−/− | −/+/+/+ | 6.6/+/+/+ | −/+/+/+ |
| Stomach | −/−/−/− | −/−/−/− | −/−/−/− | −/+/−/− | 3.3/+/−/− | −/−/−/− |
| Ileum | −/−/−/− | −/−/−/− | −/−/−/− | −/−/−/− | 3.3/+/−/− | −/−/−/− |
| Rectum | −/−/−/− | −/−/−/− | −/−/−/− | −/−/−/− | 3.3/+/−/− | −/−/−/− |
| Heart | −/−/+/− | −/−/+/− | −/−/−/− | −/−/+/+ | 3.8/+/+/+ | −/+/+/+ |
| Spleen | −/−/−/− | −/−/−/− | −/−/−/− | −/−/−/− | 5.8/+/−/− | −/+/−/− |
| Thymus | −/−/−/− | −/−/−/− | −/−/−/− | −/−/−/− | 3.8/+/−/− | 2.6/+/−/− |
| Mandibular LN | −/−/−/− | −/−/−/− | −/−/−/− | −/−/−/− | 7.1/+/−/− | −/+/−/− |
| Parotid LN | −/−/−/− | −/−/−/− | −/−/−/− | −/−/−/− | 6.8/+/−/− | −/+/−/− |
| Retropharyngeal LN | −/−/−/− | −/−/−/− | −/−/−/− | −/−/−/− | 5.8/+/−/− | −/+/−/− |
| Cervical LN | −/−/−/− | −/−/−/− | −/−/−/− | −/−/−/− | 8.3/+/−/− | −/+/−/− |
| Mandibular gland | −/−/−/− | −/−/−/− | −/−/−/− | −/−/−/− | 3.8/+/−/− | 3.3/+/−/− |
| Parotid gland | −/−/−/− | −/−/−/− | −/−/−/− | −/−/−/− | 3.1/+/−/− | −/−/−/− |
| Skin of the snout | −/−/−/− | −/−/−/− | −/−/−/− | −/+/+/+ | 7.3/+/+/+ | −/−/+/+ |
| Skin of the coronet | −/−/+/+ | −/−/+/+ | −/−/+/+ | −/+/+/+ | 9.3/+/+/+ | 5.3/+/+/+ |
dpi: day(s) post inoculation; LN: lymph node; 3.1/+/+/+: 3.1 log10TCID50/ml/PCR positive/IHC positive/histological lesion positive. Samples showed virus isolation positive and RT-PCR positive are colored orange. Samples showed virus isolation negative but RT-PCR and IHC positive are colored yellow. Samples showed virus isolation negative but RT-PCR or IHC positive are colored light orange.
No histological lesions were observed in the tonsil of the soft palate, paraepiglottic tonsil, lingual tonsil or pharyngeal tonsil, although the viral antigen was detected in the tonsillar crypt epithelium in the tonsil of the soft palate in all pigs from 1 dpi (Fig. 5a) and in the paraepiglottic tonsil and lingual tonsil in two of three pigs (pig Nos. 4 and 7) at 3 dpi. No histological lesions or viral antigen were observed in the mucosal epithelium of the oropharynx, nasopharynx, soft palate or larynx. In the esophagus, multifocal necrosis with viral antigen and ballooning degeneration of the epithelial cells were observed in the upper layer of the epithelium in three pigs at 3 dpi (Fig. 5b). In these lesions, the basal layer was intact and no viral antigen was detected in the basal layer of the epithelium. No gross lesions were observed in the hearts of any pigs, but non-suppurative myocarditis composed of lymphocyte infiltration and necrosis of cardiac muscle fibers with prominent viral antigen was histologically detected in all three pigs at 3 dpi (Fig. 5d and 5e). At 1 dpi, multifocal distribution of the viral antigen was detected in degenerated muscle fibers in two of three pigs (pig Nos. 2 and 5) (Fig. 5c). Ballooning degeneration with FMDV antigen was observed in the stratum spinosum of the skin of the snout of all three pigs at 3 dpi. The lesion in the snout was positive for TUNEL (Fig. 5f). Mild bacterial suppurative pneumonia and severe bacterial suppurative bronchopneumonia were observed in the lungs of pigs No. 2 and No. 4, respectively. The Lung lesion was not associated with FMD virus infection and FMD virus antigen was not detected in those lung lesions. No histological lesions or viral antigen were observed in the other organs including the pancreas and thyroid gland.
