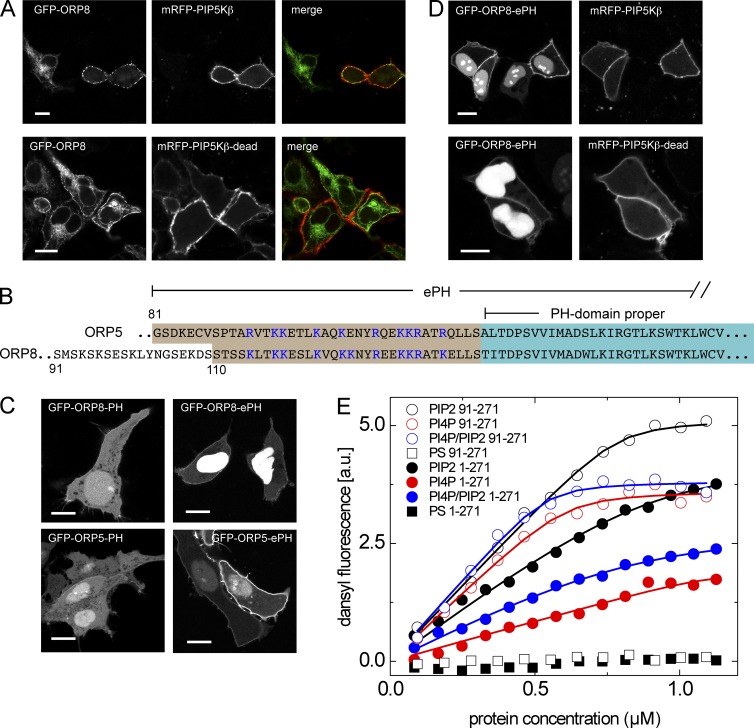
Figure 3.
Extra PI(4,5)P2 production increases the PM engagement of ORP8. (A) Representative live cell images showing intracellular localization of ORP8 under overexpression of PIP5Kβ. HEK293-AT1 cells were cotransfected with GFP-tagged ORP5 and wild-type or kinase-dead (dead) mRFP-tagged PIP5Kβ. After 1 d of transfection, cells were observed with confocal microscopy. Bars, 10 µm. (B) Comparison of amino acid sequences of N-terminal PH domains between ORP5 and ORP8. ORP5 and ORP8 share strictly defined PH domains (blue box). Conserved polybasic residues (blue) precede strict PH domains in both ORP proteins. (C) Representative live cell images showing localization of PH domains with or without adjacent polybasic residues. HEK293-AT1 cells were transfected with GFP-tagged ORP8-PH, ORP8-ePH, ORP5-PH, or ORP5-ePH. After 1 d, localization of PH domains was observed with confocal microscopy. Bars, 10 µm. (D) Representative live-cell images displaying effect of PIP5K overexpression in PM engagement of ORP8-ePH. HEK293-AT1 cells were cotransfected with GFP-tagged ORP8-ePH and mRFP-tagged PIP5Kβ (wild-type or kinase-dead). After 1 d, cells were observed with confocal microscopy. Bars, 10 µm. (E) Dependence of the Förster resonance energy transfer of ORP8 PH domain tryptophan residues to dansyl-labeled liposomes (monitored by dansyl fluorescence intensity) on the concentration of the PH domains. Solid and open circles, 1–284 and 91–284 constructs of the ORP8 PH domain. Liposomes contained 5 mol % of PI(4,5)P2 (black), 5 mol % of PI4P (red), or 2.5 mol % of PI4P plus 2.5 mol % of PI(4,5)P2 (blue). Squares, control liposomes bearing the same net charge from the negatively charged PS but no phosphoinositides. One representative result is shown from three separate experiments that gave identical results but differed in absolute fluorescent intensity values.