Fig. 5.
Histology, immunohistochemical detection of the FMDV antigen and TUNEL reaction in the other organs of pigs inoculated with 106 TCID50/ml of FMDV O/JPN/2010. (a) FMDV antigen in the tonsillar crypt epithelium in the tonsil of the soft palate of pig No. 2 at 1 dpi. IHC, scale bar=200 µm. (b) FMDV antigen in the upper layer of the epithelium in the esophagus of pig No. 4 at 3 dpi. IHC, scale bar=100 µm. (c) FMDV antigen in degenerated cardiac muscle fibers in the heart of pig No. 5 at 1 dpi. IHC, scale bar=200 µm. (d and e) Non-suppurative myocarditis composed of lymphocyte infiltration and necrosis of cardiac muscle fibers with prominent viral antigen staining in the heart of pig No. 4 at 3 dpi. d: H&E, scale bar=200 µm; e: IHC, scale bar=100 µm. (f) TUNEL-positive reaction in epidermal cells showing ballooning degeneration in the skin of the snout of pig No. 7 at 3 dpi. TUNEL, scale bar=100 µm.
Pigs inoculated with 103 TCID50/ml of FMDV O/JPN/2010
No histological lesions associated with FMD virus infection or viral antigen were detected in any organs of any pigs inoculated with 103 TCID50/ml of FMDV O/JPN/2010.
Virus isolation and RT-PCR
The results of virus isolation and RT-PCR analysis of clinical samples from pigs inoculated with 106 TCID50/ml of FMDV O/JPN/2010 are summarized in Table 2. At 1 dpi, virus shedding was detected in saliva and nasal swab samples from four of six pigs inoculated with 106 TCID50/ml of FMDV O/JPN/2010 by RT-PCR analysis. At 3 dpi, the virus was isolated from saliva and nasal swab samples from all three of the remained pigs inoculated with 106 TCID50/ml of FMDV O/JPN/2010.
Table 2. Virus shedding in saliva, nasal swab and serum samples from pigs inoculated with 106 tissue culture infectious dose (TCID)50/ml of foot-and-mouth disease virus O/JPN/2010 (virus isolation/RT-PCR).
| Pig No. | Sample | 0 dpi | 1 dpi | 2 dpi | 3 dpi |
|---|---|---|---|---|---|
| 2 | Saliva | −/− | −/+ | ||
| Nasal swab | −/− | −/− | |||
| Serum | −/− | −/+ | |||
| 5 | Saliva | −/− | 2.6/+ | ||
| Nasal swab | −/− | −/+ | |||
| Serum | −/− | −/− | |||
| 6 | Saliva | −/− | −/− | ||
| Nasal swab | −/− | −/+ | |||
| Serum | −/− | −/− | |||
| 3 | Saliva | −/− | −/− | −/+ | 3.8/+ |
| Nasal swab | −/− | −/− | −/+ | 3.3/+ | |
| Serum | −/− | −/− | −/− | −/− | |
| 4 | Saliva | −/− | −/− | 5.1/+ | 5.8/+ |
| Nasal swab | −/− | −/− | 3.3/+ | 4.6/+ | |
| Serum | −/− | −/− | 5.1/+ | 5.8/+ | |
| 7 | Saliva | −/− | −/− | −/− | 2.6/+ |
| Nasal swab | −/− | −/+ | −/− | 2.6/+ | |
| Serum | −/− | −/− | −/− | −/− | |
dpi: day(s) post inoculation; 2.6/+: 2.6 log10TCID50/ml/PCR positive; Samples showed virus isolation positive and RT-PCR positive are colored orange. −/+: virus isolation negative/PCR positive; Samples showed virus isolation negative but RT-PCR positive are colored yellow. −/−: virus isolation negative/PCR negative.
The results of virus isolation and RT-PCR analysis of tissue samples from pigs inoculated with 106 TCID50/ml of FMDV O/JPN/2010 are summarized in Table 3. At 1 dpi, the tonsil of the soft palate, soft palate, oropharynx and larynx were positive for the virus in two of three pigs inoculated with 106 TCID50/ml of FMDV O/JPN/2010 by RT-PCR. At 3 dpi, the virus was isolated from the oropharynx of all three pigs, and from the tongue, tonsil of the soft palate, soft palate, nasopharynx, larynx, trachea, lung, thymus, mandibular gland and skin of the coronet of two of the three pigs inoculated with 106 TCID50/ml of FMDV O/JPN/2010.
The results of our RT-PCR analysis were negative for all clinical and tissue samples collected from pigs inoculated with 103 TCID50/ml of FMDV O/JPN/2010. Therefore, we found no evidence of infection in pigs inoculated with 103 TCID50/ml of FMDV O/JPN/2010.
DISCUSSION
We confirmed that characteristic lesions commonly observed in pigs with FMD [3, 34] arise following inoculation with 106 TCID50/ml of FMDV O/JPN/2010 by a natural infectious route. This FMDV showed characteristic cellular tropism, infecting prickle cells in the stratum spinosum but not basal cells or epithelial cells of the papillae of the tongue. Multifocal necrosis associated with the viral antigen was observed in the upper layer of the epithelium in the esophagus in three pigs at 3 dpi. This is the first report to investigate the histological development of lesions with FMD viral antigen in the esophagus of pigs. In contrast, none of the six pigs inoculated with 103 TCID50/ml of FMDV O/JPN/2010 showed any evidence of infection up until at least 6 dpi. Our previous study in which pigs were inoculated orally with 103 TCID50/ml of FMDV O/JPN/2010 found that at least one pig was infected [14]. Additionally, no pigs inoculated intranasally with 103 TCID50/ml of FMDV O/JPN/2010 showed evidence of infection or seroconversion [14]. Therefore, an inoculation dose of 103 TCID50/ml may be close to the minimum infectious dose of FMDV O/JPN/2010 for pigs by the natural infection route.
In the tongue, viral antigen and TUNEL-positive labeling were observed in necrotic prickle cells in the stratum spinosum. Our findings confirm that the FMDV may preferentially infect prickle cells in the stratum spinosum in the tongue, which leads to death of the infected cells by apoptosis. In severe ulcerative lesions in the tongue, epithelial cells of the basal layer were also affected and destroyed. However, neither the FMD viral antigen by IHC nor TUNEL-positive label was detected in the basal layer of the epithelium in the tongue. These results indicate that, rather than arising as a direct result of the virus, the cellular injury in the basal layer might instead occur indirectly from the lesions in surrounding tissue. Epithelial basal cells have been proposed to be an early replication site for FMDV in vivo because FMDV-positive and -negative strand RNAs are prominent in the basal layer of the epithelium [11]. However, we found no evidence that FMDV O/JPN/2010 infects epithelial basal cells in the basal layer in pigs. This discrepancy may be associated with differences in the FMDV strains examined or the experimental designs between the studies.
Interestingly, the papillae of the tongue were histologically intact and were negative for the FMD viral antigen by IHC, while the surrounding epithelium was severely affected and showed prominent FMD antigen staining. There is currently no detailed study describing the lack of infection of epithelial cells in the papillae of the tongue by the FMDV. In addition, we found no histological lesions associated with the presence of the viral antigen and TUNEL-positive label in the epithelium of the corpus linguae and inferior surface of the tongue. Histological lesions associated with the presence of the viral antigen and TUNEL-positive labeling were only observed in the dorsal surface of the tongue. The cellular tropism of FMD infection is not well reported. It is important that FMD be differentiable from other viral vesicular diseases and diseases producing erosive/ulcerative lesions in the oral cavity. Gross lesions alone cannot differentiate other viral vesicular diseases such as vesicular stomatitis, vesicular exanthema and swine vesicular disease [34]. In contrast, the distribution of lesions and cellular tropism in the tongue observed in this study may be important for the differential diagnosis of FMD from other diseases producing erosive/ulcerative lesions in the oral cavity in pigs.
Viral antigen was detected by IHC in the peripheral areas of the papilla of connective tissue and dermal papillae, and prickle cells in the upper layer of the stratum spinosum in the tongue, esophagus and skin of the coronet and heel. No direct virus invasion via surface epithelial cells was observed. These findings agree with previous suggestions that direct epithelial invasion via the blood vessels is a major route of infection for FMD [5]. In this study, TUNEL-positive labeling indicating apoptosis was prominently and widely observed in the lamellar of the upper layer of the stratum spinosum in the tongue and skin from the coronet to heel before the appearance of cellular injury in FMDV-infected pigs. During this stage, the viral antigen was restricted to the surface of the stratum spinosum. Previous reports indicate that FMD infection induces cell death by apoptosis in susceptible animals [15, 18]. However, detailed studies describing the interaction between FMDV infection and TUNEL reaction are limited [18]. Our findings here suggest that the apoptotic process might start simultaneously after cellular infection with FMDV. A previous study reported that epithelial lesions form in the dermal papillae and surrounding basal keratinocytes at the periphery of vesicles, and these regions were considered facilitative for vesiculation [5]. Similar findings were observed in the skin of the coronet and heel in this study. However, our results suggest that the vesiculation in the tongue originated from the necrosis of individual prickle cells in the middle to bottom layer of the stratum spinosum along the midline of the dorsal epithelium. Unfortunately, the histological finding showing the evidence that ulceration in the tongue was developed after the process of the vesiculation was not detected in this study. Further studies are needed to clarify the detailed mechanism of FMDV-lesion development in each tissue in pigs.
Virus isolation and RT-PCR analysis of tissue samples from pigs infected with FMDV O/JPN/2010 confirmed that the intraorally inoculated FMDV primarily infected the oropharyngeal tonsil and laryngopharynx at 1 dpi, and subsequently disseminated to multi-systemic organs at 3 dpi. Distribution of lesions and the virus in pigs in the early stages of FMDV infection observed in this study support previous findings [2, 4, 19, 22, 30, 32, 33]. Pigs have three oropharyngeal tonsils (the tonsil of the soft palate, paraepiglottic tonsil and lingual tonsil) and two nasopharyngeal tonsils (the pharyngeal tonsil and tubal tonsil) [20]. In this study, four (the tonsil of the soft palate, paraepiglottic tonsil, lingual tonsil and pharyngeal tonsil) of the five tonsils were collected and examined for histology and IHC. We found no histological lesions or viral antigen in the pharyngeal tonsil. In cattle, the nasopharynx is considered an important primary infectious and replication site for the FMDV [6,7,8, 29]. Although we isolated the virus from the nasopharynx of two of three pigs at 3 dpi, we did not detect the virus antigen in the nasopharynx of any of the examined pigs. While we confirmed the occurrence of direct viral invasion via epithelial cells in the tonsil by IHC, we found no histological lesions in the tonsil, soft palate or laryngopharynx despite the presence of significant amounts of virus. There are currently no reports to have conducted detailed histological examinations, including the distribution of the immunohistochemically-labeled viral antigen associated with the presence of the virus and virus RNA across the entire body of pigs infected with FMDV. Virus titer in multi-systemic organs of pigs experimentally infected with FMDV O/Taiwan/97 [19] and FMDV A24 Cruztiro [33] have been reported. These studies isolated FMDV from all lymph nodes examined at 2 dpi at high titers. While we also isolated FMDV from lymph nodes, we found no evidence of histological lesions or viral antigen in the lymph nodes. Our IHC results were not consistent with our virus isolation and RT-PCR findings for some of the examined organs including the trachea, lung, spleen and mandibular gland. The detailed mechanisms underlying which infected cells are only used for replication or passage of the virus and which undergo cell death after replication of the virus in host animals remains unclear. Viral RNA was not detected by RT-PCR from the tongue and skin of the coronet where showed IHC positive in all three pigs on 1 dpi and this result may have come about by the collection of isolation samples from the tissue, where it was later found that there were no or minimal antigen. In this study, the virus was isolated at 3 dpi from all examined organs in pig No. 4, which had severe bronchopneumonia. We also detected high titers of virus shedding from saliva, nasal swab samples and vesicles on the skin of the coronet of pig No. 4. These results suggest that there is a risk that if FMDV infects diseased pigs, higher titers of FMDV may be distributed across the entire body of pigs and markedly shed from saliva, nasal discharge and vesicles compared to FMDV infection in healthy pigs.
We confirmed that for rapid detection of the FMDV in the early stages of infection, samples should be collected from saliva, nasal swabs and the vesicular epidermis of the foot. The FMDV antigen was prominent in the vesicular epidermis of the foot in all examined pigs and high titers of the virus were isolated from two of three pigs at 3 dpi. The skin of the coronet sample from pig No. 3 was collected from the site of a ruptured vesicle on the coronet of the right forefoot, and was negative for virus isolation. In contrast, the skin of the coronet sample from pigs No. 4 and 7 were collected from the site of a vesicle before rupture and a normal-looking region on the coronet of the right forefoot, respectively, and showed high virus titers. In IHC, the ulcerative lesion of the ruptured vesicle was negative for the FMDV antigen, although the vesicular epidermis showed prominent FMDV antigen staining. These results suggest that the vesicular epidermis is a better FMD diagnostic sample than ulcerative lesions because the FMDV is not always isolated from ulcerative lesions after vesicle ruptured.
Although no gross lesions were observed in the heart of any pigs at necropsy, myocarditis with prominent viral antigen label was histologically evident in all three pigs at 3 dpi. A few reports have described the histolopathology of cardiac lesions in FMD [15, 24, 26, 31]. We observed degenerated cardiomyocytes with viral antigen in the absence of inflammation at 1 dpi, suggesting that tissue damage in the heart may arise as a direct consequence of virus replication rather than the host’s response to infection. Our results suggest that FMDV O/JPN/2010 can enter cardiomuscular tissue as early as it enters the epidermis of the skin of the coronet after intraoral inoculation, and causes multitrophic infection of the epithelium/epidermis and muscle. Multitrophic FMDV infection has been described in cattle, mule deer [4] and lambs [15, 26]. However, there is currently no detailed histological investigation of multitrophic FMDV infections in pigs. While the cause of fatalities associated with myocarditis in young ruminants and pigs infected with FMD has been reported [31], none of the pigs in this study were in a fatal condition at 3 dpi, even though they exhibited severe myocarditis associated with the viral antigen. Further studies are necessary to clarify FMD pathogenesis in the heart and the detailed mechanisms of cardiomuscular entry by the virus that leads to myocarditis in pigs. We showed that myocarditis with viral antigen was induced via oral inoculation of FMDV O/JPN/2010, demonstrating the value of this experimental infection as a model for the study of FMD pathogenesis in the heart and the causes of fatalities due to FMD.
Acknowledgments
We thank Mr. M. Kobayashi and Miss M. Shimada for preparing the pathology sections and Mrs. H. Kimura, M. Kanda, S. Sato, K. Ishii, T. Nakamura and S. Mizumura for their care of the animals. This study was partly supported by a research project for improving food and animal health by the Ministry of Agriculture, Forestry and Fisheries of Japan.
REFERENCES
- 1.Anonymous Chapter 2.1.5. Foot and mouth disease. Manual of Diagnostic Tests and vaccines for Terrestrial Animals 2015. http://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/2.01.05_FMD.pdf [accessed on October 31, 2017].
- 2.Alexandersen S., Oleksiewicz M. B., Donaldson A. I.2001. The early pathogenesis of foot-and-mouth disease in pigs infected by contact: a quantitative time-course study using TaqMan RT-PCR. J. Gen. Virol. 82: 747–755. doi: 10.1099/0022-1317-82-4-747 [DOI] [PubMed] [Google Scholar]
- 3.Alexandersen S., Zhang Z., Donaldson A. I., Garland A. J.2003. The pathogenesis and diagnosis of foot-and-mouth disease. J. Comp. Pathol. 129: 1–36. doi: 10.1016/S0021-9975(03)00041-0 [DOI] [PubMed] [Google Scholar]
- 4.Arzt J., Baxt B., Grubman M. J., Jackson T., Juleff N., Rhyan J., Rieder E., Waters R., Rodriguez L. L.2011. The pathogenesis of foot-and-mouth disease II: viral pathways in swine, small ruminants, and wildlife; myotropism, chronic syndromes, and molecular virus-host interactions. Transbound. Emerg. Dis. 58: 305–326. doi: 10.1111/j.1865-1682.2011.01236.x [DOI] [PubMed] [Google Scholar]
- 5.Arzt J., Gregg D. A., Clavijo A., Rodriguez L. L.2009. Optimization of immunohistochemical and fluorescent antibody techniques for localization of Foot-and-mouth disease virus in animal tissues. J. Vet. Diagn. Invest. 21: 779–792. doi: 10.1177/104063870902100604 [DOI] [PubMed] [Google Scholar]
- 6.Arzt J., Pacheco J. M., Rodriguez L. L.2010. The early pathogenesis of foot-and-mouth disease in cattle after aerosol inoculation. Identification of the nasopharynx as the primary site of infection. Vet. Pathol. 47: 1048–1063. doi: 10.1177/0300985810372509 [DOI] [PubMed] [Google Scholar]
- 7.Arzt J., Pacheco J. M., Smoliga G. R., Tucker M. T., Bishop E., Pauszek S. J., Hartwig E. J., de los Santos T., Rodriguez L. L.2014. Foot-and-mouth disease virus virulence in cattle is co-determined by viral replication dynamics and route of infection. Virology 452-453: 12–22. doi: 10.1016/j.virol.2014.01.001 [DOI] [PubMed] [Google Scholar]
- 8.Arzt J., Pacheco J. M., Stenfeldt C., Rodriguez L. L.2017. Pathogenesis of virulent and attenuated foot-and-mouth disease virus in cattle. Virol. J. 14: 89. doi: 10.1186/s12985-017-0758-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bumbasirević V., Skaro-Milić A., Mircić A., Djuricić B.1995. Apoptosis induced by microtubule disrupting drugs in normal murine thymocytes in vitro. Scanning Microsc. 9: 509–516, discussion 516–518. [PubMed] [Google Scholar]
- 10.Donaldson A. I.1997. Foot-and-mouth disease in Taiwan. Vet. Rec. 140: 407. [PubMed] [Google Scholar]
- 11.Durand S., Murphy C., Zhang Z., Alexandersen S.2008. Epithelial distribution and replication of foot-and-mouth disease virus RNA in infected pigs. J. Comp. Pathol. 139: 86–96. doi: 10.1016/j.jcpa.2008.05.004 [DOI] [PubMed] [Google Scholar]
- 12.Fukai K., Morioka K., Yoshida K.2011. An experimental infection in pigs using a foot-and-mouth disease virus isolated from the 2010 epidemic in Japan. J. Vet. Med. Sci. 73: 1207–1210. doi: 10.1292/jvms.11-0063 [DOI] [PubMed] [Google Scholar]
- 13.Fukai K., Nishi T., Shimada N., Morioka K., Yamada M., Yoshida K., Sakamoto K., Kitano R., Yamazoe R., Yamakawa M.2017. Experimental infections using the foot-and-mouth disease virus O/JPN/2010 in animals administered a vaccine preserved for emergency use in Japan. J. Vet. Med. Sci. 79: 128–136. doi: 10.1292/jvms.16-0482 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fukai K., Yamada M., Morioka K., Ohashi S., Yoshida K., Kitano R., Yamazoe R., Kanno T.2015. Dose-dependent responses of pigs infected with foot-and-mouth disease virus O/JPN/2010 by the intranasal and intraoral routes. Arch. Virol. 160: 129–139. doi: 10.1007/s00705-014-2239-4 [DOI] [PubMed] [Google Scholar]
- 15.Gulbahar M. Y., Davis W. C., Guvenc T., Yarim M., Parlak U., Kabak Y. B.2007. Myocarditis associated with foot-and-mouth disease virus type O in lambs. Vet. Pathol. 44: 589–599. doi: 10.1354/vp.44-5-589 [DOI] [PubMed] [Google Scholar]
- 16.Knowles N. J., He J., Shang Y., Wadsworth J., Valdazo-González B., Onosato H., Fukai K., Morioka K., Yoshida K., Cho I. S., Kim S. M., Park J. H., Lee K. N., Luk G., Borisov V., Scherbakov A., Timina A., Bold D., Nguyen T., Paton D. J., Hammond J. M., Liu X., King D. P.2012. Southeast Asian foot-and-mouth disease viruses in Eastern Asia. Emerg. Infect. Dis. 18: 499–501. doi: 10.3201/eid1803.110908 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Knowles N. J., Samuel A. R., Davies P. R., Kitching R. P., Donaldson A. I.2001. Outbreak of foot-and-mouth disease virus serotype O in the U.K. caused by a pandemic strain. Vet. Rec. 148: 258–259. [PubMed] [Google Scholar]
- 18.Ku B. K., Kim S. B., Moon O. K., Lee S. J., Lee J. H., Lyoo Y. S., Kim H. J., Sur J. H.2005. Role of apoptosis in the pathogenesis of Asian and South American foot-and-mouth disease viruses in swine. J. Vet. Med. Sci. 67: 1081–1088. doi: 10.1292/jvms.67.1081 [DOI] [PubMed] [Google Scholar]
- 19.Lee S. H., Jong M. H., Huang T. S., Lin Y. L., Wong M. L., Liu C. I., Chang T. J.2009. Pathology and viral distributions of the porcinophilic foot-and-mouth disease virus strain (O/Taiwan/97) in experimentally infected pigs. Transbound. Emerg. Dis. 56: 189–201. doi: 10.1111/j.1865-1682.2009.01079.x [DOI] [PubMed] [Google Scholar]
- 20.Liu Z., Yu Q., Li P., Yang Q.2012. Histological and ultrastructural examinations of porcine tonsils. Anat. Rec. (Hoboken) 295: 686–690. doi: 10.1002/ar.21534 [DOI] [PubMed] [Google Scholar]
- 21.Morioka K., Fukai K., Yoshida K., Yamazoe R., Onozato H., Ohashi S., Tsuda T., Sakamoto K.2009. Neutralizing monoclonal antibody sandwich liquid-phase blocking enzyme-linked immunosorbent assay for detection of Foot-and-mouth disease virus type O antibodies. J. Vet. Diagn. Invest. 21: 499–503. doi: 10.1177/104063870902100411 [DOI] [PubMed] [Google Scholar]
- 22.Murphy C., Bashiruddin J. B., Quan M., Zhang Z., Alexandersen S.2010. Foot-and-mouth disease viral loads in pigs in the early, acute stage of disease. Vet. Rec. 166: 10–14. doi: 10.1136/vr.b5583 [DOI] [PubMed] [Google Scholar]
- 23.Muroga N., Hayama Y., Yamamoto T., Kurogi A., Tsuda T., Tsutsui T.2012. The 2010 foot-and-mouth disease epidemic in Japan. J. Vet. Med. Sci. 74: 399–404. doi: 10.1292/jvms.11-0271 [DOI] [PubMed] [Google Scholar]
- 24.Oem S. K., Yeh M. T., McKenna T. S., Hayes J. R., Rieder E., Giuffre A. C., Robida J. M., Lee K. N., Cho I. S., Fang X., Joo Y. S., Park J. H.Pathogenic characteristics of the Korean 2002. Isolate of foot-and-mouth disease virus serotype O in pigs and cattle. J. Comp. Pathol. 138: 204–214. [DOI] [PubMed] [Google Scholar]
- 25.Onozato H., Fukai K., Kitano R., Yamazoe R., Morioka K., Yamada M., Ohashi S., Yoshida K., Kanno T.2014. Experimental infection of cattle and goats with a foot-and-mouth disease virus isolate from the 2010 epidemic in Japan. Arch. Virol. 159: 2901–2908. doi: 10.1007/s00705-014-2135-y [DOI] [PubMed] [Google Scholar]
- 26.Ryan E., Horsington J., Durand S., Brooks H., Alexandersen S., Brownlie J., Zhang Z.2008. Foot-and-mouth disease virus infection in young lambs: pathogenesis and tissue tropism. Vet. Microbiol. 127: 258–274. doi: 10.1016/j.vetmic.2007.08.029 [DOI] [PubMed] [Google Scholar]
- 27.Ryoo S., Kim T., Nah J. J., Sagong M. G., Lee S., Lee K. N., Ko Y. J., Park J. H., Lee M. H., Wee S. H., Tark D., Ku B. K.2017. Complete genome sequence of a foot-and-mouth disease virus of serotype O isolated from Gimje, Republic of Korea, in 2016. Genome Announc. 5: e01694–e16. doi: 10.1128/genomeA.01694-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sakamoto K., Kanno T., Yamakawa M., Yoshida K., Yamazoe R., Murakami Y.2002. Isolation of foot-and-mouth disease virus from Japanese black cattle in Miyazaki Prefecture, Japan, 2000. J. Vet. Med. Sci. 64: 91–94. doi: 10.1292/jvms.64.91 [DOI] [PubMed] [Google Scholar]
- 29.Stenfeldt C., Eschbaumer M., Pacheco J. M., Rekant S. I., Rodriguez L. L., Arzt J.2015. Pathogenesis of primary foot-and-mouth disease virus infection in the nasopharynx of vaccinated and non-vaccinated cattle. PLOS ONE 10: e0143666. doi: 10.1371/journal.pone.0143666 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Stenfeldt C., Diaz-San Segundo F., de Los Santos T., Rodriguez L. L., Arzt J.2016. The pathogenesis of foot-and-mouth disease in pigs. Front. Vet. Sci. 3: 41. doi: 10.3389/fvets.2016.00041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Stenfeldt C., Pacheco J. M., Borca M. V., Rodriguez L. L., Arzt J.2014. Morphologic and phenotypic characteristics of myocarditis in two pigs infected by foot-and mouth disease virus strains of serotypes O or A. Acta Vet. Scand. 56: 42. doi: 10.1186/s13028-014-0042-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Stenfeldt C., Pacheco J. M., Rodriguez L. L., Arzt J.2014. Infection dynamics of foot-and-mouth disease virus in pigs using two novel simulated-natural inoculation methods. Res. Vet. Sci. 96: 396–405. doi: 10.1016/j.rvsc.2014.01.009 [DOI] [PubMed] [Google Scholar]
- 33.Stenfeldt C., Pacheco J. M., Rodriguez L. L., Arzt J.2014. Early events in the pathogenesis of foot-and-mouth disease in pigs; identification of oropharyngeal tonsils as sites of primary and sustained viral replication. PLOS ONE 9: e106859. doi: 10.1371/journal.pone.0106859 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Uzal F. A., Plattner B. L., Hostetter J. M.2016. Alimentary System. pp. 1–257. In: Jubb, Kennedy and Palmer’s Pathology of Domestic Animals Vol. 2, 6th ed. (Grant Maxie, M. ed.), Elsevier, St. Louis. [Google Scholar]